Label: LOXAPINE capsule
-
NDC Code(s):
0527-1394-01,
0527-1394-10,
0527-1395-01,
0527-1395-10, view more0527-1396-01, 0527-1396-10, 0527-1397-01, 0527-1397-10
- Packager: Lannett Company, Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated May 28, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
-
BOXED WARNING
(What is this?)
WARNING
Increased Mortality in Elderly Patients with Dementia-Related Psychosis
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. Analyses of seventeen placebo-controlled trials (modal duration of 10 weeks), largely in patients taking atypical antipsychotic drugs, revealed a risk of death in drug-treated patients of between 1.6 to 1.7 times the risk of death in placebo-treated patients. Over the course of a typical 10-week controlled trial, the rate of death in drug-treated patients was about 4.5%, compared to a rate of about 2.6% in the placebo group. Although the causes of death were varied, most of the deaths appeared to be either cardiovascular (e.g., heart failure, sudden death) or infectious (e.g., pneumonia) in nature. Observational studies suggest that, similar to atypical antipsychotic drugs, treatment with conventional antipsychotic drugs may increase mortality. The extent to which the findings of increased mortality in observational studies may be attributed to the antipsychotic drug as opposed to some characteristic(s) of the patients is not clear. Loxapine is not approved for the treatment of patients with dementia-related psychosis (see WARNINGS).
-
DESCRIPTION
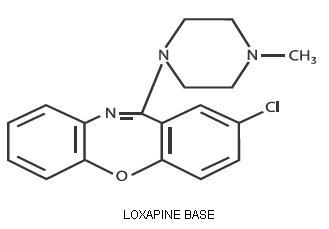
Loxapine, a dibenzoxazepine compound, represents a subclass of tricyclic antipsychotic agents, chemically distinct from the thioxanthenes, butyrophenones, and phenothiazines. Chemically, it is 2-Chloro-11-(4-methyl-1-piperazinyl)dibenz[b,f][1,4]oxazepine. It is present as the succinate salt.
Each capsule for oral administration, contains loxapine succinate, USP 6.8, 13.6, 34.0 or 68.1 mg equivalent to 5, 10, 25 or 50 mg of loxapine base respectively. It also contains the following inactive ingredients: anhydrous lactose, colloidal silicon dioxide, gelatin, magnesium stearate, polacrilin potassium, sodium lauryl sulfate, talc, titanium dioxide, D&C Yellow #10 and FD&C Blue #1. Additionally, the 5 mg capsule contains D&C Red #33, the 10 mg capsule contains D&C Red #33 and D&C Red #28, and the 25 mg capsule contains FD&C Yellow #6.
In addition, the black imprinting ink contains shellac glace in ethanol, iron oxide black, n-butyl alcohol, propylene glycol, FD&C Blue #2/indigo carmine aluminum lake, FD&C Red #40/Allurea Red AC aluminum lake, FD&C Blue #1/brilliant Blue FCF aluminum lake, D&C Yellow #10 aluminum lake, SDA 3A alcohol, and methanol. The white imprinting ink contains pharmaceutical glaze in SD-45, titanium dioxide, isopropyl alcohol, ammonium hydroxide, propylene glycol, n-butyl alcohol and simethicone.
-
CLINICAL PHARMACOLOGY
Pharmacodynamics
Pharmacologically, loxapine is an antipsychotic for which the exact mode of action has not been established. However, changes in the level of excitability of subcortical inhibitory areas have been observed in several animal species in association with such manifestations of tranquilization as calming effects and suppression of aggressive behavior.
In normal human volunteers, signs of sedation were seen within 20 to 30 minutes after administration, were most pronounced within one and one-half to three hours, and lasted through 12 hours. Similar timing of primary pharmacologic effects was seen in animals.
Absorption, Distribution, Metabolism, and Excretion
Absorption of loxapine following oral or parenteral administration is virtually complete. The drug is removed rapidly from the plasma and distributed in tissues. Animal studies suggest an initial preferential distribution in lungs, brain, spleen, heart, and kidney. Loxapine is metabolized extensively and is excreted mainly in the first 24 hours. Metabolites are excreted in the urine in the form of conjugates and in the feces unconjugated.
- INDICATIONS AND USAGE
- CONTRAINDICATIONS
-
WARNINGS
Increased Mortality in Elderly Patients with Dementia-Related Psychosis
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. Loxapine is not approved for the treatment of patients with dementia-related psychosis (see BOXED WARNING).
