Label: LAUGHING LINEN HAND SANITIZER- alcohol liquid
RAINBOW BERRY HAND SANITIZER- alcohol liquid
RED HOT CINNAMON HAND SANITIZER- alcohol liquid
THUMBS-UP LIME HAND SANITIZER- alcohol liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 29500-9815-1, 29500-9816-1, 29500-9817-1, 29500-9818-1 - Packager: GL360
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated June 10, 2018
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
- WARNINGS
- WHEN USING
- STOP USE
- KEEP OUT OF REACH OF CHILDREN
- DOSAGE & ADMINISTRATION
- INACTIVE INGREDIENT
-
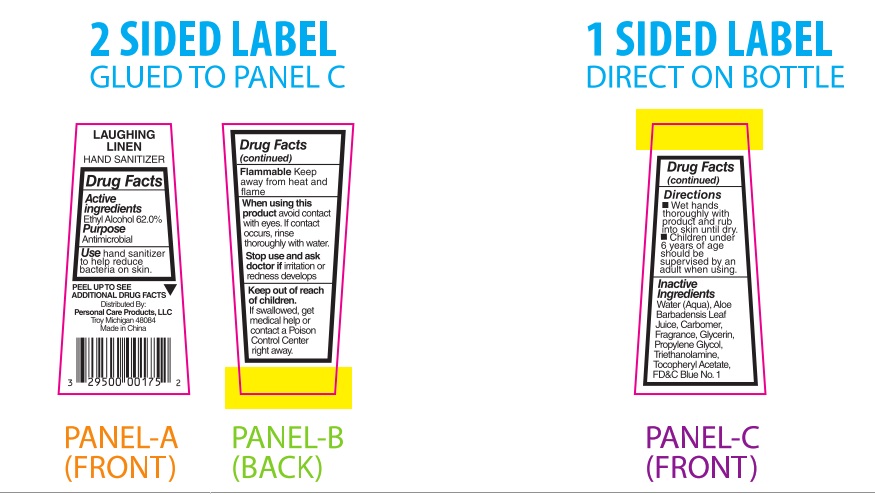
PRINCIPAL DISPLAY PANEL
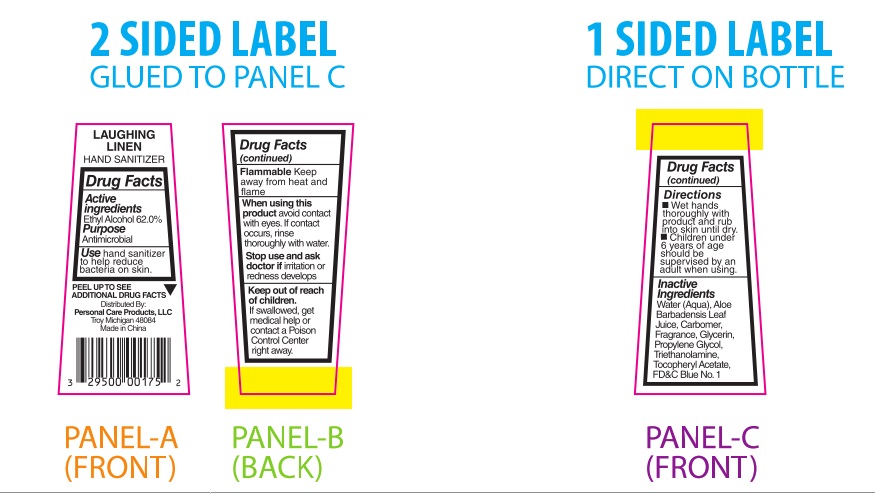
Laughing Linen
HAND SANITIZER
Drug Facts
Active
Ingredients
Ethyl Alcohol 62.0%Purpose
AntimicrobialUse hand sanitizer
to help reduce
bacteria on skin.PEEL TO SEE ▼
ADDITIONAL DRUG FACTSDistributed By:
Personal Care Products, LLC
Troy, Michigan 48084
Made in ChinaDrug Facts
(continued)Flammable Keep
away from heat and
flameWhen using this
product avoid contact
with eyes. If contact
occurs, rinse
thoroughly with water.Stop use and ask
doctor if irritation or
redness developsKeep out of reach
of children.
If swallowed, get
medical help or
contact a Poison
Control Center
right away.Drug Facts
(continued)Directions
■ Wet hands
thoroughly with
product and rub
into skin until dry.
