Label: MULTIVITAMIN WITH FLUORIDE- sodium fluoride tablet, chewable
- NDC Code(s): 59088-107-59
- Packager: PureTek Corporation
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated January 31, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- DESCRIPTION:
-
CLINICAL PHARMACOLOGY
It is well established that fluoridation of the water supply (1 ppm fluoride) during the period of tooth development leads to a significant decrease in the incidence of dental caries.
Multivitamin with 0.25 mg Fluoride Chewable Tablets provide sodium fluoride and ten essential vitamins in a chewable tablet. Because the tablets are chewable, they provide a topical as well as systemic source of fluoride. Hydroxyapatite is the principal crystal for all calcified tissue in the human body. The fluoride ion reacts with the hydroxyapatite in the tooth as it is formed to produce the more caries-resistant crystal, fluorapatite. The reaction may be expressed by the equation:
Ca 10(PO 4) 6(OH) 2 + 2F- ------- Ca 10(PO 4) 6F 2 + 2OH-
(Hydroxyapatite) (Fluorapatite)
Three stages of fluoride deposition in tooth enamel can be distinguished:
1. Small amounts (reflecting the low levels of fluoride in tissue fluids) are incorporated into the enamel crystals while they are being formed.
2. After enamel has been laid down, fluoride deposition continues in the surface enamel. Diffusion of fluoride from the surface inward is apparently restricted.
3. After eruption, the surface enamel acquires fluoride from the water, food, supplementary fluoride and smaller amounts from saliva.
-
INDICATIONS AND USAGE
Supplementation of the diet with ten essential vitamins.
Supplementation of the diet with fluoride for caries prophylaxis.The American Academy of Pediatrics recommends that children up to the age 16, in areas where drinking water contains less than optimal levels of fluoride, receive daily fluoride supplementation.Multivitamin with Fluoride 0.25 mg Chewable Tablets provide 0.25 mg fluoride in tablet form for children 6-16 years of age in areas where the drinking water fluoride level is less than 0.3 ppm.
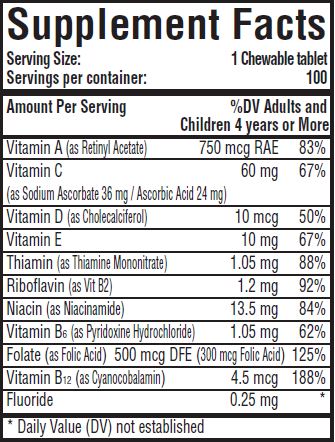
Multivitamin with Fluoride 0.25 mg Chewable Tablets supply significant amounts of Vitamins A, C, D, E, thiamine, riboflavin, niacin, vitamin B6, vitamin B12, and folate to supplement the diet, and to help assure that nutritional deficiencies of these vitamins will not develop. Thus, in a single easy-to-use preparation, children obtain ten essential vitamins and the important mineral, fluoride.Children using Multivitamin with Fluoride 0.25 mg Chewable Tablets regularly should receive semiannual dental examinations. The regular brushing of teeth and attention to good oral hygiene practices are also essential.
Multivitamin with Fluoride 0.25 mg Chewable Tablets is a prescription product for the clinical dietary management of the metabolic processes of caries prophylaxis and provides supplementation of the diet with ten essential vitamins. -
WARNING
Keep out of the reach of children. In case of accidental overdose, seek professional assistance or contact a Poison Control Center immediately.
Caution: Do not eat or drink dairy products within one hour of fluoride administration.Should be chewed. This product, as all chewable tablets, is not recommended for children under age 4 due to risk of choking.
-
PRECAUTION
The suggested dose of Multivitamin with Fluoride 0.25 mg Chewable Tablets should not be exceeded, since dental fluorosis may result from continued ingestion of large amounts of fluoride.
Before prescribing Multivitamin with Fluoride 0.25 mg Chewable Tablets:
1. Determine the fluoride content of the drinking water from all major sources.2. Make sure the child is not receiving significant amounts of fluoride from other sources such as medications and swallowed toothpaste.
