Label: VRAYLAR- cariprazine capsule, gelatin coated
VRAYLAR- cariprazine kit
-
NDC Code(s):
61874-115-07,
61874-115-11,
61874-115-17,
61874-115-20, view more61874-115-30, 61874-115-31, 61874-115-90, 61874-130-07, 61874-130-11, 61874-130-20, 61874-130-30, 61874-130-31, 61874-130-90, 61874-145-07, 61874-145-30, 61874-145-90, 61874-160-07, 61874-160-30, 61874-160-90, 61874-170-08
- Packager: Allergan, Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated November 22, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Medication Guide: HTML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use VRAYLAR safely and effectively. See full prescribing information for VRAYLAR.
VRAYLAR® (cariprazine) capsules, for oral use
Initial U.S. Approval: 2015
WARNING: INCREASED MORTALITY IN ELDERLY PATIENTS WITH DEMENTIA-RELATED PSYCHOSIS and SUICIDAL THOUGHTS AND BEHAVIORS
See full prescribing information for complete boxed warning.
-
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. VRAYLAR is not approved for the treatment of patients with dementia-related psychosis. (5.1)
- Antidepressants increased the risk of suicidal thoughts and behaviors in pediatric and young adult patients. Closely monitor all antidepressant-treated patients for clinical worsening and emergence of suicidal thoughts and behaviors. Safety and effectiveness of VRAYLAR have not been established in pediatric patients (5.2, 8.4)
RECENT MAJOR CHANGES
Dosage and Administration (2.6) 11/2024
INDICATIONS AND USAGE
VRAYLAR is an atypical antipsychotic indicated for:
- Treatment of schizophrenia in adults (1)
- Acute treatment of manic or mixed episodes associated with bipolar I disorder in adults (1)
- Treatment of depressive episodes associated with bipolar I disorder (bipolar depression) in adults (1)
- Adjunctive therapy to antidepressants for the treatment of major depressive disorder (MDD) in adults (1)
DOSAGE AND ADMINISTRATION
- Administer VRAYLAR orally once daily with or without food (2)
Starting Dose Recommended Dose Schizophrenia (2.2) 1.5 mg daily 1.5 mg to 6 mg daily Bipolar Mania (2.3) 1.5 mg daily 3 mg to 6 mg daily Bipolar Depression (2.4) 1.5 mg daily 1.5 mg or 3 mg daily Adjunctive therapy to antidepressants for MDD (2.5) 1.5 mg daily 1.5 mg or 3 mg daily - Schizophrenia and Bipolar Mania: Maximum recommended daily dosage is 6 mg. Dosages above 6 mg daily do not confer significant benefit, but increase the risk of dose-related adverse reactions (2.2, 2.3)
- Bipolar Depression: Maximum recommended daily dosage is 3 mg (2.4)
- Adjunctive therapy for treatment of MDD: Maximum recommended daily dosage is 3 mg (2.5)
DOSAGE FORMS AND STRENGTHS
Capsules: 1.5 mg, 3 mg, 4.5 mg, and 6 mg (3)
CONTRAINDICATIONS
- Known hypersensitivity to VRAYLAR (4)
WARNINGS AND PRECAUTIONS
-
Cerebrovascular Adverse Reactions in Elderly Patients with Dementia-Related Psychosis: Increased incidence of cerebrovascular adverse reactions (e.g., stroke, transient ischemic attack) (5.3)
-
Neuroleptic Malignant Syndrome: Manage with immediate discontinuation and close monitoring (5.4)
-
Tardive Dyskinesia: Discontinue if appropriate (5.5)
-
Late-Occurring Adverse Reactions: Because of VRAYLAR’s long half-life, monitor for adverse reactions and patient response for several weeks after starting VRAYLAR and with each dosage change (5.6)
-
Metabolic Changes: Monitor for hyperglycemia/diabetes mellitus, dyslipidemia and weight gain (5.7)
-
Leukopenia, Neutropenia, and Agranulocytosis: Perform complete blood counts (CBC) in patients with pre-existing low white blood cell counts (WBC) or history of leukopenia or neutropenia. Consider discontinuing VRAYLAR if a clinically significant decline in WBC occurs in absence of other causative factors (5.8)
-
Orthostatic Hypotension and Syncope: Monitor heart rate and blood pressure and warn patients with known cardiovascular or cerebrovascular disease, and risk of dehydration or syncope (5.9)
-
Seizures: Use cautiously in patients with a history of seizures or with conditions that lower the seizure threshold (5.11)
- Potential for Cognitive and Motor Impairment: Use caution when operating machinery (5.12)
ADVERSE REACTIONS
Most common adverse reactions (incidence ≥ 5% and at least twice the rate of placebo) were (6.1):
- Schizophrenia: extrapyramidal symptoms and akathisia
- Bipolar mania: extrapyramidal symptoms, akathisia, dyspepsia, vomiting, somnolence, and restlessness
- Bipolar depression: nausea, akathisia, restlessness, and extrapyramidal symptoms
- Adjunctive treatment of MDD: akathisia, restlessness, fatigue, constipation, nausea, insomnia, increased appetite, dizziness, and extrapyramidal symptoms
To report SUSPECTED ADVERSE REACTIONS, contact AbbVie at 1-800-678-1605 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.DRUG INTERACTIONS
USE IN SPECIFIC POPULATIONS
- Pregnancy: Based on animal data, may cause fetal harm. (8.1)
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 11/2024
-
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. VRAYLAR is not approved for the treatment of patients with dementia-related psychosis. (5.1)
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
WARNING: INCREASED MORTALITY IN ELDERLY PATIENTS WITH DEMENTIA-RELATED PSYCHOSIS and SUICIDAL THOUGHTS AND BEHAVIORS
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 General Dosing Information
2.2 Recommended Dosage in Schizophrenia
2.3 Recommended Dosage in Manic or Mixed Episodes Associated with Bipolar I Disorder
2.4 Recommended Dosage in Depressive Episodes Associated with Bipolar I Disorder (Bipolar Depression)
2.5 Recommended Dosage for Adjunctive Therapy to Antidepressants for the Treatment of Major Depressive Disorder
2.6 Dosage Modifications for CYP3A4 Inhibitors and Inducers
2.7 Treatment Discontinuation
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Increased Mortality in Elderly Patients with Dementia-Related Psychosis
5.2 Suicidal Thoughts and Behaviors in Children, Adolescents and Young Adults
5.3 Cerebrovascular Adverse Reactions, Including Stroke, in Elderly Patients with Dementia-Related Psychosis
5.4 Neuroleptic Malignant Syndrome (NMS)
5.5 Tardive Dyskinesia
5.6 Late-Occurring Adverse Reactions
5.7 Metabolic Changes
5.8 Leukopenia, Neutropenia, and Agranulocytosis
5.9 Orthostatic Hypotension and Syncope
5.10 Falls
5.11 Seizures
5.12 Potential for Cognitive and Motor Impairment
5.13 Body Temperature Dysregulation
5.14 Dysphagia
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Hepatic Impairment
8.7 Renal Impairment
8.8 Smoking
8.9 Other Specific Populations
9 DRUG ABUSE AND DEPENDENCE
9.1 Controlled Substance
9.2 Abuse
9.3 Dependence
10 OVERDOSAGE
10.1 Human Experience
10.2 Management of Overdosage
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
13.2 Animal Toxicology and/or Pharmacology
14 CLINICAL STUDIES
14.1 Schizophrenia
14.2 Manic or Mixed Episodes Associated with Bipolar I Disorder
14.3 Depressive Episodes Associated with Bipolar I Disorder (Bipolar Depression)
14.4 Adjunctive Treatment of Major Depressive Disorder
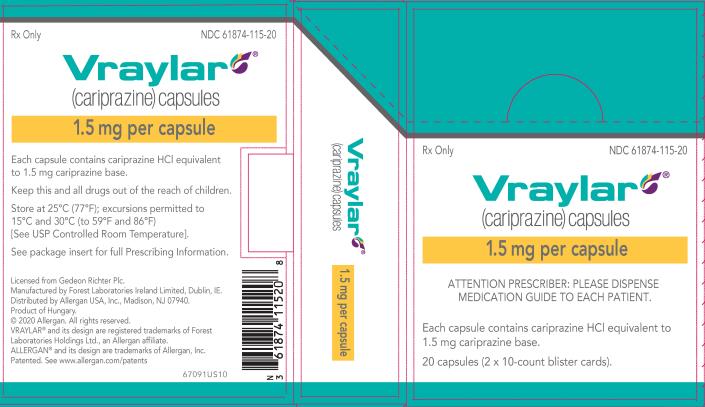
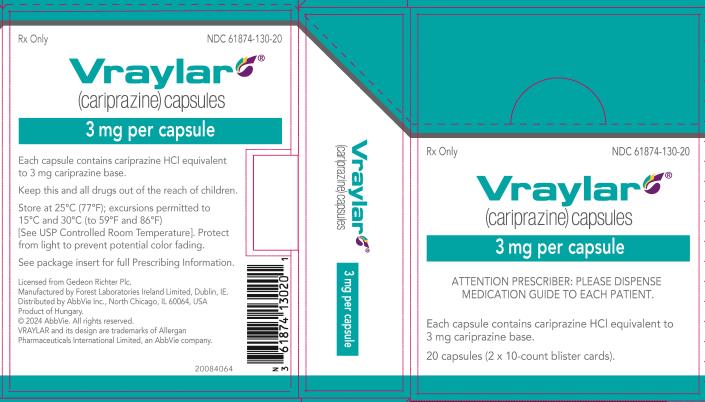
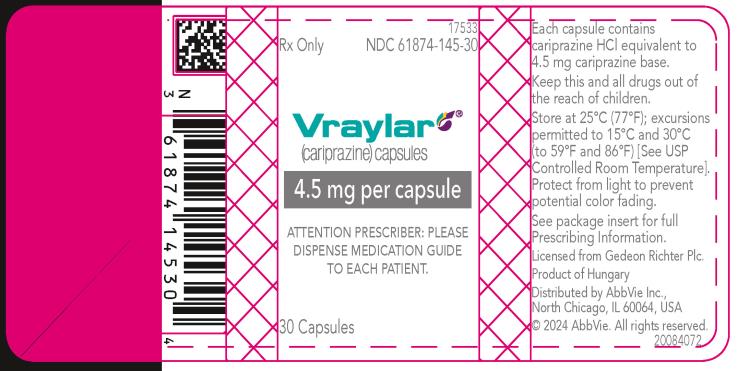
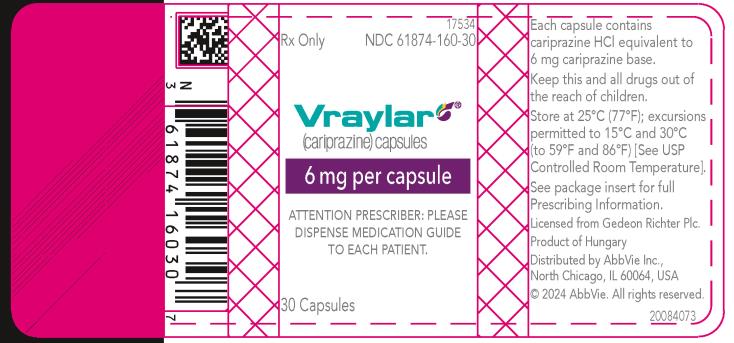
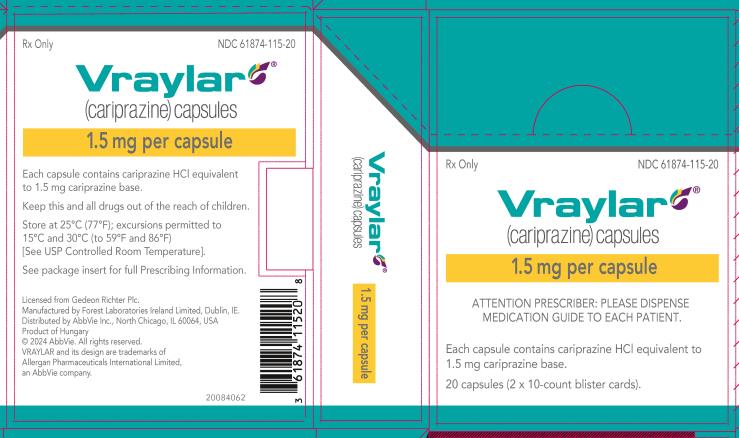
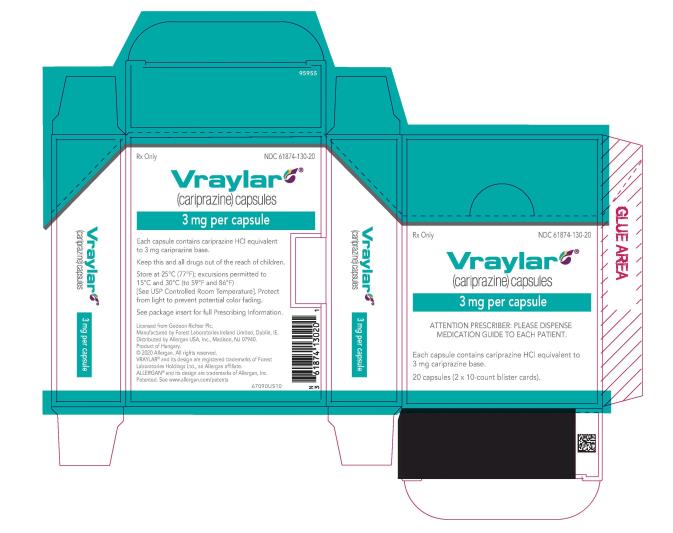
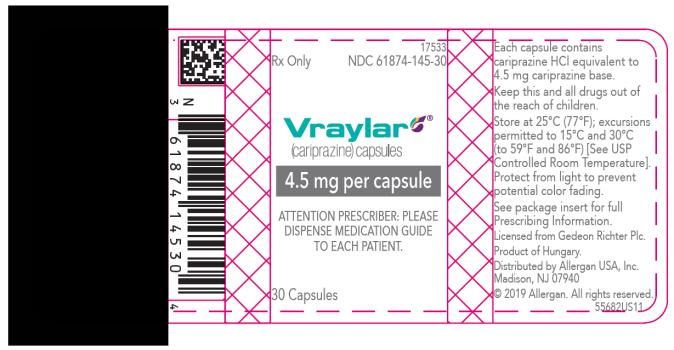
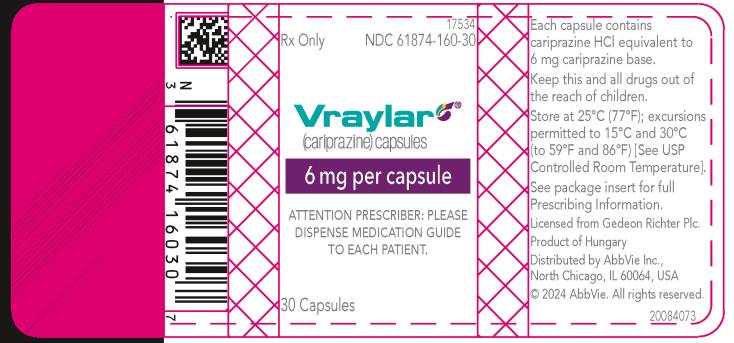
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
16.2 Storage and Handling
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
-
BOXED WARNING
(What is this?)
