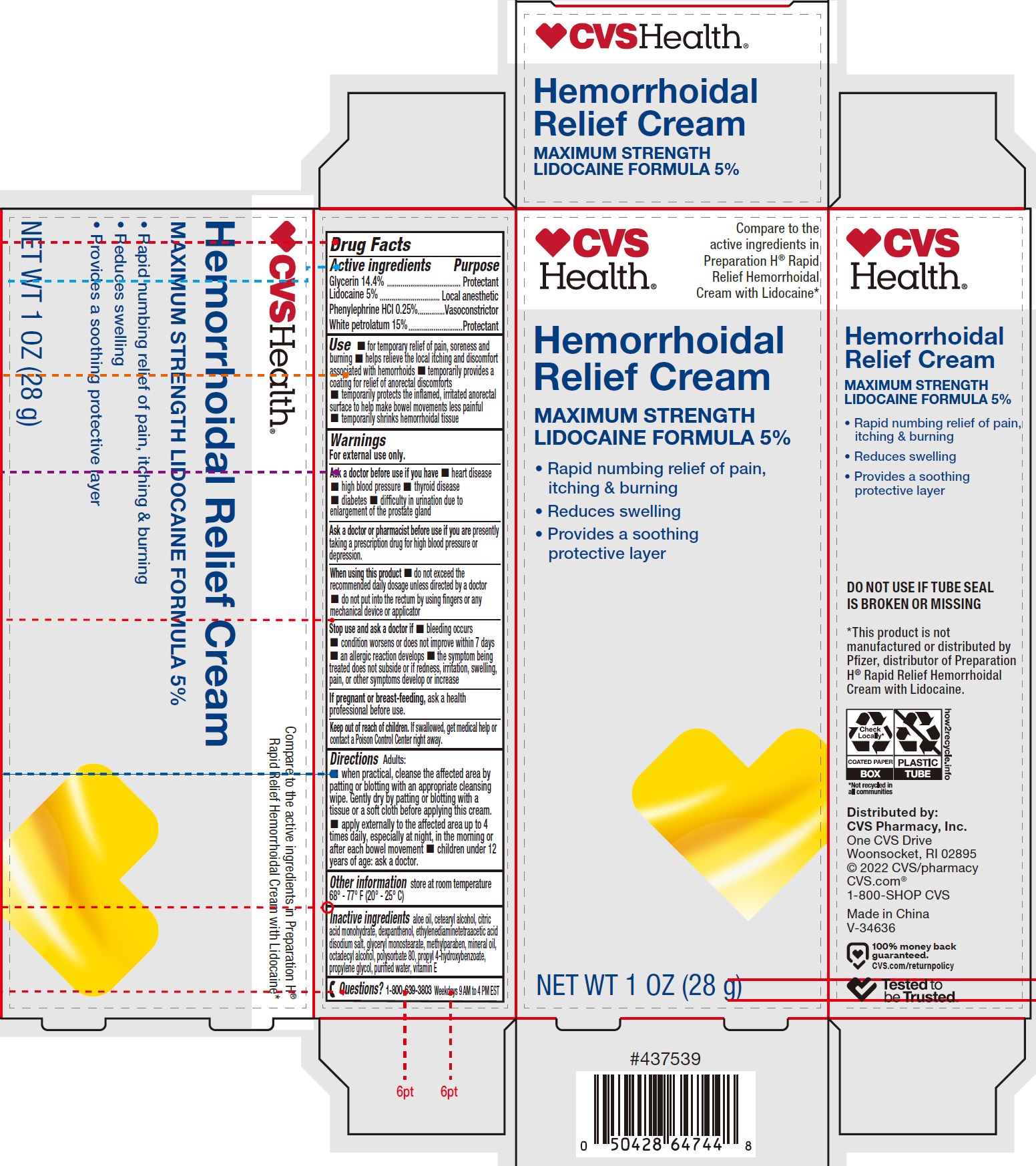
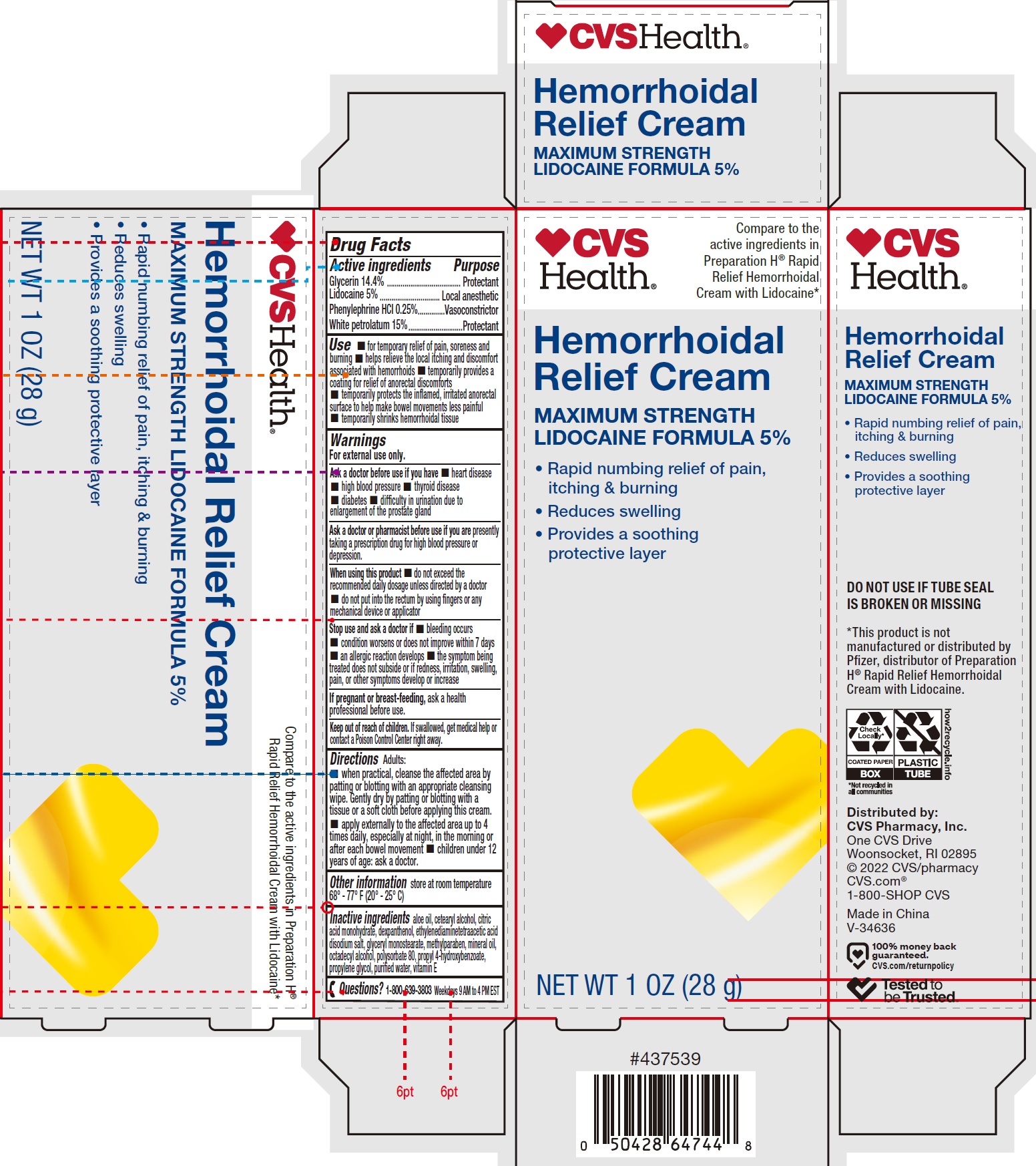
Label: HEMORRHOIDAL WITH LIDOCAINE- glycerin, lidocaine, phenylephrine hydrochloride, white petrolatum cream
- NDC Code(s): 51316-225-00
- Packager: CVS PHARMACY, INC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated October 24, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active ingredients
-
Use
- for temporary relief of pain, soreness and burning
- helps relieve the local itching and discomfort associated with hemorrhoids
- temporarily provides a coating for relief of anorectal discomforts
- temporarily protects the inflamed, irritated anorectal surface to help make bowel movements less painful
- temporarily shrinks hemorrhoidal tissue
-
Warnings
For external use only.
Ask a doctor before use if you have
- heart disease
- high blood pressure
- thyroid disease
- diabetes
- diffficulty in urination due to enlargement of the prostate gland
Ask a doctor or pharmacist before use if you are
presently taking a prescription drug for high blood pressure or depression.
When using this product
- do not exceed the recommended daily dosage unless directed by a doctor
- do not put into the rectum by using fingers or any mechanical device or applicator
-
Directions
Adults:
- when practical, cleanse the affected area by patting or blotting with an appropriate cleansing wipe. Gently dry by patting or blotting with a tissue or a soft cloth before applying this cream.
