Label: METHIMAZOLE tablet
-
Contains inactivated NDC Code(s)
NDC Code(s): 54868-5135-0, 54868-6071-0, 54868-6071-1 - Packager: Physicians Total Care, Inc.
- This is a repackaged label.
- Source NDC Code(s): 49884-640, 49884-641
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated February 6, 2013
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
DESCRIPTION
Methimazole (1-methylimidazole-2-thiol) is a white, crystalline substance that is freely soluble in water. It differs chemically from the drugs of the thiouracil series primarily because it has a 5- instead of a 6-membered ring.
Each tablet contains 5 or 10 mg (43.8 or 87.6 μmol) methimazole, an orally administered antithyroid drug. Each tablet also contains lactose monohydrate, magnesium stearate, corn starch, and talc.
The molecular weight is 114.17, and the molecular formula is C4H6N2S. The structural formula is as follows:
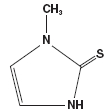
-
CLINICAL PHARMACOLOGY
Methimazole inhibits the synthesis of thyroid hormones and thus is effective in the treatment of hyperthyroidism. The drug does not inactivate existing thyroxine and tri-iodothyronine that are stored in the thyroid or circulating in the blood nor does it interfere with the effectiveness of thyroid hormones given by mouth or by injection.
Methimazole is readily absorbed in the gastrointestinal tract, metabolized in the liver, and excreted in the urine.
- INDICATIONS AND USAGE
- CONTRAINDICATIONS
-
WARNINGS
Congenital Malformations
Methimazole readily crosses placental membranes and can cause fetal harm, particularly when administered in the first trimester of pregnancy. Rare instances of congenital defects, including aplasia cutis, craniofacial malformations (facial dysmorphism; choanal atresia) and gastrointestinal malformations (esophageal atresia with or without tracheoesophageal fistula; umbilical abnormalities) have occurred in infants born to mothers who received methimazole during pregnancy. If methimazole is used during pregnancy or if the patient becomes pregnant while taking this drug, the patient should be warned of the potential hazard to the fetus.
Since the above congenital defects have been reported in offspring of patients treated with methimazole, it may be appropriate to use other agents in pregnant women requiring treatment for hyperthyroidism, particularly during organogenesis, in the first trimester of pregnancy. If methimazole is used, the lowest possible dose to control the maternal disease should be given.
Agranulocytosis
Agranulocytosis is a potentially life-threatening adverse reaction of methimazole therapy. Patients should be instructed to immediately report to their physicians any symptoms suggestive of agranulocytosis, such as fever or sore throat. Leukopenia, thrombocytopenia, and aplastic anemia (pancytopenia) may also occur. The drug should be discontinued in the presence of agranulocytosis, aplastic anemia (pancytopenia), ANCA-positive vasculitis, hepatitis, or exfoliative dermatitis, and the patient’s bone marrow indices should be monitored.
Liver Toxicity
Although there have been reports of hepatotoxicity (including acute liver failure) associated with methimazole, the risk of hepatotoxicity appears to be less with methimazole than with propylthiouracil, especially in the pediatric population. Symptoms suggestive of hepatic dysfunction (anorexia, pruritus, right upper quadrant pain, etc) should prompt evaluation of liver function (bilirubin, alkaline phosphatase) and hepatocellular integrity (ALT, AST). Drug treatment should be discontinued promptly in the event of clinically significant evidence of liver abnormality including hepatic transaminase values exceeding 3 times the upper limit of normal.
Hypothyroidism
Methimazole can cause hypothyroidism necessitating routine monitoring of TSH and free T4 levels with adjustments in dosing to maintain a euthyroid state. Because the drug readily crosses placental membranes, methimazole can cause fetal goiter and cretinism when administered to a pregnant woman. For this reason, it is important that a sufficient, but not excessive, dose be given during pregnancy (see Precautions, Pregnancy).
