Label: MINIDROPS- lubricant eye drops liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 62622-124-24, 62622-124-25, 62622-124-26 - Packager: Taiwan Biotech Co, Ltd
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated January 1, 2010
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
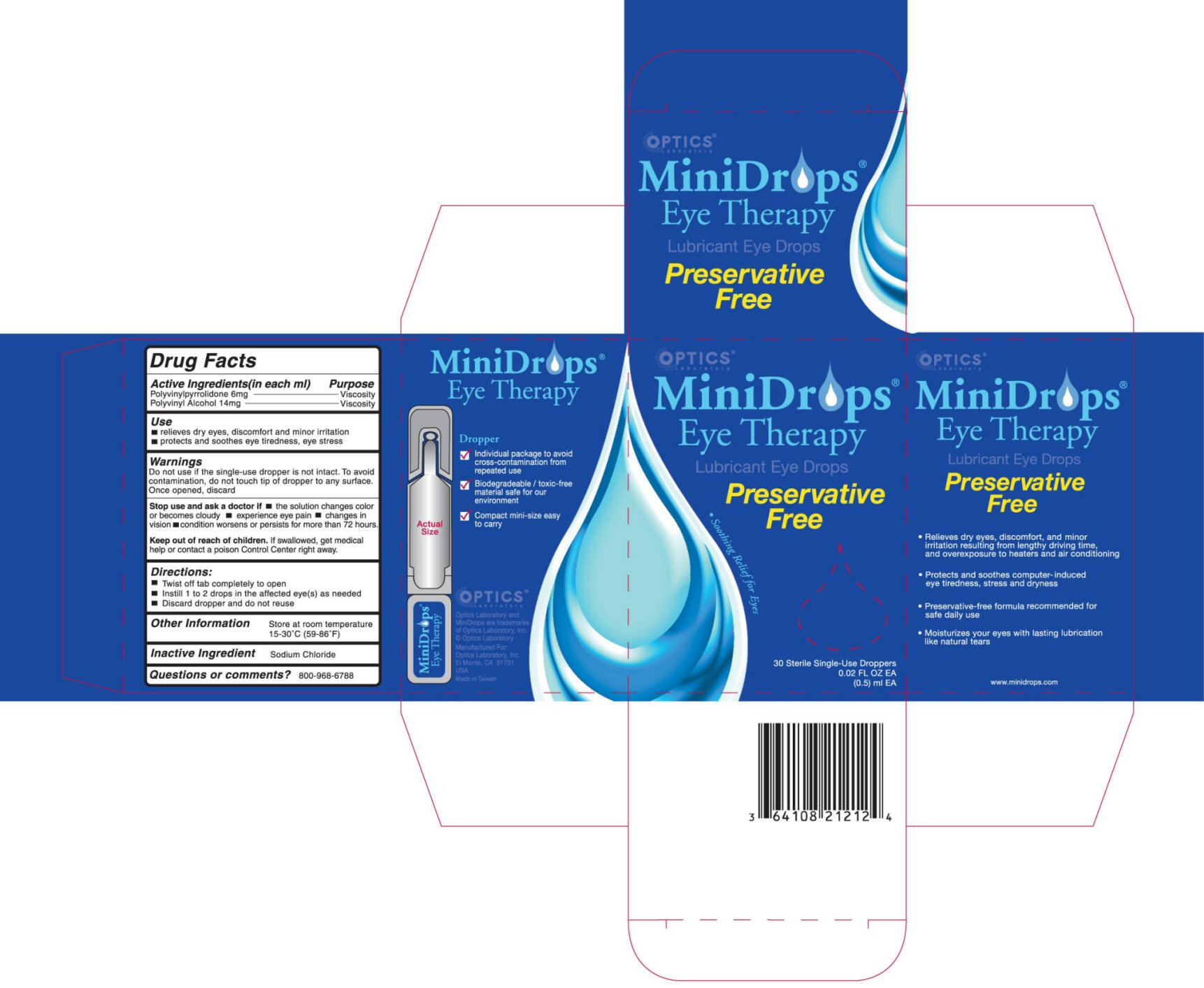
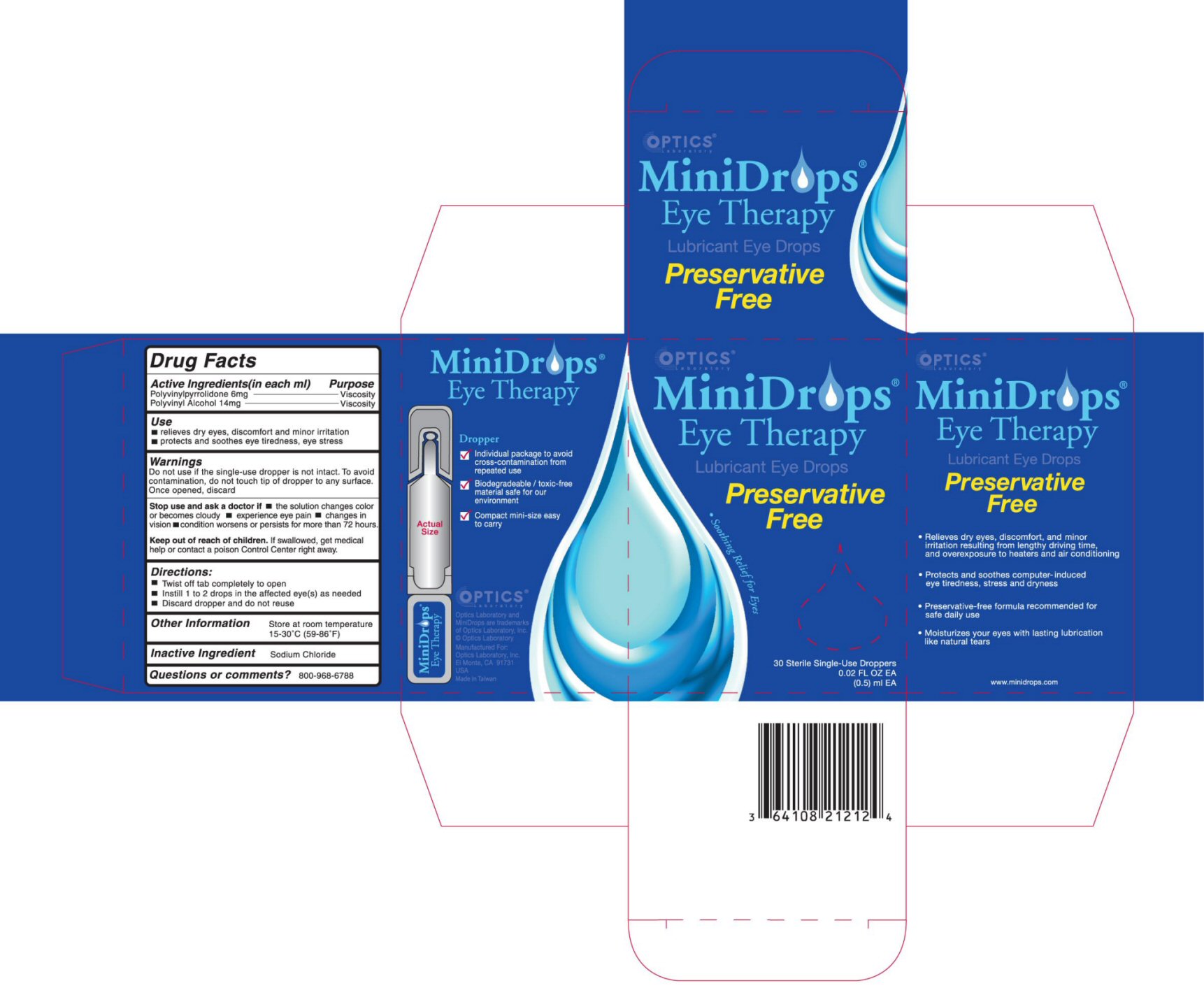
- ACTIVE INGREDIENTS (in each ml)
- PURPOSE
- USE
-
WARNINGS
DO NOT USE IF THE SINGLE-USE DROPPER IS NOT INTACT. TO AVOID CONTAMINATION, DO NOT TOUCH TIP OF DROPPER TO ANY SURFACE. ONCE OPENED, DISCARD.STOP USE AND ASK A DOCTOR IF
- THE SOLUTION CHANGES COLOR OR BECOMES CLOUDY
- EXPERIENCE EYE PAIN
- CHANGES IN VISION
- CONDITION WORSENS OR PERSISTS FOR MORE THAN 72 HOURS.