- apply externally to the affected area up to 4 times daily, especially at night, in the morning or after each bowel movement
- children under 12 years of age: ask a doctor.
- Other information
- Inactive ingredients
- Questions?
- Package Labeling:
-
INGREDIENTS AND APPEARANCE
HEMORRHOIDAL WITH LIDOCAINE
glycerin, lidocaine, phenylephrine hydrochloride, white petrolatum creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:51316-225 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength GLYCERIN (UNII: PDC6A3C0OX) (GLYCERIN - UNII:PDC6A3C0OX) GLYCERIN 144 mg in 1 g LIDOCAINE (UNII: 98PI200987) (LIDOCAINE - UNII:98PI200987) LIDOCAINE 50 mg in 1 g PHENYLEPHRINE HYDROCHLORIDE (UNII: 04JA59TNSJ) (PHENYLEPHRINE - UNII:1WS297W6MV) PHENYLEPHRINE 2.5 mg in 1 g WHITE PETROLATUM (UNII: B6E5W8RQJ4) (PHENYLEPHRINE - UNII:1WS297W6MV) PHENYLEPHRINE 150 mg in 1 g Inactive Ingredients Ingredient Name Strength CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) DEXPANTHENOL (UNII: 1O6C93RI7Z) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) METHYLPARABEN (UNII: A2I8C7HI9T) MINERAL OIL (UNII: T5L8T28FGP) STEARYL ALCOHOL (UNII: 2KR89I4H1Y) POLYSORBATE 80 (UNII: 6OZP39ZG8H) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) .ALPHA.-TOCOPHEROL (UNII: H4N855PNZ1) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:51316-225-00 1 in 1 BOX 06/01/2022 1 28 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M016 06/01/2022 Labeler - CVS PHARMACY, INC (062312574)