■ Children under
6 years of age
should be
supervised by an
adult when using.Inactive
IngredientsWater (Aqua), Aloe
Barbadensis Leaf Juice,
Carbomer, Fragrance,
Glycerin, Propylene
Glycol, Triethanolamine,
Tocopheryl Acetate,
FD&C Blue No. 1___________________________________________________________________________________________________________________
-
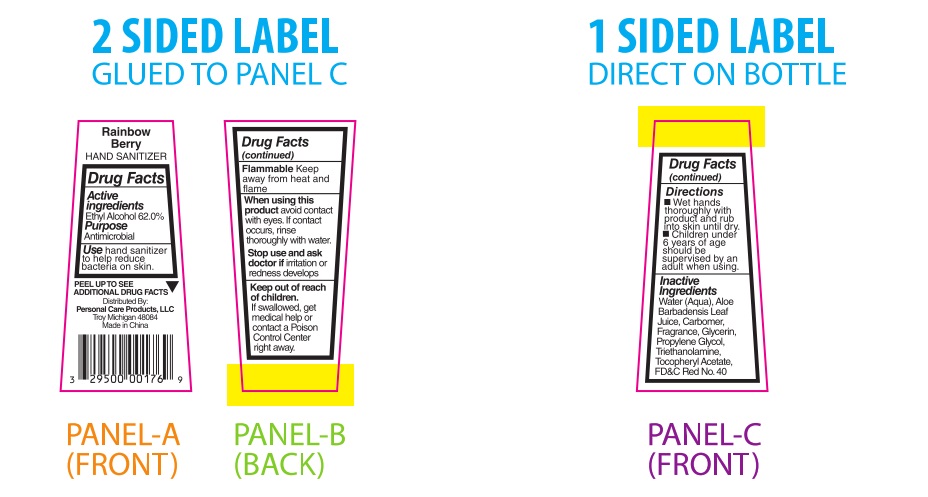
PRINCIPAL DISPLAY PANEL
Rainbow Berry
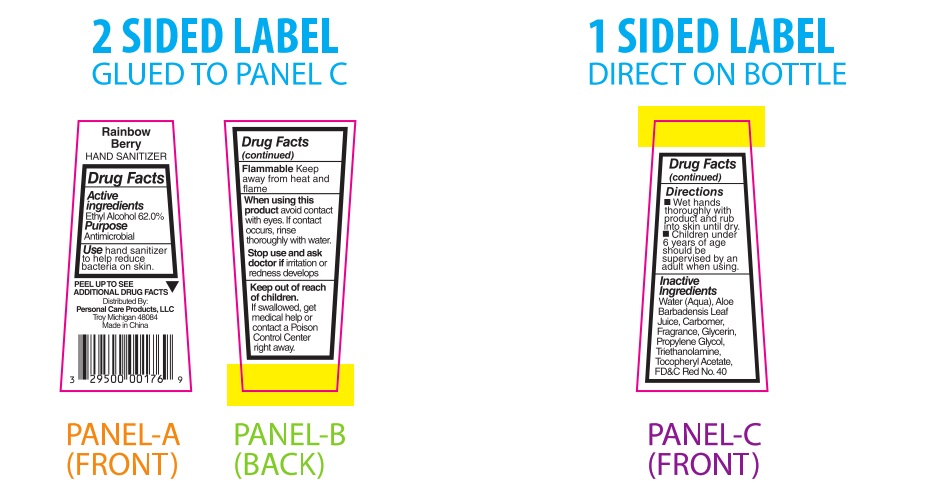
Rainbow Berry
HAND SANITIZERDrug Facts
Active
Ingredients
Ethyl Alcohol 62.0%Purpose
AntimicrobialUsehand sanitizer
to help reduce
bacteria on skin.PEEL TO SEE ▼
ADDITIONAL DRUG FACTSDistributed By:
Personal Care Products, LLC
Troy, Michigan 48084
Made in ChinaDrug Facts
(continued)Flammable Keep
away from heat and
flameWhen using this
product avoid contact
with eyes. If contact
occurs, rinse
thoroughly with water.Stop use and ask
doctor if irritation or
redness developsKeep out of reach
of children.
If swallowed, get
medical help or
contact a Poison
Control Center
right away.Drug Facts
(continued)Directions
■ Wet hands
thoroughly with
product and rub
into skin until dry.