- DIRECTIONS
- OTHER INFORMATION
- INACTIVE INGREDIENT
- QUESTIONS OR COMMENTS?
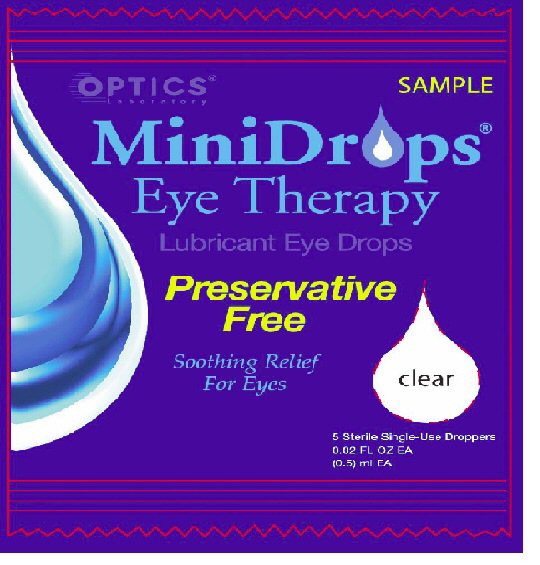
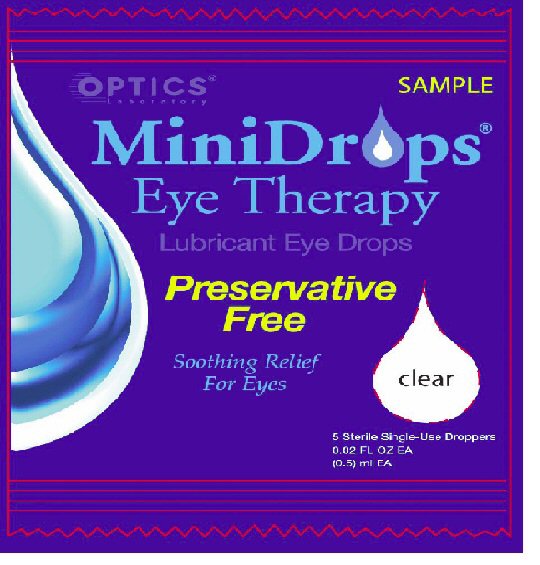
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
MINIDROPS
lubricant eye drops liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:62622-124 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength POVIDONE K30 (UNII: U725QWY32X) (POVIDONE K30 - UNII:U725QWY32X) POVIDONE K30 .6 mL in 100 mL POLYVINYL ALCOHOL (UNII: 532B59J990) (POLYVINYL ALCOHOL - UNII:532B59J990) POLYVINYL ALCOHOL 1.4 mL in 100 mL Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) .9 mL in 100 mL WATER (UNII: 059QF0KO0R) 97.1 mL in 100 mL Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:62622-124-25 5 in 1 BOX 1 NDC:62622-124-26 0.5 mL in 1 VIAL, SINGLE-USE 2 NDC:62622-124-24 30 in 1 BOX 2 NDC:62622-124-26 0.5 mL in 1 VIAL, SINGLE-USE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part349 04/01/1991 Labeler - Taiwan Biotech Co, Ltd (656127933) Registrant - Taiwan Biotech Co, Ltd (656127933) Establishment Name Address ID/FEI Business Operations Taiwan Biotech Co, Ltd 656127933 manufacture