Tardive Dyskinesia
Tardive dyskinesia, a syndrome consisting of potentially irreversible, involuntary, dyskinetic movements, may develop in patients treated with antipsychotic drugs. Although the prevalence of the syndrome appears to be highest among the elderly, especially elderly women, it is impossible to rely upon prevalence estimates to predict, at the inception of antipsychotic treatment, which patients are likely to develop the syndrome. Whether antipsychotic drug products differ in their potential to cause tardive dyskinesia is unknown.
Both the risk of developing the syndrome and the likelihood that it will become irreversible are believed to increase as the duration of treatment and the total cumulative dose of antipsychotic drugs administered to the patient increase. However, the syndrome can develop, although much less commonly, after relatively brief treatment periods at low doses.
There is no known treatment for established cases of tardive dyskinesia, although the syndrome may remit, partially or completely, if antipsychotic treatment is withdrawn. Antipsychotic treatment itself, however, may suppress (or partially suppress) the signs and symptoms of the syndrome and thereby may possibly mask the underlying disease process. The effect that symptomatic suppression has upon the long-term course of the syndrome is unknown.
Given these considerations, antipsychotics should be prescribed in a manner that is most likely to minimize the occurrence of tardive dyskinesia. Chronic antipsychotic treatment should generally be reserved for patients who suffer from a chronic illness that, 1) is known to respond to antipsychotic drugs, and 2) for whom alternative, equally effective, but potentially less harmful treatments are not available or appropriate. In patients who do require chronic treatment, the smallest dose and the shortest duration of treatment producing a satisfactory clinical response should be sought. The need for continued treatment should be reassessed periodically.
If signs and symptoms of tardive dyskinesia appear in a patient on antipsychotics, drug discontinuation should be considered. However, some patients may require treatment despite the presence of the syndrome. (See ADVERSE REACTIONS and Information for Patients sections).
Neuroleptic Malignant Syndrome (NMS)
A potentially fatal symptom complex sometimes referred to as Neuroleptic Malignant Syndrome (NMS) has been reported in association with antipsychotic drugs. Clinical manifestations of NMS are hyperpyrexia, muscle rigidity, altered mental status, and evidence of autonomic instability (irregular pulse or blood pressure, tachycardia, diaphoresis, and cardiac dysrhythmias).
The diagnostic evaluation of patients with this syndrome is complicated. In arriving at a diagnosis, it is important to identify cases where the clinical presentation includes both serious medical illness (e.g., pneumonia, systemic infection, etc.) and untreated or inadequately treated extrapyramidal signs and symptoms (EPS). Other important considerations in the differential diagnosis include central anticholinergic toxicity, heat stroke, drug fever, and primary central nervous system (CNS) pathology.
The management of NMS should include: 1) immediate discontinuation of antipsychotic drugs and other drugs not essential to concurrent therapy, 2) intensive symptomatic treatment and medical monitoring, and 3) treatment of any concomitant serious medical problems for which specific treatments are available. There is no general agreement about specific pharmacological treatment regimens for uncomplicated NMS.
If a patient requires antipsychotic drug treatment after recovery from NMS, the potential reintroduction of drug therapy should be carefully considered. The patient should be carefully monitored, since recurrences of NMS have been reported.
Loxapine, like other antipsychotics, may impair mental and/or physical abilities, especially during the first few days of therapy. Therefore, ambulatory patients should be warned about activities requiring alertness (e.g., operating vehicles or machinery) and about concomitant use of alcohol and other CNS depressants.
Loxapine has not been evaluated for the management of behavioral complications in patients with mental retardation, and therefore, it cannot be recommended.
Falls
Loxapine may cause somnolence, postural hypotension, motor and sensory instability, which may lead to falls and, consequently, fractures or other injuries. For patients with diseases, conditions, or medications that could exacerbate these effects, complete fall risk assessments when initiating antipsychotic treatment and recurrently for patients on long-term antipsychotic therapy.
-
PRECAUTIONS
Leukopenia, Neutropenia and Agranulocytosis
In clinical trial and postmarketing experience, events of leukopenia/neutropenia and agranulocytosis have been reported temporally related to antipsychotic agents.
Possible risk factors for leukopenia/neutropenia include preexisting low white blood cell count (WBC) and history of drug induced leukopenia/neutropenia. Patients with a preexisting low WBC or a history of drug induced leukopenia/neutropenia should have their complete blood count (CBC) monitored frequently during the first few months of therapy and should discontinue Loxapine Succinate Capsules, USP at the first sign of a decline in WBC in the absence of other causative factors.