■ Children under
6 years of age
should be
supervised by an
adult when using.Inactive
IngredientsWater (Aqua), Aloe
Barbadensis Leaf Juice,
Carbomer, Fragrance,
Glycerin, Propylene
Glycol, Triethanolamine,
Tocopheryl Acetate, FD&C Red No. 40 -
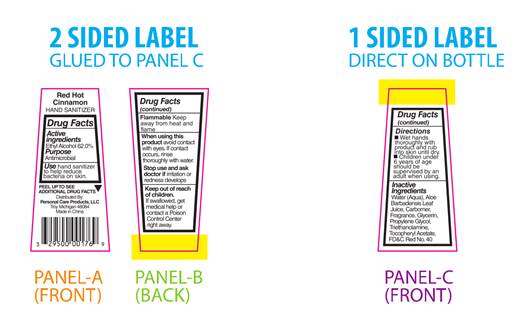
PRINCIPAL DISPLAY PANEL
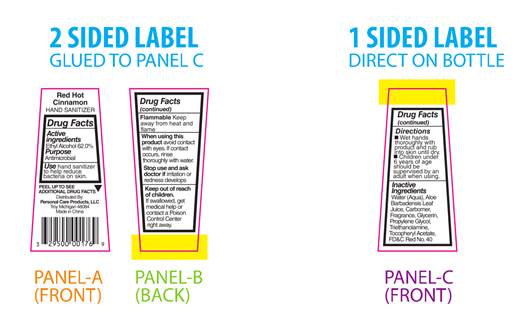
Red Hot Cinnamon
HAND SANITIZERDrug Facts
Active
Ingredients
Ethyl Alcohol 62.0%Purpose
AntimicrobialUse hand sanitizer
to help reduce
bacteria on skin.PEEL TO SEE ▼
ADDITIONAL DRUG FACTSDistributed By:
Personal Care Products, LLC
Troy, Michigan 48084
Made in ChinaDrug Facts
(continued)Flammable Keep
away from heat and
flameWhen using this
product avoid contact
with eyes. If contact
occurs, rinse
thoroughly with water.Stop use and ask
doctor if irritation or
redness developsKeep out of reach
of children.
If swallowed, get
medical help or
contact a Poison
Control Center
right away.Drug Facts
(continued)Directions
■ Wet hands
thoroughly with
product and rub
into skin until dry.
■ Children under
6 years of age
should be
supervised by an
adult when using.Inactive
Ingredients
Water (Aqua), Aloe
Barbadensis Leaf Juice,
Carbomer, Fragrance,
Glycerin, Propylene
Glycol, Triethanolamine,
Tocopheryl Acetate,
FD&C Red No. 40
-
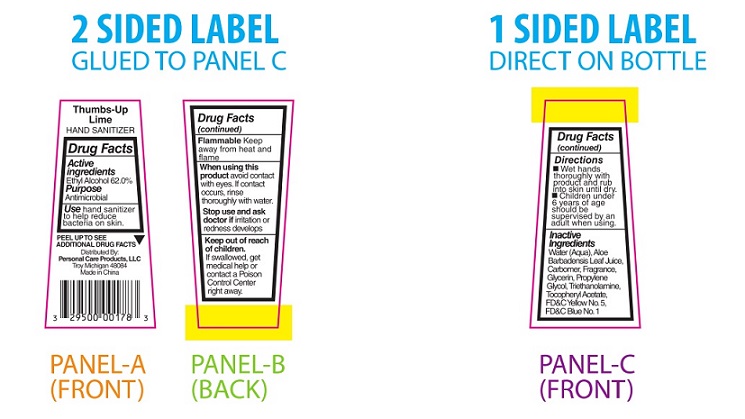
PRINCIPAL DISPLAY PANEL
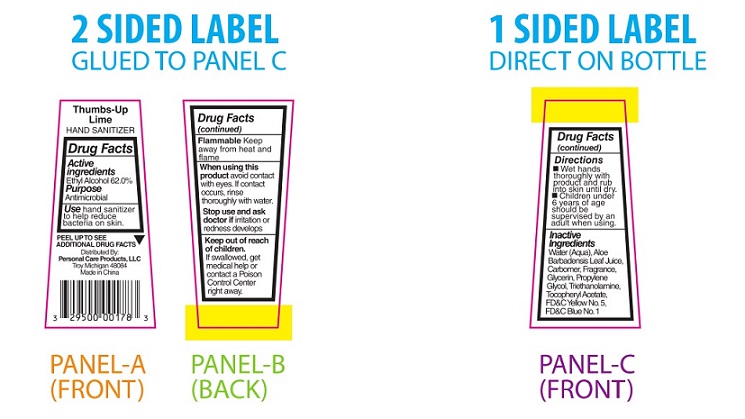
Thumbs-Up Lime
HAND SANITIZERDrug Facts
Active
Ingredients
Ethyl Alcohol 62.0%Purpose
AntimicrobialUse hand sanitizer
to help reduce
bacteria on skin.PEEL TO SEE ▼
ADDITIONAL DRUG FACTSDistributed By:
Personal Care Products, LLC
Troy, Michigan 48084
Made in ChinaDrug Facts
(continued)Flammable Keep
away from heat and
flameWhen using this
product avoid contact
with eyes. If contact
occurs, rinse
thoroughly with water.Stop use and ask
doctor if irritation or
redness developsKeep out of reach
of children.
