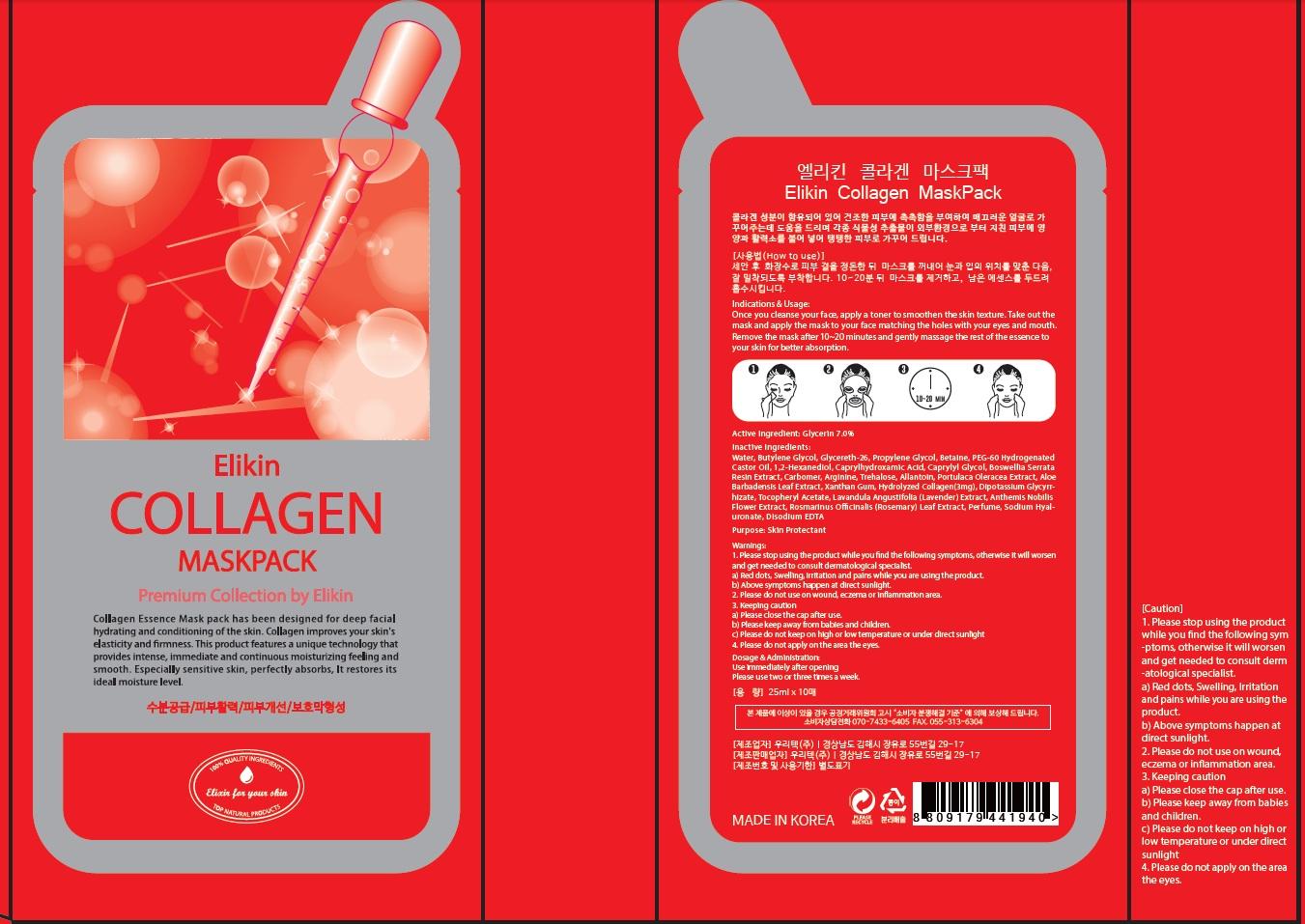
Label: ELIKIN COLLAGEN MASKPACK- glycerin patch
-
Contains inactivated NDC Code(s)
NDC Code(s): 70221-030-01 - Packager: WOORITECH CO., LTD
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated November 16, 2015
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
-
INACTIVE INGREDIENT
Inactive Ingredients: Water, Butylene Glycol, Glycereth-26, Propylene Glycol, Niacinamide, Betaine, PEG-60 Hydrogenated Castor Oil, 1,2-Hexanediol, Caprylhydroxamic Acid, Caprylyl Glycol, Boswellia Serrata Resin Extract, Carbomer, Arginine, Trehalose, Allantoin, Morus Alba Bark Extract, Glycyrrhiza Uralensis (Licorice) Root Extract, Portulaca Oleracea Extract, Aloe Barbadensis Leaf Extract, Xanthan Gum, Dipotassium Glycyrrhizate, Tocopheryl Acetate, Lavandula Angustifolia (Lavender) Extract, Anthemis Nobilis Flower Extract, Rosmarinus Officinalis (Rosemary) Leaf Extract, Perfume, Sodium Hyaluronate, Disodium EDTA
- PURPOSE
-
WARNINGS
Warnings: 1. Please stop using the product while you find the following symptoms, otherwise it will worsen and get needed to consult dermatological specialist. a) Red dots, Swelling, Irritation and pains while you are using the product. b) Above symptoms happen at direct sunlight. 2. Please do not use on wound, eczema or inflammation area. 3. Keeping caution a) Please close the cap after use. b) Please keep away from babies and children. c) Please do not keep on high or low temperature or under direct sunlight 4. Please do not apply on the area the eyes.
- KEEP OUT OF REACH OF CHILDREN
-
INDICATIONS & USAGE
Indications & Usage: Once you cleanse your face, apply a toner to smoothen the skin texture. Take out the mask and apply the mask to your face matching the holes with your eyes and mouth. Remove the mask after 10~20 minutes and gently massage the rest of the essence to your skin for better absorption.
- DOSAGE & ADMINISTRATION
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ELIKIN COLLAGEN MASKPACK
glycerin patchProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:70221-030 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Glycerin (UNII: PDC6A3C0OX) (GLYCERIN - UNII:PDC6A3C0OX) Glycerin 1.75 mg in 10 Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) Butylene Glycol (UNII: 3XUS85K0RA) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70221-030-01 10 in 1 CARTON; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 10/01/2015 Labeler - WOORITECH CO., LTD (688437735) Registrant - WOORITECH CO., LTD (688437735) Establishment Name Address ID/FEI Business Operations WOORITECH CO., LTD 688437735 manufacture(70221-030)