WARNING: INCREASED MORTALITY IN ELDERLY PATIENTS WITH DEMENTIA-RELATED PSYCHOSIS and SUICIDAL THOUGHTS AND BEHAVIORS
Increased Mortality in Elderly Patients with Dementia-Related Psychosis
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. VRAYLAR is not approved for the treatment of patients with dementia-related psychosis [see Warnings and Precautions (5.1)].
Suicidal Thoughts and Behaviors
Antidepressants increased the risk of suicidal thoughts and behaviors in pediatric and young adult patients in short-term studies. Closely monitor all antidepressant-treated patients for clinical worsening, and for the emergence of suicidal thoughts and behaviors [see Warnings and Precautions (5.2)]. The safety and effectiveness of VRAYLAR have not been established in pediatric patients [see Use in Specific Populations (8.4)].
-
1
INDICATIONS AND USAGE
VRAYLAR® is indicated for:
- Treatment of schizophrenia in adults [see Clinical Studies (14.1)]
- Acute treatment of manic or mixed episodes associated with bipolar I disorder in adults [see Clinical Studies (14.2)]
- Treatment of depressive episodes associated with bipolar I disorder (bipolar depression) in adults [see Clinical Studies (14.3)]
- Adjunctive therapy to antidepressants for the treatment of major depressive disorder (MDD) in adults [see Clinical Studies (14.4)]
- Treatment of schizophrenia in adults [see Clinical Studies (14.1)]
-
2
DOSAGE AND ADMINISTRATION
2.1 General Dosing Information
VRAYLAR is given orally once daily and can be taken with or without food.
Because of the long half-life of cariprazine and its active metabolites, changes in dose will not be fully reflected in plasma for several weeks. Prescribers should monitor patients for adverse reactions and treatment response for several weeks after starting VRAYLAR and after each dosage change [see Warnings and Precautions (5.6), Clinical Pharmacology (12.3)].
2.2 Recommended Dosage in Schizophrenia
The starting dosage of VRAYLAR is 1.5 mg orally once daily. The recommended dosage range is 1.5 mg to 6 mg orally once daily. The dosage can be increased to 3 mg on Day 2. Depending upon clinical response and tolerability, further dose adjustments can be made in 1.5 mg or 3 mg increments. The maximum recommended dosage is 6 mg orally once daily. In short-term controlled trials, dosages above 6 mg daily do not confer increased effectiveness sufficient to outweigh dose-related adverse reactions [see Adverse Reactions (6.1), Clinical Studies (14.1)].
2.3 Recommended Dosage in Manic or Mixed Episodes Associated with Bipolar I Disorder
The starting dosage of VRAYLAR is 1.5 mg orally once daily. Increase the dosage to 3 mg orally once daily on Day 2. The recommended dosage range is 3 mg to 6 mg orally once daily. Depending upon clinical response and tolerability, further dose adjustments can be made in 1.5 mg or 3 mg increments. The maximum recommended dosage is 6 mg orally once daily. In short-term controlled trials, dosages above 6 mg daily do not confer increased effectiveness sufficient to outweigh dose-related adverse reactions [see Adverse Reactions (6.1), Clinical Studies (14.2)].
2.4 Recommended Dosage in Depressive Episodes Associated with Bipolar I Disorder (Bipolar Depression)
The starting dosage of VRAYLAR is 1.5 mg orally once daily. Depending upon clinical response and tolerability, the dosage can be increased to 3 mg orally once daily on Day 15. Maximum recommended dosage is 3 mg orally once daily.
2.5 Recommended Dosage for Adjunctive Therapy to Antidepressants for the Treatment of Major Depressive Disorder
The starting dosage of VRAYLAR is 1.5 mg orally once daily. Depending upon clinical response and tolerability, the dosage can be increased to 3 mg orally once daily on Day 15. In clinical trials, dosage titration at intervals of less than 14 days resulted in a higher incidence of adverse reactions [see Adverse Reactions (6.1)]. Maximum recommended dosage is 3 mg orally once daily.
2.6 Dosage Modifications for CYP3A4 Inhibitors and Inducers
Initiating VRAYLAR While Taking a Strong or Moderate CYP3A4 Inhibitor
Dosage modifications for the starting dosage of VRAYLAR in patients taking a strong or moderate CYP3A4 inhibitor are presented in Table 1:
Table 1: Dosage Modifications for the Starting Dosage of VRAYLAR in Patients Taking a Strong or Moderate CYP3A4 Inhibitor
VRAYLAR Starting Dosage When Taking a Strong
CYP3A4 InhibitorWhen Taking a Moderate CYP3A4 Inhibitor Schizophrenia Start at 1.5 mg orally every 3 days; increase to 1.5 mg orally every other day, if needed* Start at 1.5 mg orally every other day; increase to 1.5 mg orally daily, if needed* Bipolar Mania
Bipolar Depression
1.5 mg orally every 3 days 1.5 mg orally every other day Adjunctive therapy for treatment of MDD
*Depending upon clinical response and tolerability.
Initiating a Strong or Moderate CYP3A4 Inhibitor While Taking a Stable Dosage of VRAYLAR
Dosage recommendations for patients initiating a strong or moderate CYP3A4 inhibitor while on a stable dose of VRAYLAR (see Table 2):e:
Table 2: Dosage Modifications for VRAYLAR When Initiating a Strong or Moderate CYP3A4 Inhibitor and While Taking a Stable Dose of VRAYLAR
Currently on VRAYLAR Dosage VRAYLAR Dosage When Initiating a Strong CYP3A4 Inhibitor VRAYLAR Dosage When Initiating a Moderate CYP3A4 Inhibitor 1.5 or 3 mg once daily 1.5 mg orally every 3 days 1.5 mg orally every other day 4.5 or 6 mg once daily 1.5 mg orally every other day 1.5 mg orally once daily When the strong or moderate CYP3A4 inhibitor is discontinued, the VRAYLAR dosage may need to be increased based on clinical response and tolerability [see Drug Interactions (7)].
Dosage Modifications for Patients Concomitantly Taking VRAYLAR with CYP3A4 Inducers
Concomitant use of VRAYLAR and a CYP3A4 inducer has not been evaluated and is not recommended [see Dosage and Administration (2.1), Warnings and Precautions (5.6), Drug Interactions (7), Clinical Pharmacology (12.3)].
2.7 Treatment Discontinuation
Following discontinuation of VRAYLAR, the decline in plasma concentrations of active drug and metabolites may not be immediately reflected in patients’ clinical symptoms; the plasma concentration of cariprazine and its active metabolites will decline by 50% in ~1 week [see Clinical Pharmacology (12.3)]. There are no systematically collected data to specifically address switching patients from VRAYLAR to other antipsychotics or concerning concomitant administration with other antipsychotics.
-
3
DOSAGE FORMS AND STRENGTHS
VRAYLAR (cariprazine) capsules are available in four strengths.
- 1.5 mg capsules: White cap and body imprinted with “FL 1.5”
- 3 mg capsules: Green to blue-green cap and white body imprinted with “FL 3”
- 4.5 mg capsules: Green to blue-green cap and body imprinted with “FL 4.5”
- 6 mg capsules: Purple cap and white body imprinted with “FL 6”
- 1.5 mg capsules: White cap and body imprinted with “FL 1.5”
- 4 CONTRAINDICATIONS
-
5
WARNINGS AND PRECAUTIONS
5.1 Increased Mortality in Elderly Patients with Dementia-Related Psychosis
Antipsychotic drugs increase the all-cause risk of death in elderly patients with dementia-related psychosis. Analyses of 17 dementia-related psychosis placebo-controlled trials (modal duration of 10 weeks and largely in patients taking atypical antipsychotic drugs) revealed a risk of death in the drug-treated patients of between 1.6 to 1.7 times that in placebo-treated patients. Over the course of a typical 10-week controlled trial, the rate of death in drug-treated patients was about 4.5%, compared to a rate of about 2.6% in placebo-treated patients.
Although the causes of death were varied, most of the deaths appeared to be either cardiovascular (e.g., heart failure, sudden death) or infectious (e.g., pneumonia) in nature. VRAYLAR is not approved for the treatment of patients with dementia-related psychosis [see Boxed Warning, Warnings and Precautions (5.3)].
5.2 Suicidal Thoughts and Behaviors in Children, Adolescents and Young Adults
In pooled analyses of placebo-controlled trials of antidepressant drugs (SSRIs and other antidepressant classes) that included approximately 77,000 adult patients and 4,500 pediatric patients, the incidence of suicidal thoughts and behaviors in antidepressant-treated patients age 24 years and younger was greater than in placebo-treated patients. There was considerable variation in risk of suicidal thoughts and behaviors among drugs, but there was an increased risk identified in young patients for most drugs studied. There were differences in absolute risk of suicidal thoughts and behaviors across the different indications, with the highest incidence in patients with MDD. The drug-placebo differences in the number of cases of suicidal thoughts and behaviors per 1000 patients treated are provided in Table 3.
Table 3: Risk Differences of the Number of Patients of Suicidal Thoughts and Behavior in the Pooled Placebo-Controlled Trials of Antidepressants in Pediatric* and Adult Patients Age Range Drug-Placebo Difference in Number of Patients of Suicidal Thoughts or Behaviors per 1000 Patients Treated Increases Compared to Placebo <18 years old 14 additional patients 18-24 years old 5 additional patients Decreases Compared to Placebo 25-64 years old 1 fewer patient ≥65 years old 6 fewer patients * VRAYLAR is not approved for use in pediatric patients.
It is unknown whether the risk of suicidal thoughts and behaviors in children, adolescents, and young adults extends to longer-term use, i.e., beyond four months. However, there is substantial evidence from placebo-controlled maintenance trials in adults with MDD that antidepressants delay the recurrence of depression and that depression itself is a risk factor for suicidal thoughts and behaviors.
Monitor all antidepressant-treated patients for any indication for clinical worsening and emergence of suicidal thoughts and behaviors, especially during the initial few months of drug therapy, and at times of dosage changes. Counsel family members or caregivers of patients to monitor for changes in behavior and to alert the healthcare provider. Consider changing the therapeutic regimen, including possibly discontinuing VRAYLAR, in patients whose depression is persistently worse, or who are experiencing emergent suicidal thoughts or behaviors.
5.3 Cerebrovascular Adverse Reactions, Including Stroke, in Elderly Patients with Dementia-Related Psychosis
In placebo-controlled trials in elderly patients with dementia, patients randomized to risperidone, aripiprazole, and olanzapine had a higher incidence of stroke and transient ischemic attack, including fatal stroke. VRAYLAR is not approved for the treatment of patients with dementia-related psychosis [see Boxed Warning, Warnings and Precautions (5.1)].