-
PRECAUTIONS
General
Patients who receive methimazole should be under close surveillance and should be cautioned to report immediately any evidence of illness, particularly sore throat, skin eruptions, fever, headache, or general malaise. In such cases, white-blood-cell and differential counts should be obtained to determine whether agranulocytosis has developed. Particular care should be exercised with patients who are receiving additional drugs known to cause agranulocytosis.
Laboratory Tests
Because methimazole may cause hypoprothrombinemia and bleeding, prothrombin time should be monitored during therapy with the drug, especially before surgical procedures.
Thyroid function tests should be monitored periodically during therapy. Once clinical evidence of hyperthyroidism has resolved, the finding of a rising serum TSH indicates that a lower maintenance dose of methimazole should be employed.
Drug Interactions
Anticoagulants (oral)
Due to potential inhibition of vitamin K activity by methimazole, the activity of oral anticoagulants (e.g., warfarin) may be increased; additional monitoring of PT/INR should be considered, especially before surgical procedures.
ß-adrenergic blocking agents
Hyperthyroidism may cause an increased clearance of beta blockers with a high extraction ratio. A reduced dose of beta-adrenergic blockers may be needed when a hyperthyroid patient becomes euthyroid.
Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 2 year study, rats were given methimazole at doses of 0.5, 3, and 18 mg/kg/day. These doses were 0.3, 2, and 12 times the 15 mg/day maximum human maintenance dose (when calculated on the basis of surface area). Thyroid hyperplasia, adenoma, and carcinoma developed in rats at the two higher doses. The clinical significance of these findings is unclear.
Pregnancy Category D
Due to the rare occurrence of congenital malformations associated with methimazole use, it may be appropriate to use other agents in pregnant women requiring treatment for hyperthyroidism, particularly in the first trimester of pregnancy (during organogenesis).
Patients should be advised that if they become pregnant or intend to become pregnant while taking an antithyroid drug, they should contact their physician immediately about their therapy.
In many pregnant women, the thyroid dysfunction diminishes as the pregnancy proceeds; consequently, a reduction in dosage of anti-thyroid therapy may be possible. In some instances, use of anti-thyroid therapy can be discontinued 2 or 3 weeks before delivery. Because the drug readily crosses placental membranes, methimazole can cause fetal goiter and cretinism when administered to a pregnant woman. For this reason, it is important that a sufficient, but not excessive, dose be given during pregnancy. (See WARNINGS)
Nursing Mothers
Methimazole is excreted into breast milk. However, several studies found no effect on clinical status in nursing infants of mothers taking methimazole, particularly if thyroid function is monitored at frequent (weekly or biweekly) intervals. A long-term study of 139 thyrotoxic lactating mothers and their infants failed to demonstrate toxicity in infants who are nursed by mothers receiving treatment with methimazole.
Pediatric Use
Because of postmarketing reports of severe liver injury in pediatric patients treated with propylthiouracil, methimazole is the preferred choice when an antithyroid drug is required for a pediatric patient. (See DOSAGE AND ADMINISTRATION.)
-
ADVERSE REACTIONS
Major adverse reactions (which occur with much less frequency than the minor adverse reactions) include inhibition of myelopoieses (agranulocytosis, granulocytopenia, thrombocytopenia, and aplastic anemia), drug fever, a lupuslike syndrome, insulin autoimmune syndrome (which can result in hypoglycemic coma), hepatitis (jaundice may persist for several weeks after discontinuation of the drug), periarteritis, and hypoprothrombinemia. Nephritis occurs very rarely.
Minor adverse reactions include skin rash, urticaria, nausea, vomiting, epigastric distress, arthralgia, paresthesia, loss of taste, abnormal loss of hair, myalgia, headache, pruritus, drowsiness, neuritis, edema, vertigo, skin pigmentation, jaundice, sialadenopathy, and lymphadenopathy.