Patients with neutropenia should be carefully monitored for fever or other symptoms or signs of infection and treated promptly if such symptoms or signs occur. Patients with severe neutropenia (absolute neutrophil count <1000/mm3) should discontinue Loxapine Succinate Capsules, USP and have their WBC followed until recovery.
General
Loxapine should be used with extreme caution in patients with a history of convulsive disorders since it lowers the convulsive threshold. Seizures have been reported in patients receiving loxapine at antipsychotic dose levels, and may occur in epileptic patients even with maintenance of routine anticonvulsant drug therapy.
Loxapine has an antiemetic effect in animals. Since this effect may also occur in man, loxapine may mask signs of overdosage of toxic drugs and may obscure conditions such as intestinal obstruction and brain tumor.
Loxapine should be used with caution in patients with cardiovascular disease. Increased pulse rates have been reported in the majority of patients receiving antipsychotic doses; transient hypotension has been reported. In the presence of severe hypotension requiring vasopressor therapy, the preferred drugs may be norepinephrine or angiotensin. Usual doses of epinephrine may be ineffective because of inhibition of its vasopressor effect by loxapine.
The possibility of ocular toxicity from loxapine cannot be excluded at this time. Therefore, careful observation should be made for pigmentary retinopathy and lenticular pigmentation since these have been observed in some patients receiving certain other antipsychotic drugs for prolonged periods.
Because of possible anticholinergic action, the drug should be used cautiously in patients with glaucoma or a tendency to urinary retention, particularly with concomitant administration of anticholinergic-type antiparkinson medication.
Experience to date indicates the possibility of a slightly higher incidence of extrapyramidal effects following intramuscular administration than normally anticipated with oral formulations. The increase may be attributable to higher plasma levels following intramuscular injection.
Antipsychotic drugs elevate prolactin levels; the elevation persists during chronic administration. Tissue culture experiments indicate that approximately one-third of human breast cancers are prolactin-dependent in vitro, a factor of potential importance if the prescription of these drugs is contemplated in a patient with a previously detected breast cancer. Although disturbances such as galactorrhea, amenorrhea, gynecomastia, and impotence have been reported, the clinical significance of elevated serum prolactin levels is unknown for most patients. An increase in mammary neoplasms has been found in rodents after chronic administration of antipsychotic drugs. Neither clinical studies nor epidemiologic studies conducted to date, however, have shown an association between chronic administration of these drugs and mammary tumorigenesis; the available evidence is considered too limited to be conclusive at this time.
Information for Patients
Given the likelihood that some patients exposed chronically to antipsychotics will develop tardive dyskinesia, it is advised that all patients in whom chronic use is contemplated be given, if possible, full information about this risk. The decision to inform patients and/or their guardians must obviously take into account the clinical circumstances and the competency of the patient to understand the information provided.
Drug Interactions
There have been rare reports of significant respiratory depression, stupor and/or hypotension with the concomitant use of loxapine and lorazepam.
The risk of using loxapine in combination with CNS-active drugs has not been systematically evaluated. Therefore, caution is advised if the concomitant administration of loxapine and CNS-active drugs is required.
Pregnancy
Non-teratogenic Effects
Neonates exposed to antipsychotic drugs, during the third trimester of pregnancy are at risk for extrapyramidal and/or withdrawal symptoms following delivery. There have been reports of agitation, hypertonia, hypotonia, tremor, somnolence, respiratory distress and feeding disorder in these neonates. These complications have varied in severity; while in some cases symptoms have been self-limited, in other cases neonates have required intensive care unit support and prolonged hospitalization.
Loxapine Succinate should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Safe use of loxapine during pregnancy or lactation has not been established; therefore, its use in pregnancy, in nursing mothers, or in women of childbearing potential requires that the benefits of treatment be weighed against the possible risks to mother and child. No embryotoxicity or teratogenicity was observed in studies in rats, rabbits, or dogs although, with the exception of one rabbit study, the highest dosage was only two times the maximum recommended human dose and in some studies it was below this dose. Perinatal studies have shown renal papillary abnormalities in offspring of rats treated from mid-pregnancy with doses of 0.6 and 1.8 mg/kg, doses which approximate the usual human dose but which are considerably below the maximum recommended human dose.