5.4 Neuroleptic Malignant Syndrome (NMS)
Neuroleptic Malignant Syndrome (NMS), a potentially fatal symptom complex, has been reported in association with administration of antipsychotic drugs. Clinical manifestations of NMS are hyperpyrexia, muscle rigidity, delirium, and autonomic instability. Additional signs may include elevated creatine phosphokinase, myoglobinuria (rhabdomyolysis), and acute renal failure.
If NMS is suspected, immediately discontinue VRAYLAR and provide intensive symptomatic treatment and monitoring.
5.5 Tardive Dyskinesia
Tardive dyskinesia, a syndrome consisting of potentially irreversible, involuntary, dyskinetic movements, may develop in patients treated with antipsychotic drugs, including VRAYLAR. The risk appears to be highest among the elderly, especially elderly women, but it is not possible to predict which patients are likely to develop the syndrome. Whether antipsychotic drug products differ in their potential to cause tardive dyskinesia is unknown.
The risk of tardive dyskinesia and the likelihood that it will become irreversible increase with the duration of treatment and the cumulative dose. The syndrome can develop after a relatively brief treatment period, even at low doses. It may also occur after discontinuation of treatment.
Tardive dyskinesia may remit, partially or completely, if antipsychotic treatment is discontinued. Antipsychotic treatment itself, however, may suppress (or partially suppress) the signs and symptoms of the syndrome, possibly masking the underlying process. The effect that symptomatic suppression has upon the long-term course of tardive dyskinesia is unknown.
Given these considerations, VRAYLAR should be prescribed in a manner most likely to reduce the risk of tardive dyskinesia. Chronic antipsychotic treatment should generally be reserved for patients: 1) who suffer from a chronic illness that is known to respond to antipsychotic drugs; and 2) for whom alternative, effective, but potentially less harmful treatments are not available or appropriate. In patients who do require chronic treatment, use the lowest dose and the shortest duration of treatment producing a satisfactory clinical response should be sought. Periodically reassess the need for continued treatment.
If signs and symptoms of tardive dyskinesia appear in a patient on VRAYLAR, drug discontinuation should be considered. However, some patients may require treatment with VRAYLAR despite the presence of the syndrome.
5.6 Late-Occurring Adverse Reactions
Adverse reactions may first appear several weeks after the initiation of VRAYLAR treatment, probably because plasma levels of cariprazine and its major metabolites accumulate over time. As a result, the incidence of adverse reactions in short-term trials may not reflect the rates after longer term exposures [see Dosage and Administration (2.1), Adverse Reactions (6.1), Clinical Pharmacology (12.3)].
Monitor for adverse reactions, including extrapyramidal symptoms (EPS) or akathisia, and patient response for several weeks after a patient has begun VRAYLAR and after each dosage increase. Consider reducing the dose or discontinuing the drug.
5.7 Metabolic Changes
Atypical antipsychotic drugs, including VRAYLAR, have caused metabolic changes, including hyperglycemia, diabetes mellitus, dyslipidemia, and weight gain. There have been reports of hyperglycemia in patients treated with VRAYLAR. Although all drugs in the class to date have been shown to produce some metabolic changes, each drug has its own specific risk profile.
Hyperglycemia and Diabetes Mellitus
Hyperglycemia, in some cases extreme and associated with ketoacidosis or hyperosmolar coma or death, has been reported in patients treated with atypical antipsychotics. Assess fasting plasma glucose before or soon after initiation of antipsychotic medication and monitor periodically during long-term treatment.
Schizophrenia
In the 6-week, placebo-controlled trials of adult patients with schizophrenia, the proportion of patients with shifts in fasting glucose from normal (<100 mg/dL) to high (≥126 mg/dL) and borderline (≥100 and <126 mg/dL) to high were similar in patients treated with VRAYLAR and placebo. In the long-term, open-label schizophrenia studies, 4% patients with normal hemoglobin A1c baseline values developed elevated levels (≥ 6.5%).
Bipolar Disorder
In six placebo-controlled trials up to 8-weeks of adult patients with bipolar disorder (mania or depression), the proportion of patients with shifts in fasting glucose from normal (<100 mg/dL) to high (≥126 mg/dL) and borderline (≥100 and <126 mg/dL) to high were similar in patients treated with VRAYLAR and placebo. In the long-term, open-label bipolar disorder studies, 4% patients with normal hemoglobin A1c baseline values developed elevated levels (≥ 6.5%).
Adjunctive Treatment of Major Depressive Disorder
In two 6-week placebo-controlled trials of adult patients with major depressive disorder, the proportion of patients with shifts in fasting glucose from normal (<100 mg/dL) to high (≥126 mg/dL) was greatest in the VRAYLAR 3 mg per day + antidepressant therapy arm (3.2%) compared with those taking VRAYLAR 1.5 mg per day + antidepressant therapy (2%) or those placebo-treated (1.3%). The proportion of patients with shifts from normal to borderline (≥100 and <126 mg/dL) or from borderline to high were similar in patients treated with VRAYLAR and placebo. In a long-term, open-label adjunctive treatment of MDD study, 7% patients with normal hemoglobin A1c baseline values developed elevated levels (> 6%).
In one 8-week placebo-controlled trial of adult patients with major depressive disorder, the changes from baseline to end of the trial in fasting glucose were similar among the VRAYLAR and placebo + antidepressant therapy treatment groups. During the 8-week trial, serum insulin levels increased by 12 pmol/L in the VRAYLAR 1 mg to 2 mg per day group, 20 pmol/L in the VRAYLAR 2 mg to 4.5 mg per day group, and 8.5 pmol/L in the placebo group.
Dyslipidemia
Atypical antipsychotics cause adverse alterations in lipids. Before or soon after initiation of antipsychotic medication, obtain a fasting lipid profile at baseline and monitor periodically during treatment.
Schizophrenia
In the 6-week, placebo-controlled trials of adult patients with schizophrenia, the proportion of patients with shifts in fasting total cholesterol, LDL, HDL, and triglycerides were similar in patients treated with VRAYLAR and placebo.
Bipolar Disorder
In six placebo-controlled trials up to 8-weeks of adult patients with bipolar disorder (mania or depression), the proportion of patients with shifts in fasting total cholesterol, LDL, HDL, and triglycerides were similar in patients treated with VRAYLAR and placebo.
Adjunctive Treatment of Major Depressive Disorder
In two 6-week placebo-controlled trials of adult patients with major depressive disorder, the proportion of patients with shifts in total cholesterol, fasting LDL, HDL, and fasting triglycerides were similar in patients treated with VRAYLAR and placebo.
Weight Gain
Weight gain has been observed with use of atypical antipsychotics, including VRAYLAR. Monitor weight at baseline and frequently thereafter. Tables 4, 5, 6, and 7 show the change in body weight occurring from baseline to endpoint in 6-week trials of schizophrenia, 3-week bipolar mania trials, 6-week and 8-week bipolar depression trials, and 6-week and 8-week trials of adjunctive treatment for major depressive disorder, respectively.
Table 4. Change in Body Weight (kg) in 6-Week Schizophrenia Trials VRAYLAR* Placebo
(N=573)1.5 - 3 mg/day
(N=512)4.5 - 6
mg/day
(N=570)9 - 12⸰
mg/day
(N=203)Mean Change at Endpoint +0.3 +0.8 +1 +1 Proportion of Patients with Weight Increase (≥7%) 5% 8% 8% 17% *Data shown by modal daily dose, defined as most frequently administered dose per patient
⸰The maximum recommended daily dose is 6 mg. Doses above 6 mg daily do not confer increased effectiveness sufficient to outweigh dose-related adverse reactions.In long-term, uncontrolled trials with VRAYLAR in schizophrenia, the mean changes from baseline in weight at 12, 24, and 48 weeks were 1.2 kg, 1.7 kg, and 2.5 kg, respectively.
Table 5. Change in Body Weight (kg) in 3-Week Bipolar Mania Trials VRAYLAR* Placebo
(N=439)3 - 6
mg/day
(N=259)9 - 12⸰
mg/day
(N=360)Mean Change at Endpoint +0.2 +0.5 +0.6 Proportion of Patients with Weight Increase (≥7%) 2% 1% 3% *Data shown by modal daily dose, defined as most frequently administered dose per patient
⸰The maximum recommended daily dose is 6 mg. Doses above 6 mg daily do not confer increased effectiveness sufficient to outweigh dose-related adverse reactions.Table 6. Change in Body Weight (kg) in two 6-Week and one 8-Week Bipolar Depression Trials VRAYLAR Placebo 1.5 mg/day 3 mg/day (N=463) (N=467) (N=465) Mean Change at Endpoint -0.1 +0.7 +0.4 Proportion of Patients with Weight Increase (≥7%) 1% 3% 3% Table 7. Change in Body Weight (kg) in two 6-Week and one 8-Week Adjunctive Treatment for Major Depressive Disorder Trials VRAYLAR 6-week Trials Placebo +ADT 1.5 mg/day +ADT 3 mg/day +ADT (N=503) (N=502) (N=503) Mean Change at Endpoint +0.2 +0.7 +0.7 Proportion of Patients with Weight Increase (≥7%) 1% 2% 2% 8-week Trial Placebo +
ADT
(N=266)1 to 2 mg/day + ADT
(N=273)2 to 4.5 mg/day + ADT
(N=273)Mean Change at Endpoint 0 +0.9 +0.9 Proportion of Patients with Weight Increase (≥7%) 2% 2% 3% In the long-term, open-label adjunctive treatment of MDD trial, 2 patients (0.6%) discontinued due to weight increase. VRAYLAR was associated with mean change from baseline in weight of 1.7 kg at Week 26. In the long-term, open-label adjunctive treatment of MDD trial, 19% of patients demonstrated a ≥7% increase in body weight, and 5% demonstrated a ≥7% decrease in body weight.
5.8 Leukopenia, Neutropenia, and Agranulocytosis
Leukopenia and neutropenia have been reported during treatment with antipsychotic agents, including VRAYLAR. Agranulocytosis (including fatal cases) has been reported with other agents in the class.
Possible risk factors for leukopenia and neutropenia include pre-existing low white blood cell count (WBC) or absolute neutrophil count (ANC) and history of drug-induced leukopenia or neutropenia. In patients with a pre-existing low WBC or ANC or a history of drug-induced leukopenia or neutropenia, perform a complete blood count (CBC) frequently during the first few months of therapy. In such patients, consider discontinuation of VRAYLAR at the first sign of a clinically significant decline in WBC in the absence of other causative factors.
Monitor patients with clinically significant neutropenia for fever or other symptoms or signs of infection and treat promptly if such symptoms or signs occur. Discontinue VRAYLAR in patients with absolute neutrophil count < 1000/mm3 and follow their WBC until recovery.
5.9 Orthostatic Hypotension and Syncope
Atypical antipsychotics cause orthostatic hypotension and syncope. Generally, the risk is greatest during initial dose titration and when increasing the dose. Symptomatic orthostatic hypotension was infrequent in trials of VRAYLAR and was not more frequent on VRAYLAR than placebo. Syncope was not observed.
Orthostatic vital signs should be monitored in patients who are vulnerable to hypotension (e.g., elderly patients, patients with dehydration, hypovolemia, and concomitant treatment with antihypertensive medications), patients with known cardiovascular disease (history of myocardial infarction, ischemic heart disease, heart failure, or conduction abnormalities), and patients with cerebrovascular disease. VRAYLAR has not been evaluated in patients with a recent history of myocardial infarction or unstable cardiovascular disease. Such patients were excluded from pre-marketing clinical trials.
5.10 Falls
Antipsychotics, including VRAYLAR, may cause somnolence, postural hypotension, motor and sensory instability, which may lead to falls and, consequently, fractures or other injuries. For patients with diseases, conditions, or medications that could exacerbate these effects, complete fall risk assessments when initiating antipsychotic treatment and recurrently for patients on long-term antipsychotic therapy.
5.11 Seizures
Like other antipsychotic drugs, VRAYLAR may cause seizures. This risk is greatest in patients with a history of seizures or with conditions that lower the seizure threshold. Conditions that lower the seizure threshold may be more prevalent in older patients.
5.12 Potential for Cognitive and Motor Impairment
VRAYLAR, like other antipsychotics, may cause somnolence and has the potential to impair judgment, thinking, or motor skills.
In 6-week schizophrenia trials, somnolence (hypersomnia, sedation, and somnolence) was reported in 7% of VRAYLAR-treated patients compared to 6% of placebo-treated patients. In 3-week bipolar mania trials, somnolence was reported in 8% of VRAYLAR-treated patients compared to 4% of placebo-treated patients. In two 6-week and one 8-week trials of depressive episodes of bipolar I disorder, VRAYLAR-treated patients reported 7% somnolence and 4% in the placebo-treated patients. In 6-week adjunctive treatment of major depressive disorder trials, somnolence was reported in 6% of VRAYLAR-treated patients compared to 4% of placebo-treated patients. In one 8-week adjunctive treatment of major depressive disorder trial, somnolence was reported in 11% of VRAYLAR-treated patients compared to 6% of placebo-treated patients.
Patients should be cautioned about operating hazardous machinery, including motor vehicles, until they are reasonably certain that therapy with VRAYLAR does not affect them adversely.