-
OVERDOSAGE
Signs and Symptoms
Symptoms may include nausea, vomiting, epigastric distress, headache, fever, joint pain, pruritus, and edema. Aplastic anemia (pancytopenia) or agranulocytosis may be manifested in hours to days. Less frequent events are hepatitis, nephrotic syndrome, exfoliative dermatitis, neuropathies, and CNS stimulation or depression.
No information is available on the median lethal dose of the drug or the concentration of methimazole in biologic fluids associated with toxicity and/or death.
Treatment
To obtain up-to-date information about the treatment of overdose, a good resource is the certified Regional Poison Control Center. In managing overdosage, consider the possibility of multiple drug overdoses, interaction among drugs, and unusual drug kinetics in the patient.
In the event of an overdose, appropriate supportive treatment should be initiated as dictated by the patient’s medical status.
-
DOSAGE AND ADMINISTRATION
Methimazole is administered orally. The total daily dosage is usually given in 3 divided doses at approximately 8-hour intervals.
-
HOW SUPPLIED
Methimazole Tablets, USP 5 mg - white to off-white, round, flat-faced, bevelled-edged tablets, scored with “EM 5” on one side and plain on the other.
They are available in:
Bottles of 30
NDC 54868-6071-0
Bottles of 90
NDC 54868-6071-1
Store at controlled room temperature 15° to 30°C (59° to 86°F).
Dispense in tight, light-resistant container.
Manufactured and Distributed by:
PAR PHARMACEUTICAL COMPANIES, INC.
Spring Valley, NY 10977Revised: 01/12 OS640-01-1-09
Distributed by:
Physicians Total Care, Inc.
Tulsa, Oklahoma 74146
-
PRINCIPAL DISPLAY PANEL
PRINCIPAL DISPLAY PANEL - 5 mg
NDC 54868-6071-0

Methimazole
Tablets, USP
5 mgRx only
Each tablet contains:
Methimazole ................................ 5 mgUSUAL DOSAGE:
See package insert for dosage information.WARNING: This drug may cause toxic
reactions. If such reactions occur,
discontinue the drug. Constant
supervision of patient is essential.KEEP THIS AND ALL DRUGS
OUT OF REACH OF CHILDREN.Dispense in a tight, light-resistant
container. Keep tightly closed.Store at controlled room
temperature 15° – 30°C (59° – 86°F).R01/06 LA640-01-1-04
-
INGREDIENTS AND APPEARANCE
METHIMAZOLE
methimazole tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:54868-6071(NDC:49884-640) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength METHIMAZOLE (UNII: 554Z48XN5E) (METHIMAZOLE - UNII:554Z48XN5E) METHIMAZOLE 5 mg Inactive Ingredients Ingredient Name Strength LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) STARCH, CORN (UNII: O8232NY3SJ) TALC (UNII: 7SEV7J4R1U) Product Characteristics Color WHITE (off-white) Score 2 pieces Shape ROUND Size 6mm Flavor Imprint Code EM;5 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:54868-6071-0 30 in 1 BOTTLE 2 NDC:54868-6071-1 90 in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA040350 10/13/2009 METHIMAZOLE
methimazole tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:54868-5135(NDC:49884-641) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength METHIMAZOLE (UNII: 554Z48XN5E) (METHIMAZOLE - UNII:554Z48XN5E) METHIMAZOLE 10 mg Inactive Ingredients Ingredient Name Strength LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) STARCH, CORN (UNII: O8232NY3SJ) TALC (UNII: 7SEV7J4R1U) Product Characteristics Color WHITE (off-white) Score 2 pieces Shape ROUND Size 8mm Flavor Imprint Code EM;10 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:54868-5135-0 100 in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA040350 08/13/2004 06/30/2011 Labeler - Physicians Total Care, Inc. (194123980) Establishment Name Address ID/FEI Business Operations Physicians Total Care, Inc. 194123980 relabel(54868-6071) , repack(54868-6071)

