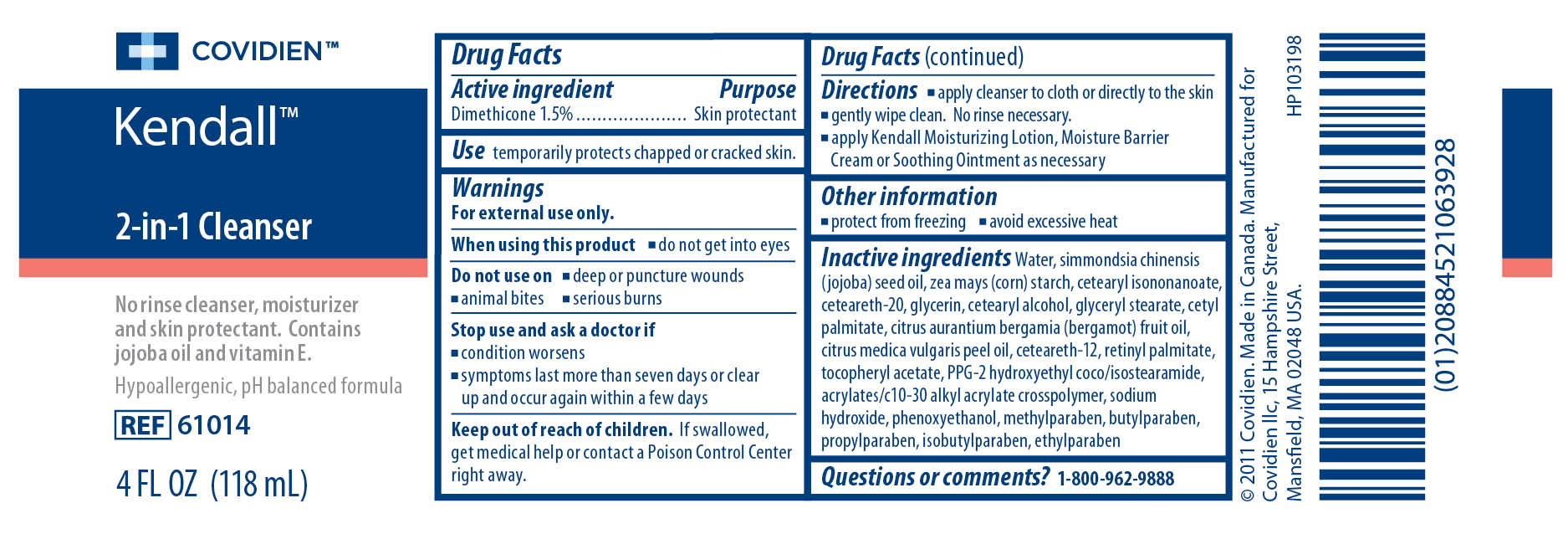
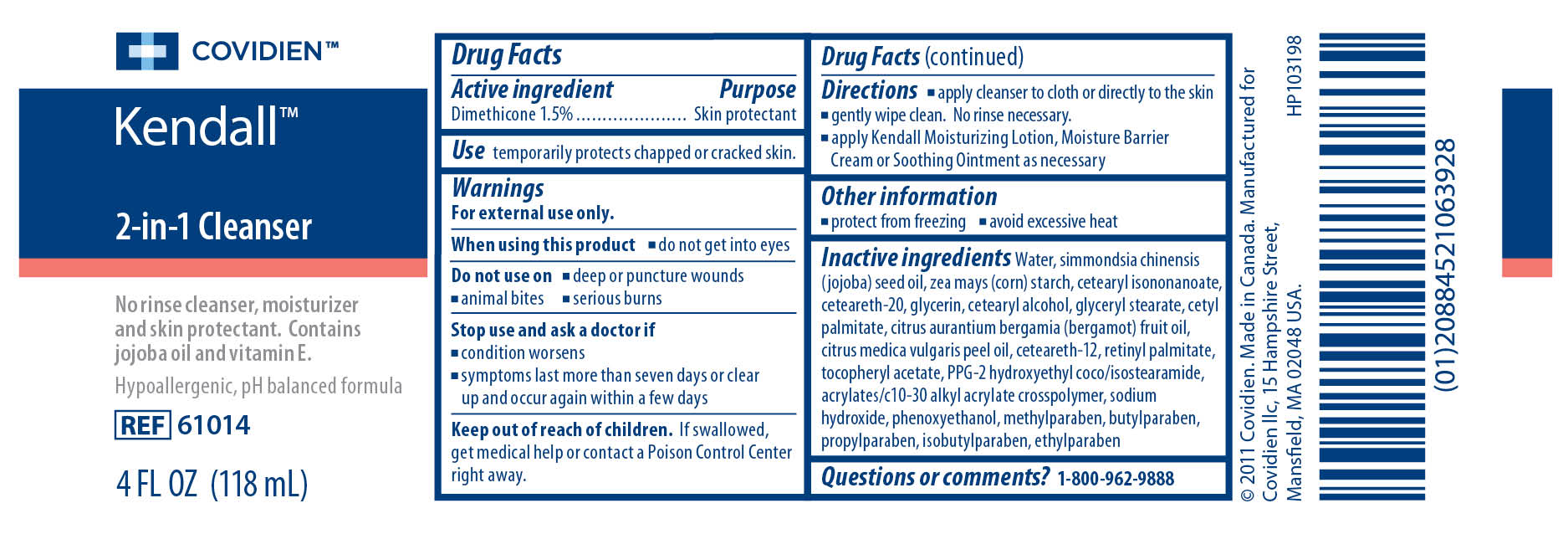
Label: KENDALL 2-IN-1 CLEANSER- dimethicone liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 28851-690-01, 28851-690-02 - Packager: Covidien Inc.
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated April 5, 2011
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
-
WARNINGS
Warnings
For external use only.
When using this product
- do not get into eyes
- deep or puncture wounds
- animal bites
- serious burns
- condition worsens
- symptoms last more than seven days or clear up and occur again within a few days
- do not get into eyes
- KEEP OUT OF REACH OF CHILDREN
- DOSAGE & ADMINISTRATION
- SPL UNCLASSIFIED SECTION
-
INACTIVE INGREDIENT
Inactive ingredients
Water, simmondsia chinensis (jojoba) seed oil, zea mays (corn) starch, cetearyl isononanoate, ceteareth-20, glycerin, cetearyl alcohol, glyceryl stearate, cetyl palmitate, cirtus aurantium bergamia (bergamot) fruit oil, citrus medica vulgaris peel oil, ceteareth-12, retinyl palmitate, tocopheryl acetate, PPG-2 hydroxyethyl coco/isostearamide, acrylates/c10-30 alkyl acrylate crosspolymer, sodium hydroxide, phenoxyethanol, methylparaben, butylparaben, propylparaben, isobutylparaben, ethylparaben