5.13 Body Temperature Dysregulation
Atypical antipsychotics may disrupt the body’s ability to reduce core body temperature. Strenuous exercise, exposure to extreme heat, dehydration, and anticholinergic medications may contribute to an elevation in core body temperature; use VRAYLAR with caution in patient who may experience these conditions.
-
6
ADVERSE REACTIONS
The following adverse reactions are discussed in more detail in other sections of the labeling:
- Increased Mortality in Elderly Patients with Dementia-Related Psychosis [see Boxed Warning and Warnings and Precautions (5.1)]
- Suicidal Thoughts and Behaviors [see Boxed Warning and Warnings and Precautions (5.2)]
- Cerebrovascular Adverse Reactions, Including Stroke, in Elderly Patients with Dementia-Related Psychosis [see Warnings and Precautions (5.3)]
- Neuroleptic Malignant Syndrome [see Warnings and Precautions (5.4)]
- Tardive Dyskinesia [see Warnings and Precautions (5.5)]
- Late Occurring Adverse Reactions [see Warnings and Precautions (5.6)]
- Metabolic Changes [see Warnings and Precautions (5.7)]
- Leukopenia, Neutropenia, and Agranulocytosis [see Warnings and Precautions (5.8)]
- Orthostatic Hypotension and Syncope [see Warnings and Precautions (5.9)]
- Falls [see Warnings and Precautions (5.10)]
- Seizures [see Warnings and Precautions (5.11)]
- Potential for Cognitive and Motor Impairment [see Warnings and Precautions (5.12)]
- Body Temperature Dysregulation [see Warnings and Precautions (5.13)]
- Dysphagia [see Warnings and Precautions (5.14)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The information below is derived from an integrated clinical study database for VRAYLAR consisting of 6,722 adult patients exposed to one or more doses of VRAYLAR for the treatment of schizophrenia, manic or mixed episodes associated with bipolar I disorder, bipolar depression, and adjunctive treatment of major depressive disorder in placebo-controlled studies. This experience corresponds with a total experience of 1,182.8 patient-years. A total of 4,329 VRAYLAR-treated patients had at least 6 weeks and 296 VRAYLAR-treated patients had at least 48 weeks of exposure.
Patients with Schizophrenia
The following findings are based on four placebo-controlled, 6-week schizophrenia trials with VRAYLAR doses ranging from 1.5 to 12 mg once daily. The maximum recommended dosage is 6 mg daily.
Adverse Reactions Associated with Discontinuation of Treatment: There was no single adverse reaction leading to discontinuation that occurred at a rate of ≥ 2% in VRAYLAR-treated patients and at least twice the rate of placebo.
Common Adverse Reactions (≥ 5% and at least twice the rate of placebo): extrapyramidal symptoms and akathisia.
Adverse Reactions with an incidence of ≥ 2% and greater than placebo, at any dose are shown in Table 8.
Table 8. Adverse Reactions Occurring in ≥ 2% of VRAYLAR-treated Patients and > Placebo-treated Adult Patients in 6-Week Schizophrenia Trials VRAYLAR* System Organ Class /
Preferred Term
Placebo
(N= 584)
(%)1.5 to 3 mg/day
(N=539)
(%)4.5 to 6 mg/day
(N=575)
(%)9 to 12 mg/day⸰
(N=203)
(%)Cardiac Disorders Tachycardiaa 1 2 2 3 Gastrointestinal Disorders Abdominal painb 5 3 4 7 Constipation 5 6 7 10 Diarrheac 3 1 4 5 Dry Mouth 2 1 2 3 Dyspepsia 4 4 5 5 Nausea 5 5 7 8 Toothache 4 3 3 6 Vomiting 3 4 5 5 General Disorders/Administration Site Conditions Fatigued 1 1 3 2 Infections and Infestations Nasopharyngitis 1 1 1 2 Urinary tract infection 1 1 <1 2 Investigations Blood creatine phosphokinase increased 1 1 2 3 Hepatic enzyme increasede <1 1 1 2 Weight increased 1 3 2 3 Metabolism and Nutrition Disorders Decreased appetite 2 1 3 2 Musculoskeletal and Connective Tissue Disorders Arthralgia 1 2 1 2 Back pain 2 3 3 1 Pain in extremity 3 2 2 4 Nervous System Disorders Akathisia 4 9 13 14 Extrapyramidal symptomsf 8 15 19 20 Headacheg 13 9 11 18 Somnolenceh 5 5 8 10 Dizziness 2 3 5 5 Psychiatric Disorders Agitation 4 3 5 3 Insomniai 11 12 13 11 Restlessness 3 4 6 5 Anxiety 4 6 5 3 Respiratory, Thoracic and Mediastinal Disorders Cough 2 1 2 4 Skin and Subcutaneous Disorders Rash 1 <1 1 2 Vascular Disorders Hypertensionj 1 2 3 6 Note: Figures rounded to the nearest integer
* Data shown by modal daily dose, defined as most frequently administered dose per patient
aTachycardia terms: heart rate increased, sinus tachycardia, tachycardia
bAbdominal pain terms: abdominal discomfort, abdominal pain, abdominal pain lower, abdominal pain upper, gastrointestinal pain
cDiarrhea terms: diarrhea, frequent bowel movements
dFatigue terms: asthenia, fatigue
eHepatic enzyme increase terms: alanine aminotransferase increased, aspartate aminotransferase increased, hepatic enzyme increased
fExtrapyramidal Symptoms terms: bradykinesia, cogwheel rigidity, drooling, dyskinesia, dystonia, extrapyramidal disorder, hypokinesia, masked facies, muscle rigidity, muscle tightness, Musculoskeletal stiffness, oculogyric crisis, oromandibular dystonia, parkinsonism, salivary hypersecretion, tardive dyskinesia, torticollis, tremor, trismus
gHeadache terms: headache, tension headache
hSomnolence terms: hypersomnia, sedation, somnolence
iInsomnia terms: initial insomnia, insomnia, middle insomnia, terminal insomnia
jHypertension terms: blood pressure diastolic increased, blood pressure increased, blood pressure systolic increased, hypertension
⸰ The maximum recommended daily dose is 6 mg. Doses above 6 mg daily do not confer increased effectiveness sufficient to outweigh dose-related adverse reactions.Patients with Bipolar Mania
The following findings are based on three placebo-controlled, 3-week bipolar mania trials with VRAYLAR doses ranging from 3 to 12 mg once daily. The maximum recommended dosage is 6 mg daily.
Adverse Reactions Associated with Discontinuation of Treatment: The adverse reaction leading to discontinuation that occurred at a rate of ≥ 2% in VRAYLAR-treated patients and at least twice the rate of placebo was akathisia (2%). Overall, 12% of the patients who received VRAYLAR discontinued treatment due to an adverse reaction, compared with 7% of placebo-treated patients in these trials.
Common Adverse Reactions (≥ 5% and at least twice the rate of placebo): extrapyramidal symptoms, akathisia, dyspepsia, vomiting, somnolence, and restlessness.
Adverse Reactions with an incidence of ≥ 2% and greater than placebo at any dose are shown in Table 9.
Table 9. Adverse Reactions Occurring in ≥ 2% of VRAYLAR-treated Patients and > Placebo-treated Adult Patients in 3-Week Bipolar Mania Trials VRAYLAR* System Organ Class /
Preferred Term
Placebo
(N= 442)
(%)3 to 6
mg/day
(N=263)
(%)9 to 12 mg/day⸰
(N=360)
(%)Cardiac Disorders Tachycardiaa 1 2 1 Eye Disorders Vision blurred 1 4 4 Gastrointestinal Disorders Nausea 7 13 11 Constipation 5 6 11 Vomiting 4 10 8 Dry mouth 2 3 2 Dyspepsia 4 7 9 Abdominal painb 5 6 8 Diarrheac 5 5 6 Toothache 2 4 3 General Disorders/Administration Site Conditions Fatigued 2 4 5 Pyrexiae 2 1 4 Investigations Blood creatine phosphokinase increased 2 2 3 Hepatic enzymes increasedf <1 1 3 Weight increased 2 2 3 Metabolism and Nutrition Disorders Decreased appetite 3 3 4 Musculoskeletal and Connective Tissue Disorders Pain in extremity 2 4 2 Back pain 1 1 3 Nervous System Disorders Akathisia 5 20 21 Extrapyramidal Symptomsg 12 26 29 Headacheh 13 14 13 Dizziness 4 7 6 Somnolencei 4 7 8 Psychiatric Disorders Insomniaj 7 9 8 Restlessness 2 7 7 Respiratory, thoracic and mediastinal disorders Oropharyngeal pain 2 1 3 Vascular Disorders Hypertensionk 1 5 4 Note: Figures rounded to the nearest integer
*Data shown by modal daily dose, defined as most frequently administered dose per patient
aTachycardia terms: heart rate increased, sinus tachycardia, tachycardia
bAbdominal pain terms: abdominal discomfort, abdominal pain, abdominal pain upper, abdominal tenderness,
cDiarrhea: diarrhea, frequent bowel movements
dFatigue terms: asthenia, fatigue
ePyrexia terms: body temperature increased, pyrexia
fHepatic enzymes increased terms: alanine aminotransferase increased, aspartate aminotransferase increased, hepatic enzyme increased, transaminases increased
gExtrapyramidal Symptoms terms: bradykinesia, drooling, dyskinesia, dystonia, extrapyramidal disorder, hypokinesia, muscle rigidity, muscle tightness, musculoskeletal stiffness, oromandibular dystonia, parkinsonism, salivary hypersecretion, tremor
hHeadache terms: headache, tension headache
iSomnolence terms: hypersomnia, sedation, somnolence
jInsomnia terms: initial insomnia, insomnia, middle insomnia
kHypertension terms: blood pressure diastolic increased, blood pressure increased, hypertension
⸰ The maximum recommended daily dose is 6 mg. Doses above 6 mg daily do not confer increased effectiveness sufficient to outweigh dose-related adverse reactions.Patients with Bipolar Depression
The following findings are based on three placebo-controlled, two 6-week and one 8-week bipolar depression trials with VRAYLAR doses of 1.5 mg and 3 mg once daily.
Adverse Reactions Associated with Discontinuation of Treatment: There were no adverse reaction leading to discontinuation that occurred at a rate of ≥ 2% in VRAYLAR-treated patients and at least twice the rate of placebo. Overall, 6% of the patients who received VRAYLAR discontinued treatment due to an adverse reaction, compared with 5% of placebo-treated patients in these trials.
Common Adverse Reactions (≥ 5% and at least twice the rate of placebo): nausea, akathisia, restlessness, and extrapyramidal symptoms.
Adverse Reactions with an incidence of ≥ 2% and greater than placebo at 1.5 mg or 3 mg doses are shown in Table 10.
Table 10. Adverse Reactions Occurring in ≥ 2% of VRAYLAR-treated Patients and > Placebo-treated Adult Patients in Two 6-Week and One 8-Week Bipolar Depression Trials VRAYLAR Placebo
(N=468)
(%)1.5 mg/day
(N=470)
(%)3 mg/day
(N=469)
(%)Restlessness 3 2 7 Akathisia 2 6 10 Extrapyramidal symptomsa 2 4 6 Dizziness 2 4 3 Somnolenceb 4 7 6 Nausea 3 7 7 Increased appetite 1 3 3 Weight increase <1 2 2 Fatiguec 2 4 3 Insomniad 7 7 10 aExtrapyramidal symptoms terms: akinesia, drooling, dyskinesia, dystonia, extrapyramidal disorder, hypokinesia, muscle tightness, musculoskeletal stiffness, myoclonus, oculogyric crisis, salivary hypersecretion, tardive dyskinesia, tremor
bSomnolence terms: hypersomnia, sedation, somnolence
cFatigue terms: asthenia, fatigue, malaise
dInsomnia terms: initial insomnia, insomnia, insomnia related to another mental condition, middle insomnia, sleep disorder, terminal insomniaAdjunctive Therapy in Major Depressive Disorder
The following findings are based on two placebo-controlled, fixed-dose 6-week trials with VRAYLAR doses of 1.5 and 3 mg once daily plus an antidepressant and one placebo-controlled, flexible-dose 8-week trial with VRAYLAR doses of (1 to 2 mg) and (2 to 4.5 mg) once daily plus an antidepressant for adjunctive therapy in MDD.
Adverse Reactions Associated with Discontinuation of Treatment: The adverse reaction leading to discontinuation that occurred at a rate of ≥ 2% in VRAYLAR-treated patients and at least twice the rate of placebo was akathisia (2%). Overall, 6% of the patients who received VRAYLAR discontinued treatment due to an adverse reaction, compared with 3% of placebo-treated patients in these trials.
Common Adverse Reactions (≥ 5% and at least twice the rate of placebo): Akathisia, nausea, and insomnia occurred in two 6-week, fixed-dose trials. Akathisia, restlessness, fatigue, constipation, nausea, increased appetite, dizziness, insomnia, and extrapyramidal symptoms occurred in one 8-week flexible-dose trial.
Adverse Reactions with an incidence of ≥ 2% and greater than placebo at 1.5 mg or 3 mg doses are shown in Table 11.