-
ADVERSE REACTIONS
CNS Effects: Manifestations of adverse effects on the central nervous system, other than extrapyramidal effects, have been seen infrequently. Drowsiness, usually mild, may occur at the beginning of therapy or when dosage is increased. It usually subsides with continued loxapine therapy. The incidence of sedation has been less than that of certain aliphatic phenothiazines and slightly more than the piperazine phenothiazines. Dizziness, faintness, staggering gait, shuffling gait, muscle twitching, weakness, insomnia, agitation, tension, seizures, akinesia, slurred speech, numbness, and confusional states have been reported. Neuroleptic malignant syndrome (NMS) has been reported (see WARNINGS).
Extrapyramidal Symptoms - Neuromuscular (extrapyramidal) reactions during the administration of loxapine have been reported frequently, often during the first few days of treatment. In most patients, these reactions involved parkinsonian-like symptoms such as tremor, rigidity, excessive salivation, and masked facies. Akathisia (motor restlessness) also has been reported relatively frequently. These symptoms are usually not severe and can be controlled by reduction of loxapine dosage or by administration of antiparkinson drugs in usual dosage.
Dystonia - Class effect: Symptoms of dystonia, prolonged abnormal contractions of muscle groups, may occur in susceptible individuals during the first few days of treatment. Dystonic symptoms include: spasm of the neck muscles, sometimes progressing to tightness of the throat, swallowing difficulty, difficulty breathing, and/or protrusion of the tongue. While these symptoms can occur at low doses, they occur more frequently and with greater severity with high potency and at higher doses of first generation antipsychotic drugs. An elevated risk of acute dystonia is observed in males and younger age groups.
Persistent Tardive Dyskinesia - As with all antipsychotic agents, tardive dyskinesia may appear in some patients on long-term therapy or may appear after drug therapy has been discontinued. The risk appears to be greater in elderly patients on high-dose therapy, especially females. The symptoms are persistent and in some patients appear to be irreversible. The syndrome is characterized by rhythmical involuntary movement of the tongue, face, mouth or jaw (e.g., protrusion of tongue, puffing of cheeks, puckering of mouth, chewing movements). Sometimes these may be accompanied by involuntary movements of extremities.
There is no known effective treatment for tardive dyskinesia; antiparkinson agents usually do not alleviate the symptoms of this syndrome. It is suggested that all antipsychotic agents be discontinued if these symptoms appear. Should it be necessary to reinstitute treatment, or increase the dosage of the agent, or switch to a different antipsychotic agent, the syndrome may be masked. It has been suggested that fine vermicular movements of the tongue may be an early sign of the syndrome, and if the medication is stopped at that time the syndrome may not develop.
Cardiovascular Effects: Tachycardia, hypotension, hypertension, orthostatic hypotension, lightheadedness, and syncope have been reported.
A few cases of ECG changes similar to those seen with phenothiazines have been reported. It is not known whether these were related to loxapine administration.
Hematologic: Rarely, agranulocytosis, thrombocytopenia, leukopenia.
Skin: Dermatitis, edema (puffiness of face), pruritus, rash, alopecia, and seborrhea have been reported with loxapine.
Anticholinergic Effects: Dry mouth, nasal congestion, constipation, blurred vision, urinary retention, and paralytic ileus have occurred.
Gastrointestinal: Nausea and vomiting have been reported in some patients. Hepatocellular injury (i.e., SGOT/SGPT elevation) has been reported in association with loxapine administration and rarely, jaundice and/or hepatitis questionably related to loxapine treatment.
Other Adverse Reactions: Weight gain, weight loss, dyspnea, ptosis, hyperpyrexia, flushed facies, headache, paresthesia, and polydipsia have been reported in some patients. Rarely, galactorrhea, amenorrhea, gynecomastia, and menstrual irregularity of uncertain etiology have been reported.
-
OVERDOSAGE
Signs and symptoms of overdosage will depend on the amount ingested and individual patient tolerance. As would be expected from the pharmacologic actions of the drug, the clinical findings may range from mild depression of the CNS and cardiovascular systems to profound hypotension, respiratory depression, and unconsciousness. The possibility of occurrence of extrapyramidal symptoms and/or convulsive seizures should be kept in mind. Renal failure following loxapine overdosage has also been reported.
