Label: TRIPLE ANTIBIOTIC PLUS PAIN RELIEF- bacitracin zinc, neomycin sulfate, polymyxin b sulfate, and pramoxine hydrochloride ointment
- NDC Code(s): 50066-202-01
- Packager: GENOMMA LAB USA, INC.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated June 13, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- Uses
- Warnings
- Directions
- Other information
- Inactive ingredient
- Questions?
- SPL UNCLASSIFIED SECTION
-
PRINCIPAL DISPLAY PANEL - 28.4 g Tube Carton
cicatricure ®
OPEN WOUND
FOR MINOR CUTS, SCRAPES AND BURNS
PAIN, ITCH & SCAR
- Triple Antibiotic Ointment
- Up to 24-Hour Infection Protection
- Maximum Strength: Infection Protection,
Pain & Itch Relief - Helps Healing Wounds, Minimizing
the Appearance of Scars**
FIRST AID ANTIBIOTIC
PAIN RELIEVING OINTMENTBacitracin Zinc – Neomycin Sulfate – Polyximin B Sulfate – Pramoxine HCl
*Based on published studies. For best results use sterile bandage or gauze.
**Preventing infection helps minimize appearance of scars.NET WT.
1 oz (28.4 g)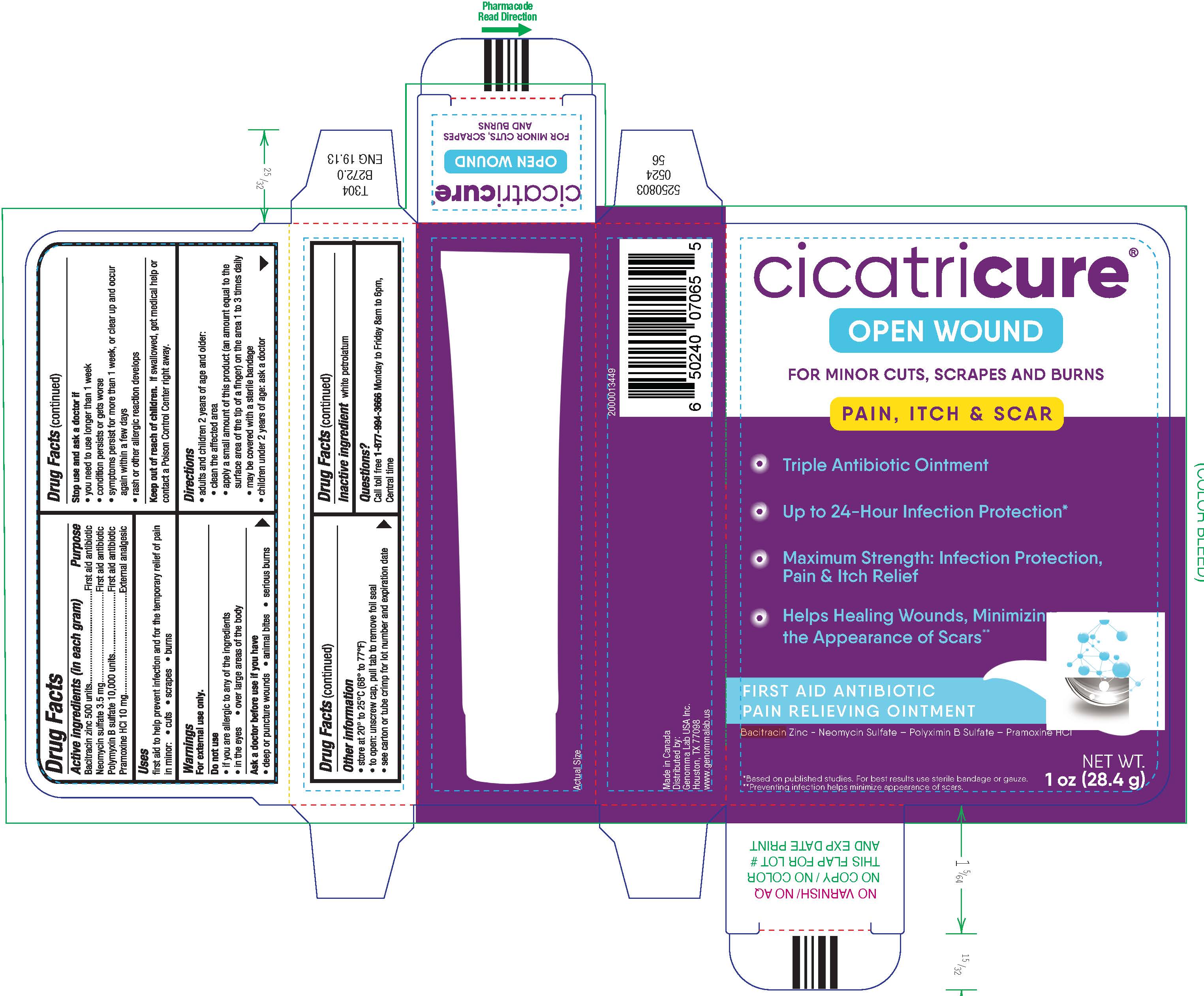
-
INGREDIENTS AND APPEARANCE
TRIPLE ANTIBIOTIC PLUS PAIN RELIEF
bacitracin zinc, neomycin sulfate, polymyxin b sulfate, and pramoxine hydrochloride ointmentProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:50066-202 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BACITRACIN ZINC (UNII: 89Y4M234ES) (BACITRACIN - UNII:58H6RWO52I) BACITRACIN ZINC 500 [USP'U] in 1 g NEOMYCIN SULFATE (UNII: 057Y626693) (NEOMYCIN - UNII:I16QD7X297) NEOMYCIN SULFATE 3.5 mg in 1 g POLYMYXIN B SULFATE (UNII: 19371312D4) (POLYMYXIN B - UNII:J2VZ07J96K) POLYMYXIN B 10000 [USP'U] in 1 g PRAMOXINE HYDROCHLORIDE (UNII: 88AYB867L5) (PRAMOXINE - UNII:068X84E056) PRAMOXINE HYDROCHLORIDE 10 mg in 1 g Inactive Ingredients Ingredient Name Strength PETROLATUM (UNII: 4T6H12BN9U) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:50066-202-01 1 in 1 CARTON 12/21/2023 1 28.4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M004 12/21/2023 Labeler - GENOMMA LAB USA, INC. (832323534) Establishment Name Address ID/FEI Business Operations Taro Pharmaceuticals Inc. 206263295 manufacture(50066-202)