Table 11. Adverse Reactions Occurring in ≥ 2% of VRAYLAR-Treated Patients and > Placebo-Treated Adult Patients in Two Fixed-Dose 6-Week Placebo-Controlled Trials of Adjunctive Treatment of Major Depressive Disorder System Organ Class/
Preferred TermPlacebo + ADT
(N=503)
(%)VRAYLAR 1.5 mg/day + ADT
(N=502)
(%)3 mg/day + ADT
(N=503)
(%)Eye Disorders Vision Blurred <1 <1 2 Gastrointestinal Disorders Nausea 3 7 6 Dry Mouth 2 3 3 Constipation 1 2 2 Vomiting 1 1 2 General Disorders Fatigue 2 3 3 Investigations Weight increased 1 2 2 Nervous System Disorders Akathisiaa 2 7 10 Somnolenceb 4 5 7 Extrapyramidal
Symptomsc4 5 6 Psychiatric Disorders Insomniad 5 9 10 Restlessness 2 4 4 Anxiety 1 2 1 Skin and Subcutaneous Tissue Disorders Hyperhidrosis 1 1 2 Note: Figures rounded to the nearest integer
aAkathisia terms: akathisia, psychomotor hyperactivity, feeling jittery, nervousness, tension
bSomnolence terms: hypersomnia, sedation, lethargy, somnolence
cExtrapyramidal symptoms terms: drooling, dyskinesia, extrapyramidal disorder, hypotonia, muscle contractions involuntary, muscle rigidity, muscle spasms, muscle tightness, muscle twitching, musculoskeletal stiffness, myoclonus, oromandibular dystonia, parkinsonism, resting tremor, restless legs syndrome, stiff leg syndrome, salivary hypersecretion, stiff tongue, tardive dyskinesia, tremor, trismus
dInsomnia terms: initial insomnia, insomnia, middle insomnia, poor sleep quality, sleep disorder, terminal insomniaAdverse Reactions with an incidence of ≥ 2% and greater than placebo at 1 mg to 2 mg per day or 2 mg to 4.5 mg per day doses are shown in Table 12.
Table 12. Adverse Reactions Occurring in ≥ 2% of VRAYLAR-Treated Patients and > Placebo-Treated Adult Patients in a Flexible-dose 8-Week Placebo-Controlled Trial of Adjunctive Treatment of Major Depressive Disorder System Organ Class/ Preferred Term Placebo + ADT
(N=266)
(%)VRAYLAR
1 to 2 mg/day + ADT
(N=273)
(%)VRAYLAR
2 to 4.5 mg/day + ADT
(N=273)
(%)Cardiac disorders Palpitations 1 2 <1 Eye disorders Vision blurred 1 1 4 Gastrointestinal disorders Nausea 5 7 13 Constipation 2 2 5 Dry mouth 3 5 4 Vomiting <1 1 3 General disorders Fatigue 4 7 10 Edema <1 2 1 Infections Nasopharyngitis 2 4 1 Investigations Increased appetite 2 2 5 Weight increased 1 2 3 Musculoskeletal and Connective Tissue disorders Back pain 1 2 3 Myalgia 0 1 2 Nervous System disorders Akathisiaa 3 8 23 Extrapyramidal symptomsb 5 12 18 Somnolencec 6 10 11 Dizziness 2 4 5 Psychiatric disorders Insomniad 8 14 16 Restlessness 3 8 8 Agitation <1 <1 3 Anxiety <1 1 3 aAkathisia terms: akathisia, feeling jittery, nervousness, tension
bExtrapyramidal symptoms terms: cogwheel rigidity, drooling, dyskinesia, extrapyramidal disorder, hypertonia, jaw stiffness, muscle contractions involuntary, muscle disorder, muscle rigidity, muscle spasms, muscle tightness, muscle twitching, musculoskeletal stiffness, nuchal rigidity, parkinsonism, psychomotor retardation, reduced facial expression, resting tremor, restless legs syndrome, sensation of heaviness, salivary hypersecretion, tremor
cSomnolence terms: hypersomnia, sedation, lethargy, somnolence
dInsomnia terms: initial insomnia, insomnia, middle insomnia, terminal insomnia, sleep disorder, poor sleep quality
Dystonia
Symptoms of dystonia, prolonged abnormal contractions of muscle groups, may occur in susceptible individuals during the first few days of treatment. Dystonic symptoms include: spasm of the neck muscles, sometimes progressing to tightness of the throat, swallowing difficulty, difficulty breathing, and/or protrusion of the tongue. Although these symptoms can occur at low doses, they occur more frequently and with greater severity with high potency and higher doses of first-generation antipsychotic drugs. An elevated risk of acute dystonia is observed in males and younger age groups.
Extrapyramidal Symptoms (EPS) and Akathisia
In schizophrenia, bipolar mania, bipolar depression, and adjunctive treatment of major depressive disorder trials, data were objectively collected using the Simpson Angus Scale (SAS) for treatment-emergent EPS (parkinsonism) (SAS total score ≤ 3 at baseline and > 3 post-baseline) and the Barnes Akathisia Rating Scale (BARS) for treatment-emergent akathisia (BARS total score ≤ 2 at baseline and > 2 post-baseline).
In 6-week schizophrenia trials, the incidence of reported adverse reactions related to extrapyramidal symptoms (EPS), excluding akathisia and restlessness was 17% for VRAYLAR-treated patients versus 8% for placebo-treated patients. These reactions led to discontinuation in 0.3% of VRAYLAR-treated patients versus 0.2% of placebo-treated patients. The incidence of akathisia was 11% for VRAYLAR-treated patients versus 4% for placebo-treated patients. These reactions led to discontinuation in 0.5% of VRAYLAR-treated patients versus 0.2% of placebo-treated patients.
In 3-week bipolar mania trials, the incidence of reported adverse reactions related to extrapyramidal symptoms (EPS), excluding akathisia and restlessness, was 28% for VRAYLAR-treated patients versus 12% for placebo-treated patients. These reactions led to a discontinuation in 1% of VRAYLAR-treated patients versus 0.2% of placebo-treated patients. The incidence of akathisia was 20% for VRAYLAR-treated patients versus 5% for placebo-treated patients. These reactions led to discontinuation in 2% of VRAYLAR-treated patients versus 0% of placebo-treated patients.
In the two 6-week and one 8-week bipolar depression trials, the incidence of reported adverse reactions related to EPS, excluding akathisia and restlessness was 4% for VRAYLAR-treated patients versus 2% for placebo-treated patients. These reactions led to discontinuation in 0.4% of VRAYLAR-treated patients versus 0% of placebo-treated patients. The incidence of akathisia was 8% for VRAYLAR-treated patients versus 2% for placebo-treated patients. These reactions led to discontinuation in 1.5% of VRAYLAR-treated patients versus 0% of placebo-treated patients.
In the two 6-week adjunctive treatment of major depressive disorder trials, the incidence of reported adverse reactions related to EPS, excluding akathisia and restlessness, was 6% for VRAYLAR-treated patients versus 4% for placebo-treated patients. These reactions led to discontinuation in 0.3% of VRAYLAR-treated patients versus 0.6% of placebo-treated patients. The combined incidence of akathisia and restlessness was 12% for VRAYLAR-treated patients versus 4% for placebo-treated patients. These reactions led to discontinuation in 2% of VRAYLAR-treated patients versus 0.4% of placebo-treated patients.
In one 8-week adjunctive treatment of major depressive disorder trial, the incidence of reported adverse reactions related to EPS, excluding akathisia and restlessness, was 12% for VRAYLAR-treated patients versus 5% for placebo-treated patients. These reactions led to discontinuation in 1% of VRAYLAR-treated patients versus 0.4% of placebo-treated patients. The incidence of akathisia and restlessness was 22% for VRAYLAR-treated patients versus 6% for placebo-treated patients. These reactions led to discontinuation in 3% of VRAYLAR-treated patients versus 0.0% of placebo-treated patients.
Cataracts
The development of cataracts was observed in nonclinical studies [see Nonclinical Toxicology (13.2)]. Cataracts were reported during the premarketing clinical trials of cariprazine; however, the duration of trials was too short to assess any association to cariprazine usage.
Vital Signs Changes
There were no clinically meaningful differences between VRAYLAR-treated patients and placebo-treated patients in mean change from baseline to endpoint in supine blood pressure parameters except for an increase in supine diastolic blood pressure in the 9 - 12 mg/day VRAYLAR-treated patients with schizophrenia.
Pooled data from 6-week schizophrenia trials are shown in Table 13, and from 3-week bipolar mania trials are shown in Table 14.
Table 13. Mean Change in Blood Pressure at Endpoint in 6-Week Schizophrenia Trials Placebo
(N=574)VRAYLAR* 1.5 - 3 mg/day
(N=512)4.5 - 6 mg/day
(N=570)9 - 12 mg/day⸰
(N=203)Supine Systolic Blood Pressure (mmHg) +0.9 +0.6 +1.3 +2.1 Supine Diastolic Blood Pressure (mmHg) +0.4 +0.2 +1.6 +3.4 * Data shown by modal daily dose, defined as most frequently administered dose per patient
⸰ The maximum recommended daily dose is 6 mg. Doses above 6 mg daily do not confer increased effectiveness sufficient to outweigh dose-related adverse reactions.Table 14. Mean Change in Blood Pressure at Endpoint in 3-Week Bipolar Mania Trials Placebo
(N=439)VRAYLAR* 3 - 6 mg/day
(N=259)9 – 12
mg/day⸰
(N=360)Supine Systolic Blood Pressure (mmHg) -0.5 +0.8 +1.8 Supine Diastolic Blood Pressure (mmHg) +0.9 +1.5 +1.9 * Data shown by modal daily dose, defined as most frequently administered dose per patient
⸰ The maximum recommended daily dose is 6 mg. Doses above 6 mg daily do not confer increased effectiveness sufficient to outweigh dose-related adverse reactions.In the two 6-week and one 8-week bipolar depression trials, there were no clinically meaningful differences between VRAYLAR-treated patients and placebo-treated patients in mean change from baseline to endpoint in supine systolic and diastolic blood pressure. VRAYLAR-treated patients’ supine blood pressure increased by 0.1 to 0.3 mmHg; placebo-treated patients’ supine blood pressure increased by 0.2 mmHg.
In two 6-week and one 8-week adjunctive treatment of major depressive disorder trials, there were no clinically meaningful differences between VRAYLAR-treated patients and placebo-treated patients in mean change from baseline to endpoint in supine systolic and diastolic blood pressure. At the end of the 6-week trials, VRAYLAR-treated patients’ supine systolic blood pressure decreased by 0.1 to 0.7 mmHg; placebo-treated patients’ supine systolic blood pressure decreased by 0.1 mmHg. VRAYLAR-treated patients’ supine diastolic blood pressure increased by 0.1 mmHg and placebo-treated patients’ supine diastolic blood pressure increased by 0.2 mmHg.
Changes in Laboratory Tests
The proportions of patients with transaminase elevations of ≥3 times the upper limits of the normal reference range in 6-week schizophrenia trials ranged between 1% and 2% for VRAYLAR-treated patients, increasing with dose, and was 1% for placebo-treated patients. The proportions of patients with transaminase elevations of ≥3 times the upper limits of the normal reference range in 3-week bipolar mania trials ranged between 2% and 4% for VRAYLAR-treated patients depending on dose group administered and 2% for placebo-treated patients. The proportions of patients with transaminase elevations of ≥3 times the upper limits of the normal reference range in 6-week and 8-week bipolar depression trials ranged between 0% and 0.5% for VRAYLAR-treated patients depending on dose group administered and 0.4% for placebo-treated patients. The proportions of patients with transaminase elevations of ≥3 times the upper limits of the normal reference range in two 6-week adjunctive treatment of major depressive disorder trials ranged between 0% and 1% for VRAYLAR-treated patients depending on dose group administered and 0% for placebo-treated patients.
The proportions of patients with elevations of creatine phosphokinase (CPK) greater than 1000 U/L in 6-week schizophrenia trials ranged between 4% and 6% for VRAYLAR-treated patients, increasing with dose, and was 4% for placebo-treated patients. The proportions of patients with elevations of CPK greater than 1000 U/L in 3-week bipolar mania trials was about 4% in VRAYLAR and placebo-treated patients. The proportions of patients with elevations of CPK greater than 1000 U/L in 6-week and 8-week bipolar depression trials ranged between 0.2% and 1% for VRAYLAR-treated patients versus 0.2% for placebo-treated patients. The proportions of patients with elevations of CPK greater than 1000 U/L in two 6-week adjunctive treatment of major depressive disorder trials ranged between 0.6% and 0.8% for VRAYLAR-treated patients versus 0% for placebo-treated patients.
Other Adverse Reactions Observed During the Pre-marketing Evaluation of VRAYLAR
Adverse reactions listed below were reported by patients treated with VRAYLAR at doses of ≥ 1.5 mg once daily within the premarketing database of 5,763 VRAYLAR-treated patients. The reactions listed are those that could be of clinical importance, as well as reactions that are plausibly drug-related on pharmacologic or other grounds. Reactions that appear elsewhere in the VRAYLAR label are not included.