The treatment of overdosage is essentially symptomatic and supportive. Early gastric lavage and extended dialysis might be expected to be beneficial. Centrally-acting emetics may have little effect because of the antiemetic action of loxapine. In addition, emesis should be avoided because of the possibility of aspiration of vomitus. Avoid analeptics, such as pentylenetetrazol, which may cause convulsions. Severe hypotension might be expected to respond to the administration of norepinephrine or phenylephrine. EPINEPHRINE SHOULD NOT BE USED SINCE ITS USE IN A PATIENT WITH PARTIAL ADRENERGIC BLOCKADE MAY FURTHER LOWER THE BLOOD PRESSURE. Severe extrapyramidal reactions should be treated with anticholinergic antiparkinson agents or diphenhydramine hydrochloride, and anticonvulsant therapy should be initiated as indicated. Additional measures include oxygen and intravenous fluids.
-
DOSAGE AND ADMINISTRATION
Loxapine Capsules, USP are administered, usually in divided doses, two to four times a day. Daily dosage (in terms of base equivalents) should be adjusted to the individual patient's needs as assessed by the severity of symptoms and previous history of response to antipsychotic drugs.
Oral Administration
Initial dosage of 10 mg twice daily is recommended, although in severely disturbed patients initial dosage up to a total of 50 mg daily may be desirable. Dosage should then be increased fairly rapidly over the first seven to ten days until there is effective control of symptoms of schizophrenia. The usual therapeutic and maintenance range is 60 mg to 100 mg daily. However, as with other drugs used to treat schizophrenia, some patients respond to lower dosage and others require higher dosage for optimal benefit. Daily dosage higher than 250 mg is not recommended.
-
HOW SUPPLIED
Loxapine Capsules, USP are available in the following strengths:
Loxapine Succinate, USP 6.8 mg equivalent to 5 mg loxapine, opaque, with a dark green body and cap, imprinted with logo "LANNETT" on the cap and "1394" on the body are supplied as follows:
NDC 0527-1394-01 –Bottle of 100s
NDC 0527-1394-10 –Bottle of 1000sLoxapine Succinate, USP 13.6 mg equivalent to 10 mg loxapine, yellow opaque body and dark green opaque cap, imprinted with logo "LANNETT" on the cap and "1395" on the body are supplied as follows:
NDC 0527-1395-01 –Bottle of 100s
NDC 0527-1395-10 –Bottle of 1000sLoxapine Succinate, USP 34.0 mg equivalent to 25 mg loxapine, light green opaque body and dark green opaque cap, imprinted with logo "LANNETT" on the cap and "1396" on the body are supplied as follows:
NDC 0527-1396-01 –Bottle of 100s
NDC 0527-1396-10 –Bottle of 1000sLoxapine Succinate, USP 68.1 mg equivalent to 50 mg loxapine, light blue opaque body and dark green opaque cap, imprinted with logo "LANNETT" on the cap and "1397" on the body are supplied as follows:
NDC 0527-1397-01 –Bottle of 100s
NDC 0527-1397-10 –Bottle of 1000sStore at 20°-25°C (68°-77°F). [See USP controlled room temperature.] Dispense in a tight, child-resistant container.
Distributed by:
Lannett Company, Inc.