If swallowed, get
medical help or
contact a Poison
Control Center
right away.Drug Facts
(continued)Directions
■ Wet hands
thoroughly with
product and rub
into skin until dry.
■ Children under
6 years of age
should be
supervised by an
adult when using.Inactive
Ingredients
Water (Aqua), Aloe
Barbadensis Leaf Juice,
Carbomer, Fragrance,
Glycerin, Propylene
Glycol, Triethanolamine,
Tocopheryl Acetate,
FD&C Yellow No. 5,
FD&C Blue No.1 -
INGREDIENTS AND APPEARANCE
LAUGHING LINEN HAND SANITIZER
alcohol liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:29500-9815 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 17.57 g in 30 mL Inactive Ingredients Ingredient Name Strength water (UNII: 059QF0KO0R) ALOE VERA LEAF (UNII: ZY81Z83H0X) CARBOXYPOLYMETHYLENE (UNII: 0A5MM307FC) GLYCERIN (UNII: PDC6A3C0OX) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) TROLAMINE (UNII: 9O3K93S3TK) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:29500-9815-1 30 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 07/01/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333E 07/01/2018 RAINBOW BERRY HAND SANITIZER
alcohol liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:29500-9816 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 17.57 g in 30 mL Inactive Ingredients Ingredient Name Strength water (UNII: 059QF0KO0R) ALOE VERA LEAF (UNII: ZY81Z83H0X) CARBOXYPOLYMETHYLENE (UNII: 0A5MM307FC) GLYCERIN (UNII: PDC6A3C0OX) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) TROLAMINE (UNII: 9O3K93S3TK) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) FD&C RED NO. 40 (UNII: WZB9127XOA) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:29500-9816-1 30 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 07/01/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333E 07/01/2018 RED HOT CINNAMON HAND SANITIZER
alcohol liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:29500-9817 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 17.57 g in 30 mL Inactive Ingredients Ingredient Name Strength water (UNII: 059QF0KO0R) ALOE VERA LEAF (UNII: ZY81Z83H0X) CARBOXYPOLYMETHYLENE (UNII: 0A5MM307FC) GLYCERIN (UNII: PDC6A3C0OX) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) TROLAMINE (UNII: 9O3K93S3TK) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) FD&C RED NO. 40 (UNII: WZB9127XOA) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:29500-9817-1 30 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 07/01/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333E 07/01/2018 THUMBS-UP LIME HAND SANITIZER
alcohol liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:29500-9818 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 17.57 g in 30 mL Inactive Ingredients Ingredient Name Strength water (UNII: 059QF0KO0R) ALOE VERA LEAF (UNII: ZY81Z83H0X) CARBOXYPOLYMETHYLENE (UNII: 0A5MM307FC) GLYCERIN (UNII: PDC6A3C0OX) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) TROLAMINE (UNII: 9O3K93S3TK) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:29500-9818-1 30 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 07/01/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333E 07/01/2018 Labeler - GL360 (966155082) Registrant - GL360 (966155082) Establishment Name Address ID/FEI Business Operations Ningbo Liyuan Daily Chemical Products Co., Ltd 530766098 manufacture(29500-9815, 29500-9816, 29500-9817, 29500-9818)