Reactions are further categorized by organ class and listed in order of decreasing frequency, according to the following definition: those occurring in at least 1/100 patients (frequent) [only those not already listed in the tabulated results from placebo-controlled studies appear in this listing]; those occurring in 1/100 to 1/1000 patients (infrequent); and those occurring in fewer than 1/1,000 patients (rare).
Gastrointestinal Disorders: Infrequent: gastroesophageal reflux disease, gastritis
Hepatobiliary Disorders: Rare: hepatitis
Metabolism and Nutrition Disorders: Frequent: decreased appetite; Rare: hyponatremia
Musculoskeletal and Connective Tissue Disorders: Rare: rhabdomyolysis
Nervous System Disorders: Rare: ischemic stroke
Psychiatric Disorders: Infrequent: suicide ideation; Rare: completed suicide, suicide attempts
Renal and Urinary Disorders: Infrequent: pollakiuria
Skin and Subcutaneous Tissue Disorders: Infrequent: hyperhidrosis6.2 Postmarketing Experience
The following adverse reaction has been identified during post approval use of VRAYLAR. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to estimate their frequency or establish a causal relationship to drug exposure.
Skin and Subcutaneous Tissue Disorders – Stevens-Johnson syndrome
- Increased Mortality in Elderly Patients with Dementia-Related Psychosis [see Boxed Warning and Warnings and Precautions (5.1)]
-
7
DRUG INTERACTIONS
Table 15 displays clinically significant drug interactions with VRAYLAR.
Table 15. Clinically Significant Drug Interactions with VRAYLAR Strong or Moderate CYP3A4 Inhibitors Clinical Impact: Concomitant use of VRAYLAR with a strong or moderate CYP3A4 inhibitor increases the exposures of cariprazine and its major active metabolite, didesmethylcariprazine (DDCAR), compared to use of VRAYLAR alone [see Clinical Pharmacology (12.3)].
Intervention: If VRAYLAR is used with a strong or moderate CYP3A4 inhibitor, reduce VRAYLAR dosage [see Dosage and Administration (2.6)].
CYP3A4 Inducers Clinical Impact: CYP3A4 is responsible for the formation and elimination of the active metabolites of cariprazine. The effect of CYP3A4 inducers on the exposure of VRAYLAR has not been evaluated, and the net effect is unclear [see Clinical Pharmacology (12.3)].
Intervention: Concomitant use of VRAYLAR with a CYP3A4 inducer is not recommended [see Dosage and Administration (2.1, 2.6)].
-
8
USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Exposure Registry
There is a pregnancy exposure registry that monitors pregnancy outcomes in women exposed to VRAYLAR during pregnancy. For more information, contact the National Pregnancy Registry for Atypical Antipsychotics at 1-866-961-2388 or visit http://womensmentalhealth.org/clinical-and-research-programs/pregnancyregistry/.
Risk Summary
Neonates exposed to antipsychotic drugs during the third trimester of pregnancy are at risk for extrapyramidal and/or withdrawal symptoms following delivery (see Clinical Considerations). There are no available data on VRAYLAR use in pregnant women to inform any drug-associated risks for birth defects or miscarriage. The major active metabolite of cariprazine, DDCAR, has been detected in adult patients up to 12 weeks after discontinuation of VRAYLAR [see Clinical Pharmacology (12.3)].
Based on animal data, VRAYLAR may cause fetal harm.Administration of cariprazine to rats during the period of organogenesis caused malformations, lower pup survival, and developmental delays at drug exposures less than the human exposure at the maximum recommended human dose (MRHD) of 6 mg/day. However, cariprazine was not teratogenic in rabbits at doses up to 4.6 times the MRHD of 6 mg/day [see Data].
The estimated background risk of major birth defects and miscarriage for the indicated populations is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively. Advise pregnant women of the potential risk to a fetus.
Clinical Considerations
Fetal/Neonatal Adverse Reactions
Extrapyramidal and/or withdrawal symptoms, including agitation, hypertonia, hypotonia, tremor, somnolence, respiratory distress, and feeding disorder have been reported in neonates whose mothers were exposed to antipsychotic drugs during the third trimester of pregnancy. These symptoms have varied in severity. Some neonates recovered within hours or days without specific treatment; others required prolonged hospitalization. Monitor neonates for extrapyramidal and/or withdrawal symptoms and manage symptoms appropriately.
Data
Animal Data
Administration of cariprazine to pregnant rats during the period of organogenesis at oral doses of 0.5, 2.5, and 7.5 mg/kg/day, which are 0.2 to 3.5 times the maximum recommended human dose (MRHD) of 6 mg/day based on AUC of total cariprazine (i.e. sum of cariprazine, DCAR, and DDCAR), caused fetal developmental toxicity at all doses, which included reduced body weight, decreased male anogenital distance, and skeletal malformations of bent limb bones, scapula, and humerus. These effects occurred in the absence or presence of maternal toxicity. Maternal toxicity, observed as a reduction in body weight and food consumption, occurred at doses 1.2 and 3.5-times the MRHD of 6 mg/day based on AUC of total cariprazine. At these doses, cariprazine caused fetal external malformations (localized fetal thoracic edema), visceral variations (undeveloped/underdeveloped renal papillae and/or distended urethrae), and skeletal developmental variations (bent ribs, unossified sternebrae). Cariprazine had no effect on fetal survival.
Administration of cariprazine to pregnant rats during pregnancy and lactation at oral doses of 0.1, 0.3, and 1 mg/kg/day, which are 0.03 to 0.4 times the MRHD of 6 mg/day based on AUC of total cariprazine, caused a decrease in postnatal survival, birth weight, and post-weaning body weight of first generation pups at the dose that is 0.4 times the MRHD of 6 mg/day based on AUC of total cariprazine in absence of maternal toxicity. First generation pups also had pale, cold bodies and developmental delays (renal papillae not developed or underdeveloped and decreased auditory startle response in males). Reproductive performance of the first generation pups was unaffected; however, the second generation pups had clinical signs and lower body weight similar to those of the first generation pups.
Administration of cariprazine to pregnant rabbits during the period of organogenesis at oral doses of 0.1, 1, and 5 mg/kg/day, which are 0.02 to 4.6 times the MRHD of 6 mg/day based on AUC of total cariprazine, was not teratogenic. Maternal body weight and food consumption were decreased at 4.6 times the MRHD of 6 mg/day based on AUC of total cariprazine; however, no adverse effects were observed on pregnancy parameters or reproductive organs.
8.2 Lactation
Risk Summary
Lactation studies have not been conducted to assess the presence of cariprazine in human milk, the effects on the breastfed infant, or the effects on milk production. Cariprazine is present in rat milk. The development and health benefits of breastfeeding should be considered along with the mother’s clinical need for VRAYLAR and any potential adverse effects on the breastfed infant from VRAYLAR or from the underlying maternal condition.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established. Pediatric studies of VRAYLAR have not been conducted. Antidepressants increased the risk of suicidal thoughts and behaviors in pediatric patients [see Boxed Warning, Warnings and Precautions (5.2)].
8.5 Geriatric Use
Clinical trials of VRAYLAR did not include sufficient numbers of patients aged 65 and older to determine whether or not they respond differently from younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
Antipsychotic drugs increase the risk of death in elderly patients with dementia-related psychosis. VRAYLAR is not approved for the treatment of patients with dementia-related psychosis [see Boxed Warning and Warnings and Precautions (5.1, 5.3)].
8.6 Hepatic Impairment
No dosage adjustment for VRAYLAR is required in patients with mild to moderate hepatic impairment (Child-Pugh score between 5 and 9) [see Clinical Pharmacology (12.3)]. Usage of VRAYLAR is not recommended in patients with severe hepatic impairment (Child-Pugh score between 10 and 15). VRAYLAR has not been evaluated in this patient population.
8.7 Renal Impairment
No dosage adjustment for VRAYLAR is required in patients with mild to moderate (CrCL ≥ 30 mL/minute) renal impairment [see Clinical Pharmacology (12.3)].
Usage of VRAYLAR is not recommended in patients with severe renal impairment (CrCL < 30 mL/minute). VRAYLAR has not been evaluated in this patient population.
8.8 Smoking
No dosage adjustment for VRAYLAR is needed for patients who smoke. VRAYLAR is not a substrate for CYP1A2; smoking is not expected to have an effect on the pharmacokinetics of VRAYLAR.
8.9 Other Specific Populations
No dosage adjustment is required based on patient’s age, sex, or race. These factors do not affect the pharmacokinetics of VRAYLAR [see Clinical Pharmacology (12.3)].
- 9 DRUG ABUSE AND DEPENDENCE
-
10
OVERDOSAGE
10.1 Human Experience
In pre-marketing clinical trials involving VRAYLAR in approximately 5000 patients or healthy subjects, accidental acute overdosage (48 mg/day) was reported in one patient. This patient experienced orthostasis and sedation. The patient fully recovered the same day.
10.2 Management of Overdosage
No specific antidotes for VRAYLAR are known. In managing overdose, provide supportive care, including close medical supervision and monitoring, and consider the possibility of multiple drug involvement. In case of an overdose, consult a Certified Poison Control Center (1-800-222-1222) for up-to-date guidance and advice.
-
11
DESCRIPTION
The active ingredient of VRAYLAR is cariprazine, an atypical antipsychotic, in hydrochloride salt form. The chemical name is trans-N-{4-[2-[4-(2,3-dichlorophenyl)piperazine-1-yl]ethyl]cyclohexyl}-N’,N’-dimethylurea hydrochloride; its empirical formula is C21H32Cl2N4O•HCl and its molecular weight is 463.9 g/mol. The chemical structure is:
![The chemical structure for VRAYLAR is cariprazine HCl, an atypical antipsychotic. The chemical name is trans-N-{4-[2-[4-(2,3 dichlorophenyl)piperazine-1-yl]ethyl]cyclohexyl}-N’,N’-dimethylurea hydrochloride; its empirical formula is C21H33Cl3N4O and its molecular weight is 463.9 g/mol.](/dailymed/image.cfm?name=vraylar-01.jpg&setid=4b5f7c65-aa2d-452a-b3db-bc85c06ff12f)
VRAYLAR capsules are intended for oral administration only. Each hard gelatin capsule contains a white to off-white powder of cariprazine HCl, which is equivalent to 1.5, 3, 4.5, or 6 mg of cariprazine base. In addition, capsules include the following inactive ingredients: gelatin, magnesium stearate, pregelatinized starch, shellac, and titanium dioxide. Colorants include black iron oxide (1.5, 3, and 6 mg), FD&C Blue 1 (3, 4.5, and 6 mg), FD&C Red 3 (6 mg), FD&C Red 40 (3 and 4.5 mg), or yellow iron oxide (3 and 4.5 mg).
-
12
CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
The mechanism of action of cariprazine is unknown. However, the efficacy of cariprazine could be mediated through a combination of partial agonist activity at central dopamine D2 and serotonin 5-HT1A receptors and antagonist activity at serotonin 5-HT2A receptors. Cariprazine forms two major metabolites, desmethylcariprazine (DCAR) and didesmethylcariprazine (DDCAR), that have in vitro receptor binding profiles similar to the parent drug.
12.2 Pharmacodynamics
Cariprazine acts as a partial agonist at the dopamine D3 and D2 receptors with high binding affinity (Ki values 0.085 nM, and 0.49 nM (D2L) and 0.69 nM (D2S), respectively) and at the serotonin 5-HT1A receptors (Ki value 2.6 nM). Cariprazine acts as an antagonist at 5-HT2B and 5-HT2A receptors with high and moderate binding affinity (Ki values 0.58 nM and 18.8 nM respectively) as well as it binds to the histamine H1 receptors (Ki value 23.2 nM). Cariprazine shows lower binding affinity to the serotonin 5-HT2C and α1A- adrenergic receptors (Ki values 134 nM and 155 nM, respectively) and has no appreciable affinity for cholinergic muscarinic receptors (IC50>1000 nM).
Effect on QTc Interval
At a dose three-times the maximum recommended dose, cariprazine does not prolong the QTc interval to clinically relevant extent.
12.3 Pharmacokinetics
VRAYLAR activity is thought to be mediated by cariprazine and its two major active metabolites, desmethylcariprazine (DCAR) and didesmethylcariprazine (DDCAR), which are pharmacologically equipotent to cariprazine.
After multiple dose administration of VRAYLAR, mean cariprazine and DCAR concentrations reached steady state at around Week 1 to Week 2 and mean DDCAR concentrations appeared to be approaching steady state at around Week 4 to Week 8 in a 12-week study (Figure 1). The half-lives based on time to reach steady state, estimated from the mean concentration-time curves, are 2 to 4 days for cariprazine, about 1 to 2 days for DCAR, and approximately 1 to 3 weeks for DDCAR. The time to reach steady state for the major active metabolite DDCAR was variable across patients, with some patients not achieving steady state at the end of the 12 week treatment [see Dosage and Administration (2.1), Warnings and Precautions (5.6)]. Mean concentrations of DCAR and DDCAR are approximately 30% and 400%, respectively, of cariprazine concentrations by the end of 12-week treatment.