Philadelphia, PA 19136CIB70541E
Rev. 05/20
- PRINCIPAL DISPLAY PANEL - 5 mg Container Label
- PRINCIPAL DISPLAY PANEL - 10 mg Container Label
- PRINCIPAL DISPLAY PANEL - 25 mg Container Label
- PRINCIPAL DISPLAY PANEL - 50 mg Container Label
-
INGREDIENTS AND APPEARANCE
LOXAPINE
loxapine capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0527-1394 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LOXAPINE SUCCINATE (UNII: X59SG0MRYU) (LOXAPINE - UNII:LER583670J) LOXAPINE 5 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) GELATIN (UNII: 2G86QN327L) MAGNESIUM STEARATE (UNII: 70097M6I30) POLACRILIN POTASSIUM (UNII: 0BZ5A00FQU) SODIUM LAURYL SULFATE (UNII: 368GB5141J) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) D&C RED NO. 33 (UNII: 9DBA0SBB0L) SHELLAC (UNII: 46N107B71O) ALCOHOL (UNII: 3K9958V90M) ISOPROPYL ALCOHOL (UNII: ND2M416302) AMMONIA (UNII: 5138Q19F1X) BUTYL ALCOHOL (UNII: 8PJ61P6TS3) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) Product Characteristics Color green (Dark Green Opaque) Score no score Shape CAPSULE Size 18mm Flavor Imprint Code LANNETT;1394 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0527-1394-01 100 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 09/26/2011 2 NDC:0527-1394-10 1000 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 09/26/2011 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA090695 09/26/2011 LOXAPINE
loxapine capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0527-1395 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LOXAPINE SUCCINATE (UNII: X59SG0MRYU) (LOXAPINE - UNII:LER583670J) LOXAPINE 10 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) GELATIN (UNII: 2G86QN327L) MAGNESIUM STEARATE (UNII: 70097M6I30) POLACRILIN POTASSIUM (UNII: 0BZ5A00FQU) SODIUM LAURYL SULFATE (UNII: 368GB5141J) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) D&C RED NO. 33 (UNII: 9DBA0SBB0L) D&C RED NO. 28 (UNII: 767IP0Y5NH) SHELLAC (UNII: 46N107B71O) ALCOHOL (UNII: 3K9958V90M) ISOPROPYL ALCOHOL (UNII: ND2M416302) AMMONIA (UNII: 5138Q19F1X) BUTYL ALCOHOL (UNII: 8PJ61P6TS3) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) FERROSOFERRIC OXIDE (UNII: XM0M87F357) FD&C BLUE NO. 2 (UNII: L06K8R7DQK) ALUMINUM OXIDE (UNII: LMI26O6933) FD&C RED NO. 40 (UNII: WZB9127XOA) METHYL ALCOHOL (UNII: Y4S76JWI15) Product Characteristics Color green (Dark Green Opaque) , yellow (Yellow Opaque) Score no score Shape CAPSULE Size 18mm Flavor Imprint Code LANNETT;1395 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0527-1395-01 100 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 09/26/2011 2 NDC:0527-1395-10 1000 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 09/26/2011 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA090695 09/26/2011 LOXAPINE
loxapine capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0527-1396 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LOXAPINE SUCCINATE (UNII: X59SG0MRYU) (LOXAPINE - UNII:LER583670J) LOXAPINE 25 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) GELATIN (UNII: 2G86QN327L) MAGNESIUM STEARATE (UNII: 70097M6I30) POLACRILIN POTASSIUM (UNII: 0BZ5A00FQU) SODIUM LAURYL SULFATE (UNII: 368GB5141J) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) SHELLAC (UNII: 46N107B71O) ALCOHOL (UNII: 3K9958V90M) ISOPROPYL ALCOHOL (UNII: ND2M416302) AMMONIA (UNII: 5138Q19F1X) BUTYL ALCOHOL (UNII: 8PJ61P6TS3) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) FERROSOFERRIC OXIDE (UNII: XM0M87F357) FD&C BLUE NO. 2 (UNII: L06K8R7DQK) ALUMINUM OXIDE (UNII: LMI26O6933) FD&C RED NO. 40 (UNII: WZB9127XOA) METHYL ALCOHOL (UNII: Y4S76JWI15) Product Characteristics Color green (Light Green Opaque/Dark Green Opaque) Score no score Shape CAPSULE Size 18mm Flavor Imprint Code LANNETT;1396 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0527-1396-01 100 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 09/26/2011 2 NDC:0527-1396-10 1000 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 09/26/2011 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA090695 09/26/2011 LOXAPINE
loxapine capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0527-1397 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LOXAPINE SUCCINATE (UNII: X59SG0MRYU) (LOXAPINE - UNII:LER583670J) LOXAPINE 50 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) GELATIN (UNII: 2G86QN327L) MAGNESIUM STEARATE (UNII: 70097M6I30) POLACRILIN POTASSIUM (UNII: 0BZ5A00FQU) SODIUM LAURYL SULFATE (UNII: 368GB5141J) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) SHELLAC (UNII: 46N107B71O) ALCOHOL (UNII: 3K9958V90M) ISOPROPYL ALCOHOL (UNII: ND2M416302) AMMONIA (UNII: 5138Q19F1X) BUTYL ALCOHOL (UNII: 8PJ61P6TS3) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) Product Characteristics Color green (Dark Green Opaque) , blue (Light Blue Opaque) Score no score Shape CAPSULE Size 19mm Flavor Imprint Code LANNETT;1397 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0527-1397-01 100 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 09/26/2011 2 NDC:0527-1397-10 1000 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 09/26/2011 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA090695 09/26/2011 Labeler - Lannett Company, Inc. (002277481)