- QUESTIONS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
KENDALL 2-IN-1 CLEANSER
dimethicone liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:28851-690 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Dimethicone (UNII: 92RU3N3Y1O) (Dimethicone - UNII:92RU3N3Y1O) Dimethicone 1.5 mL in 100 mL Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) JOJOBA OIL (UNII: 724GKU717M) STARCH, CORN (UNII: O8232NY3SJ) CETEARYL ISONONANOATE (UNII: P5O01U99NI) POLYOXYL 20 CETOSTEARYL ETHER (UNII: YRC528SWUY) GLYCERIN (UNII: PDC6A3C0OX) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) CETYL PALMITATE (UNII: 5ZA2S6B08X) BERGAMOT OIL (UNII: 39W1PKE3JI) CETEARETH-12 (UNII: 7V4MR24V5P) VITAMIN A PALMITATE (UNII: 1D1K0N0VVC) ACETATE ION (UNII: 569DQM74SC) SODIUM HYDROXIDE (UNII: 55X04QC32I) PHENOXYETHANOL (UNII: HIE492ZZ3T) METHYLPARABEN (UNII: A2I8C7HI9T) BUTYLPARABEN (UNII: 3QPI1U3FV8) PROPYLPARABEN (UNII: Z8IX2SC1OH) ISOBUTYLPARABEN (UNII: 0QQJ25X58G) ETHYLPARABEN (UNII: 14255EXE39) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:28851-690-01 118 mL in 1 BOTTLE, SPRAY 2 NDC:28851-690-02 236 mL in 1 BOTTLE, SPRAY Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part347 04/05/2011 Labeler - Covidien Inc. (805770828)