After discontinuation of VRAYLAR, cariprazine, DCAR, and DDCAR plasma concentrations declined in a multi-exponential manner. Mean plasma concentrations of DDCAR decreased by about 50% 1 week after the last dose, and mean cariprazine and DCAR concentration dropped by about 50% in about 1 day. There was an approximately 90% decline in plasma exposure within 1 week for cariprazine and DCAR, and at about 4 weeks for DDCAR. Following a single dose of 1 mg of cariprazine administration, DDCAR remained detectable 8 weeks post-dose.
After multiple dosing of VRAYLAR, plasma exposure of cariprazine, DCAR, and DDCAR increases approximately proportionally over the therapeutic dose range.
Figure 1. Plasma Concentration (Mean ± SE)-Time Profile During and Following
12-weeks of Treatment with Cariprazine 6 mg/daya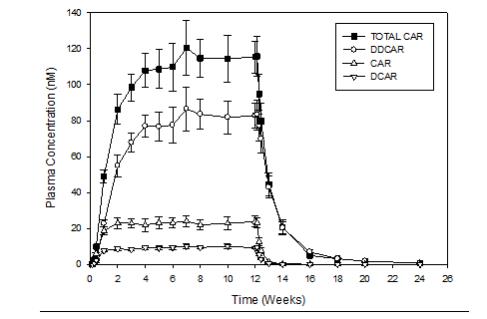
a Trough concentrations shown during treatment with cariprazine 6 mg/day.
SE: standard error; TOTAL CAR: sum concentration of cariprazine, DCAR and DDCAR; CAR: cariprazine
Absorption
After single dose administration of VRAYLAR, the peak plasma cariprazine concentration occurred in approximately 3-6 hours.
Administration of a single dose of 1.5 mg VRAYLAR capsule with a high-fat meal did not significantly affect the Cmax and AUC of cariprazine or DCAR.
Distribution
Cariprazine and its major active metabolites are highly bound (91 to 97%) to plasma proteins.
Elimination
Metabolism
Cariprazine is extensively metabolized by CYP3A4 and, to a lesser extent, by CYP2D6 to DCAR and DDCAR. DCAR is further metabolized into DDCAR by CYP3A4 and CYP2D6. DDCAR is then metabolized by CYP3A4 to a hydroxylated metabolite.
Excretion
Following administration of 12.5 mg/day cariprazine to patients with schizophrenia for 27 days, about 21% of the daily dose was found in urine, with approximately 1.2% of the daily dose excreted in urine as unchanged cariprazine.
Studies in Specific Populations
Hepatic Impairment
Compared to healthy subjects, exposure (Cmax and AUC) in patients with either mild or moderate hepatic impairment (Child-Pugh score between 5 and 9) was approximately 25% higher for cariprazine and 20% to 30% lower for the major metabolites (DCAR and DDCAR) following daily doses of 0.5 mg cariprazine for 14 days [see Use in Specific Populations (8.6)].
Renal Impairment
Cariprazine and its major active metabolites are minimally excreted in urine. Pharmacokinetic analyses indicated no significant relationship between plasma clearance and creatinine clearance [see Use in Specific Populations (8.7)].
CYP2D6 Poor Metabolizers
CYP2D6 poor metabolizer status does not have clinically relevant effect on pharmacokinetics of cariprazine, DCAR, or DDCAR.
Age, Sex, Race
Age, sex, or race does not have clinically relevant effect on pharmacokinetics of cariprazine, DCAR, or DDCAR.
Drug Interaction Studies
In vitro studies
Cariprazine and its major active metabolites did not induce CYP1A2 and CYP3A4 enzymes and were weak inhibitors of CYP1A2, CYP2C9, CYP2D6, and CYP3A4 in vitro. Cariprazine was also a weak inhibitor of CYP2C19, CYP2A6, and CYP2E1 in vitro.
Cariprazine and its major active metabolites are not substrates of P-glycoprotein (P-gp), the organic anion transporting polypeptides 1B1 and 1B3 (OATP1B1 and OATP1B3), or the breast cancer resistance protein (BCRP).
Cariprazine and its major active metabolites were poor or non-inhibitors of transporters OATP1B1, OATP1B3, BCRP, organic cation transporter 2 (OCT2), and organic anion transporters 1 and 3 (OAT1 and OAT3) in vitro. The major active metabolites were also poor or non-inhibitors of transporter P-gp although cariprazine was probably a P-gp inhibitor based on the theoretical GI concentrations at high doses in vitro.
Based on in vitro studies, VRAYLAR is unlikely to cause clinically significant pharmacokinetic drug interactions with substrates of CYP1A2, CYP2A6, CYP2C9, CYP2C19, CYP2D6, CYP2E, and CYP3A4, or OATP1B1, OATP1B3, BCRP, OCT2, OAT1 and OAT3.
In vivo studies and Model-based Approaches
CYP3A4 inhibitors
In the clinical drug-drug interaction studies, co-administration of ketoconazole (400 mg/day for four days), a strong CYP3A4 inhibitor, with VRAYLAR (0.5 mg/day) increased total cariprazine (the sum of cariprazine, DCAR and DDCAR) Cmax and AUC0-24h by approximately 100%. Co-administration of erythromycin (500 mg twice daily for 21 days), a moderate CYP3A4 inhibitor, with VRAYLAR (1.5 mg/day) increased total cariprazine Cmax and AUC0-24h by approximately 50%.
Physiologically based pharmacokinetic model-based analyses suggest that co-administration of ketoconazole (400 mg/day) with VRAYLAR (0.5 mg/day) at steady state is predicted to result in up to about 5.5-fold and 6-fold increase in Cmax and AUC0-24h, respectively, of total cariprazine. Co-administration of fluconazole (200 mg/day), a moderate CYP3A inhibitor, with VRAYLAR (0.5 mg/day) at steady state is predicted to result in up to about 3-fold increase in Cmax and AUC0-24h of total cariprazine.
CYP3A4 inducers
CYP3A4 is responsible for the formation and elimination of the active metabolites of cariprazine. The effect of CYP3A4 inducers on the plasma exposure of cariprazine and its major active metabolites has not been evaluated, and the net effect is unclear.
CYP2D6 inhibitors
CYP2D6 inhibitors are not expected to influence pharmacokinetics of cariprazine, DCAR, or DDCAR based on the observations in CYP2D6 poor metabolizers.
Proton pump inhibitors
Co-administration of pantoprazole (40 mg/day), a proton pump inhibitor, with VRAYLAR (6 mg/day) in patients with schizophrenia for 15 days did not affect cariprazine exposure at steady-state, based on Cmax and AUC0-24. Similarly, no significant change in exposure to DCAR and DDCAR was observed.
-
13
NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
There was no increase in the incidence of tumors following daily oral administration of cariprazine to rats for 2 years and to Tg.rasH2 mice for 6 months at doses which are up to 4 and 19 times respectively, the MRHD of 6 mg/day based on AUC of total cariprazine, (i.e. sum of AUC values of cariprazine, DCAR and DDCAR).
Rats were administered cariprazine at oral doses of 0.25, 0.75, and 2.5 (males)/1, 2.5, and 7.5 mg/kg/day (females) which are 0.2 to 1.8 (males)/ 0.8 to 4.1 (females) times the MRHD of 6 mg/day based on AUC of total cariprazine.
Tg.rasH2 mice were administered cariprazine at oral doses of 1, 5, and 15 (males)/5, 15, and 50 mg/kg/day (females) which are 0.2 to 7.9 (males)/2.6 to 19 (females) times the MRHD of 6 mg/day based on AUC of total cariprazine.
Mutagenesis
Cariprazine was not mutagenic in the in vitro bacterial reverse mutation assay, nor clastogenic in the in vitro human lymphocyte chromosomal aberration assay or in the in vivo mouse bone marrow micronucleus assay. However, cariprazine increased the mutation frequency in the in vitro mouse lymphoma assay under conditions of metabolic activation. The major human metabolite DDCAR was not mutagenic in the in vitro bacterial reverse mutation assay, however, it was clastogenic and induced structural chromosomal aberration in the in vitro human lymphocyte chromosomal aberration assay.
Impairment of Fertility
Cariprazine was administered orally to male and female rats before mating, through mating, and up to day 7 of gestation at doses of 1, 3, and 10 mg/kg/day which are 1.6 to 16 times the MRHD of 6 mg/day based on mg/m2. In female rats, lower fertility and conception indices were observed at all dose levels which are equal to or higher than 1.6 times the MRHD of 6 mg/day based on mg/m2. No effects on male fertility were noted at any dose up to 4.3 times the MRHD of 6 mg/day based on AUC of total cariprazine.
13.2 Animal Toxicology and/or Pharmacology
Cariprazine caused bilateral cataract and cystic degeneration of the retina in the dog following oral daily administration for 13 weeks and/or 1 year and retinal degeneration/atrophy in the rat following oral daily administration for 2 years. Cataract in the dog was observed at 4 mg/kg/day which is 7.1 (male) and 7.7 (female) times the MRHD of 6 mg/day based on AUC of total cariprazine. The NOEL for cataract and retinal toxicity in the dog is 2 mg/kg/day which is 5 (males) to 3.6 (females) times the MRHD of 6 mg/day based on AUC of total cariprazine. Increased incidence and severity of retinal degeneration/atrophy in the rat occurred at all doses tested, including the low dose of 0.75 mg/kg/day, at total cariprazine plasma levels less than clinical exposure (AUC) at the MRHD of 6 mg/day. Cataract was not observed in other repeat dose studies in pigmented mice or albino rats.
Phospholipidosis was observed in the lungs of rats, dogs, and mice (with or without inflammation) and in the adrenal gland cortex of dogs at clinically relevant exposures (AUC) of total cariprazine. Phospholipidosis was not reversible at the end of the 1-2 month drug-free periods. Inflammation was observed in the lungs of dogs dosed daily for 1 year with a NOEL of 1 mg/kg/day which is 2.7 (males) and 1.7 (females) times the MRHD of 6 mg/day based on AUC of total cariprazine. No inflammation was observed at the end of 2-month drug free period following administration of 2 mg/kg/day which is 5 (males) and 3.6 (females) times the MRHD of 6 mg/day based on AUC of total cariprazine; however, inflammation was still present at higher doses.
Hypertrophy of the adrenal gland cortex was observed at clinically relevant total cariprazine plasma concentrations in rats (females only) and mice following daily oral administration of cariprazine for 2 years and 6 months, respectively. Reversible hypertrophy/hyperplasia and vacuolation/vesiculation of the adrenal gland cortex were observed following daily oral administration of cariprazine to dogs for 1 year. The NOEL was 2 mg/kg/day which is 5 (males) and 3.6 (females) times the MRHD of 6 mg/day based on AUC of total cariprazine. The relevance of these findings to human risk is unknown.
-
14
CLINICAL STUDIES
14.1 Schizophrenia
The efficacy of VRAYLAR for the treatment of schizophrenia was established in three, 6-week, randomized, double-blind, placebo-controlled trials in patients (mean age of 37 years, aged 18 to 60 years; 31% were female; and 45% were Caucasian) who met Diagnostic and Statistical Manual of Mental Disorders 4th edition, Text Revision (DSM-IV-TR) criteria for schizophrenia. An active control arm (risperidone or aripiprazole) was included in two trials to assess assay sensitivity. In all three trials, VRAYLAR was superior to placebo.
Positive and Negative Syndrome Scale (PANSS) and Clinical Global Impressions-Severity (CGI-S) rating scales were used as the primary and secondary efficacy measures, respectively, for assessing psychiatric signs and symptoms in each trial:
- PANSS is a 30-item scale that measures positive symptoms of schizophrenia (7 items), negative symptoms of schizophrenia (7 items), and general psychopathology (16 items), each rated on a scale of 1 (absent) to 7 (extreme). The PANSS total score may range from 30 to 210 with the higher score reflecting greater severity.
- The CGI-S is a validated clinician-related scale that measures the patient’s current illness state and overall clinical state on a 1 (normal, not at all ill) to 7-point (extremely ill) scale.
In each study, the primary endpoint was change from baseline in PANSS total score at the end of week 6. The change from baseline for VRAYLAR and active control groups was compared to placebo. The results of the trials are shown in Table 16. The time course of efficacy results of Study 2 is shown in Figure 2.
Study 1: In a 6-week, placebo-controlled trial (N = 711) involving three fixed doses of VRAYLAR (1.5, 3, or 4.5 mg/day) and an active control (risperidone), all VRAYLAR doses and the active control were superior to placebo on the PANSS total score and the CGI-S.
Study 2: In a 6-week, placebo-controlled trial (N = 604) involving two fixed doses of VRAYLAR (3 or 6 mg/day) and an active control (aripiprazole), both VRAYLAR doses and the active control were superior to placebo on the PANSS total score and the CGI-S.
Study 3: In a 6-week, placebo-controlled trial (N = 439) involving two flexible-dose range groups of VRAYLAR (3 to 6 mg/day or 6 to 9 mg/day), both VRAYLAR groups were superior to placebo on the PANSS total score and the CGI-S.