3. Periodically check to make sure that the child does not develop significant dental fluorosis.
Phenylketonurics: Contains Phenylalanine 3.0 mg Per Tablet.
- ADVERSE REACTIONS
- DOSAGE AND ADMINISTRATION
-
HOW SUPPLIED
Multivitamin chewable tablets containing 0.25 mg fluoride are purple-colored, grape flavor, un-scored, round debossed
“107” tablets are available on prescription only in bottles of 100 tablets. NDC: 59088-107-59
Store at 20°- 25°C (68°- 77°F); excursions permitted to 15°– 30°C (59°– 86°F) [see USP Controlled Room Temperature].
Dispense in a tight, light resistant container with a child–resistant closure as defined in the USP/NF. All prescription substitutions using this product shall be pursuant to state statutes as applicable. This is not an Orange Book product. -
STORAGE
Store at 20°-25°C (68°-77°F); excursions permitted to 15°-30°C (59°-86°F). See USP Controlled Room Temperature.
Dispense in a tight, light resistant container with a child-resistant closure as defined in the USP/NF. All prescription substitutions using this product shall be pursuant to state statutes as applicable. This is not an Orange Book product.
- Multivitamin with Fluoride Chewable Tablets 0.25 mg
-
INGREDIENTS AND APPEARANCE
MULTIVITAMIN WITH FLUORIDE
sodium fluoride tablet, chewableProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:59088-107 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SODIUM FLUORIDE (UNII: 8ZYQ1474W7) (FLUORIDE ION - UNII:Q80VPU408O) FLUORIDE ION 0.25 mg VITAMIN A (UNII: 81G40H8B0T) (VITAMIN A - UNII:81G40H8B0T) VITAMIN A 750 ug ASCORBIC ACID (UNII: PQ6CK8PD0R) (ASCORBIC ACID - UNII:PQ6CK8PD0R) ASCORBIC ACID 24 mg SODIUM ASCORBATE (UNII: S033EH8359) (ASCORBIC ACID - UNII:PQ6CK8PD0R) ASCORBIC ACID 36 mg CHOLECALCIFEROL (UNII: 1C6V77QF41) (CHOLECALCIFEROL - UNII:1C6V77QF41) CHOLECALCIFEROL 10 ug .ALPHA.-TOCOPHEROL ACETATE, DL- (UNII: WR1WPI7EW8) (.ALPHA.-TOCOPHEROL, DL- - UNII:7QWA1RIO01) .ALPHA.-TOCOPHEROL, DL- 10 mg THIAMINE MONONITRATE (UNII: 8K0I04919X) (THIAMINE ION - UNII:4ABT0J945J) THIAMINE 1.05 mg RIBOFLAVIN (UNII: TLM2976OFR) (RIBOFLAVIN - UNII:TLM2976OFR) RIBOFLAVIN 1.2 mg NIACINAMIDE (UNII: 25X51I8RD4) (NIACINAMIDE - UNII:25X51I8RD4) NIACINAMIDE 13.5 mg PYRIDOXINE HYDROCHLORIDE (UNII: 68Y4CF58BV) (PYRIDOXINE - UNII:KV2JZ1BI6Z) PYRIDOXINE 1.05 mg FOLIC ACID (UNII: 935E97BOY8) (FOLIC ACID - UNII:935E97BOY8) FOLIC ACID 300 ug CYANOCOBALAMIN (UNII: P6YC3EG204) (CYANOCOBALAMIN - UNII:P6YC3EG204) CYANOCOBALAMIN 4.5 ug Inactive Ingredients Ingredient Name Strength SUCROSE (UNII: C151H8M554) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) D&C RED NO. 27 (UNII: 2LRS185U6K) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) STEARIC ACID (UNII: 4ELV7Z65AP) ASPARTAME (UNII: Z0H242BBR1) Product Characteristics Color purple (Dark purple) Score no score Shape ROUND Size 13mm Flavor GRAPE (Grape flavor) Imprint Code 107 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:59088-107-59 100 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 06/01/2011 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 06/01/2011 Labeler - PureTek Corporation (785961046)