The efficacy of VRAYLAR was demonstrated at doses ranging from 1.5 to 9 mg/day compared to placebo. There was, however, a dose-related increase in certain adverse reactions, particularly above 6 mg. Therefore, the maximum recommended dose is 6 mg/day.
Examination of population subgroups based on age (there were few patients over 55), sex, and race did not suggest any clear evidence of differential responsiveness.
Table 16. Primary Analysis Results from Schizophrenia Trials Study
NumberTreatment Group
(# ITT patients)Primary Efficacy Endpoint: PANSS Total Mean
Baseline
Score (SD)LS Mean
Change from
Baseline (SE)Placebo-subtracted
Differencea (95% CI)Study 1 VRAYLAR (1.5 mg/day)* (n=140) 97.1 (9.1) -19.4 (1.6) -7.6 (-11.8, -3.3) VRAYLAR (3 mg/day)*
(n=140)97.2 (8.7) -20.7 (1.6) -8.8 (-13.1, -4.6) VRAYLAR (4.5 mg/day)*
(n=145)96.7 (9.0) -22.3 (1.6) -10.4 (-14.6, -6.2) Placebo
(n=148)97.3 (9.2) -11.8 (1.5) -- Study 2 VRAYLAR (3 mg/day)* (n=151) 96.1 (8.7) -20.2 (1.5) -6.0 (-10.1, -1.9) VRAYLAR (6 mg/day)*
(n=154)95.7 (9.4) -23.0 (1.5) -8.8 (-12.9, -4.7) Placebo
(n=149)96.5 (9.1) -14.3 (1.5) -- Study 3 VRAYLAR (3-6 mg/day)*
(n=147)96.3 (9.3) -22.8 (1.6) -6.8 (-11.3, -2.4) VRAYLAR (6-9 mg/day)*b
(n=147)96.3 (9.0) -25.9 (1.7) -9.9 (-14.5, -5.3) Placebo
(n=145)96.6 (9.3) -16.0 (1.6) -- ITT: intent-to-treat; SD: standard deviation; SE: standard error; LS Mean: least-squares mean; CI: unadjusted confidence interval
aDifference (drug minus placebo) in least-squares mean change from baseline
*Doses that are statistically significantly superior to placebo
bThe maximum recommended daily dose is 6 mg. Doses above 6 mg daily do not confer increased effectiveness sufficient to outweigh dose-related adverse reactions.Figure 2. Change from Baseline in PANSS total score by weekly visits (Study 2)
The safety and efficacy of VRAYLAR as maintenance treatment in adults with schizophrenia were demonstrated in a randomized withdrawal trial that included 200 patients meeting DSM-IV criteria for schizophrenia who were clinically stable following 20 weeks of open-label cariprazine at doses of 3 to 9 mg/day. Patients were randomized to receive either placebo or cariprazine at the same dose for up to 72 weeks for observation of relapse. The primary endpoint was time to relapse. Relapse during the double-blind phase (DBP) was defined as meeting any one of the following criteria: hospitalization due to worsening of schizophrenia, increase in the PANSS total score by ≥ 30%, increase in CGI-S score by ≥ 2 points, deliberate self-injury, aggressive or violent behavior, clinically significant suicidal or homicidal ideation, or score >4 on one or more of the following PANSS items: delusions (P1), conceptual disorganization (P2), hallucination (P3), suspiciousness or persecution (P6), hostility (P7), uncooperativeness (G8), or poor impulse control (G14).
The efficacy of VRAYLAR was demonstrated at doses ranging from 3 to 9 mg/day compared to placebo. There was, however, a dose-related increase in certain adverse reactions, particularly above 6 mg. Therefore, the maximum recommended dose is 6 mg/day.
The Kaplan-Meier curves of the time to relapse during the double-blind, placebo-controlled, randomized withdrawal phase of the long-term trial are shown in Figure 3. Time to relapse was statistically significantly longer in the VRAYLAR-treated group compared to the placebo group.
Figure 3. Kaplan-Meier Curves of Cumulative Rate of Relapse During the Double-Blind Treatment Period
DB = double-blind
*The maximum recommended daily dose is 6 mg. Doses above 6 mg daily do not confer increased effectiveness sufficient to outweigh dose-related adverse reactions.14.2 Manic or Mixed Episodes Associated with Bipolar I Disorder
The efficacy of VRAYLAR in the acute treatment of bipolar mania was established in three, 3-week placebo-controlled trials in patients (mean age of 39 years, range 18 to 65 years; 40% were female; and 48% were Caucasian) who met DSM-IV-TR criteria for bipolar 1 disorder with manic or mixed episodes with or without psychotic features. In all three trials, VRAYLAR was superior to placebo.
Young Mania Rating Scale (YMRS) and Clinical Global Impressions-Severity scale (CGI-S) were used as the primary and secondary efficacy measures, respectively, for assessing psychiatric signs and symptoms in each trial:
- The YMRS is an 11-item clinician-rated scale traditionally used to assess the degree of manic symptomatology. YMRS total score may range from 0 to 60 with a higher score reflecting greater severity.
- The CGI-S is validated clinician-related scale that measures the patient’s current illness state and overall clinical state on a 1 (normal, not at all ill) to 7-point (extremely ill) scale.
In each study, the primary endpoint was decrease from baseline in YMRS total score at the end of week 3. The change from baseline for each VRAYLAR dose group was compared to placebo. The results of the trials are shown in Table 17. The time course of efficacy results is shown in Figure 4.
Study 4: In a 3-week, placebo-controlled trial (N = 492) involving two flexible-dose range groups of VRAYLAR (3 to 6 mg/day or 6 to 12 mg/day), both VRAYLAR dose groups were superior to placebo on the YMRS total score and the CGI-S. The 6 to 12 mg/day dose group showed no additional advantage.
Study 5: In a 3-week, placebo-controlled trial (N = 235) involving a flexible-dose range of VRAYLAR (3 to 12 mg/day), VRAYLAR was superior to placebo on the YMRS total score and the CGI-S.
Study 6: In a 3-week, placebo-controlled trial (N = 310) involving a flexible-dose range of VRAYLAR (3 to 12 mg/day), VRAYLAR was superior to placebo on the YMRS total score and the CGI-S.
The efficacy of VRAYLAR was established at doses ranging from 3 to 12 mg/day. Doses above 6 mg did not appear to have additional benefit over lower doses (Table 17), and there was a dose-related increase in certain adverse reactions. Therefore, the maximum recommended dose is 6 mg/day.
Examination of population subgroups based on age (there were few patients over 55), sex, and race did not suggest any clear evidence of differential responsiveness.
Table 17. Primary Analysis Results from Manic or Mixed Episodes Associated with Bipolar I Disorder Trials Study
NumberTreatment Group
(# ITT patients)Primary Efficacy Endpoint: YMRS Total Mean
Baseline
Score (SD)LS Mean
Change from
Baseline (SE)Placebo-subtracted
Differencea (95% CI)Study 4 VRAYLAR (3-6 mg/day)*
(n=165)33.2 (5.6) -18.6 (0.8) -6.1 (-8.4, -3.8) VRAYLAR (6-12 mg/day)*b
(n=167)32.9 (4.7) -18.5 (0.8) -5.9 (-8.2, -3.6) Placebo
(n=160)32.6 (5.8) -12.5 (0.8) -- Study 5 VRAYLAR (3-12 mg/day)*b
(n=118)30.6 (5.0) -15.0 (1.1) -6.1 (-8.9, -3.3) Placebo
(n=117)30.2 (5.2) -8.9 (1.1) -- Study 6 VRAYLAR (3-12 mg/day)*b
(n=158)32.3 (5.8) -19.6 (0.9) -4.3 (-6.7, -1.9) Placebo
(n=152)32.1 (5.6) -15.3 (0.9) -- ITT: intent-to-treat; SD: standard deviation; SE: standard error; LS Mean: least-squares mean; CI: unadjusted confidence interval
aDifference (drug minus placebo) in least-squares mean change from baseline
*Doses that are statistically significantly superior to placebo
bThe maximum recommended daily dose is 6 mg. Doses above 6 mg daily do not confer increased effectiveness sufficient to outweigh dose-related adverse reactions.
Figure 4. Change from Baseline in YMRS total score by study visit (Study 4)
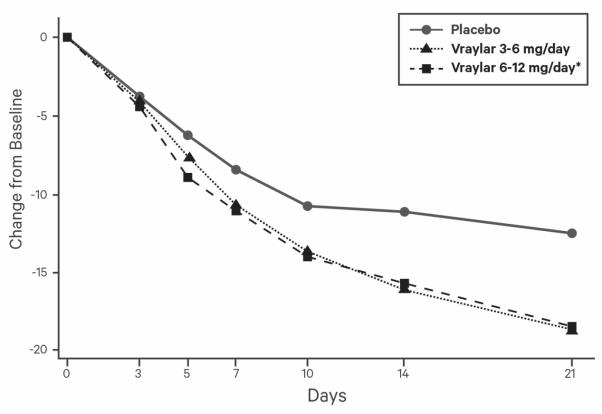
* The maximum recommended daily dose is 6 mg. Doses above 6 mg daily do not confer increased effectiveness sufficient to outweigh dose-related adverse reactions.
14.3 Depressive Episodes Associated with Bipolar I Disorder (Bipolar Depression)
The efficacy of VRAYLAR in the treatment of depressive episodes associated with bipolar I disorder (bipolar depression) was established in one 8-week and two 6-week placebo-controlled trials in patients (mean age of 43 years, range 18 to 65 years; 61% were female; and 75% were Caucasian) who met DSM-IV-TR or DSM-5 criteria for depressive episodes associated with bipolar I disorder.
In each study, the primary endpoint was change from baseline in Montgomery-Asberg Depression Rating Scale (MADRS) total score at the end of Week 6. The MADRS is a 10-item clinician-rated scale with total scores ranging from 0 (no depressive features) to 60 (maximum score). The MADRS total score change from baseline for VRAYLAR compared to placebo is shown in Table 18. The time course of efficacy results of Study 8 is shown in Figure 5. In each study, the VRAYLAR 1.5 mg dose demonstrated statistical significance over placebo. The secondary endpoint was change from baseline to Week 6 in CGI-S. The CGI-S is validated clinician-related scale that measures the patient’s current illness state and overall clinical state on a 1 (normal, not at all ill) to 7-point (extremely ill) scale.
Study 7: In an 8-week, placebo-controlled trial (N = 571) involving three-fixed doses of VRAYLAR (0.75 mg/day, 1.5 mg/day, and 3 mg/day), VRAYLAR 1.5 mg was superior to placebo at end of Week 6 on the MADRS total score and the CGI-S.
Study 8: In a 6-week, placebo-controlled trial (N = 474) involving two-fixed doses of VRAYLAR (1.5 mg/day and 3 mg/day), VRAYLAR 1.5 mg and 3 mg were superior to placebo at end of Week 6 on the MADRS total score.
Study 9: In a 6-week, placebo-controlled trial (N = 478) involving two-fixed doses of VRAYLAR (1.5 mg/day and 3 mg/day), VRAYLAR 1.5 mg was superior to placebo at end of Week 6 on the MADRS total score and the CGI-S.
Examination of population subgroups based on age (there were few patients over 55), sex, and race did not suggest any clear evidence of differential responsiveness.
Table 18. Primary Analysis Results from Bipolar Depression Trials Study Number Treatment Group
(# ITT patients)Primary Efficacy Endpoint: MADRS Total Mean
Baseline
Score (SD)LS Mean
Change
from
Baseline
(SE)Placebo-subtracted
Differencea (95% CI)Study 7 VRAYLAR (1.5 mg/day)* (n=145)
VRAYLAR (3 mg/day)
(n=145)
Placebo
(n=141)
30.3 (4.4)
30.6 (4.7)
30.4 (4.6)
-15.1 (0.8)
-13.7 (0.9)
-11.1 (0.9)
-4.0 (-6.3, -1.6)
-2.5 (-4.9, -0.1)Study 8 VRAYLAR (1.5 mg/day)* (n=154) 30.7 (4.3) -15.1 (0.8) -2.5 (-4.6, -0.4) VRAYLAR (3 mg/day)*
(n=164)31.0 (4.9) -15.6 (0.8) -3.0 (-5.1, -0.9) Placebo
(n=156)30.2 (4.4) -12.6 (0.8) Study 9 VRAYLAR (1.5 mg/day)*
(n=162)31.5 (4.3) -14.8 (0.8) -2.5 (-4.6, -0.4) VRAYLAR (3 mg/day)
(n=153)31.5 (4.8) -14.1 (0.8) -1.8 (-3.9, 0.4) Placebo
(n=163)31.4 (4.5) -12.4 (0.8) ITT: intent-to-treat; SD: standard deviation; SE: standard error; LS Mean: least-squares mean; CI: confidence interval
aDifference (drug minus placebo) in least-squares mean change from baseline
*Doses that are statistically significantly superior to placebo
Figure 5. LS Mean* Change from Baseline in MADRS Total Score by Visits (Study 8)
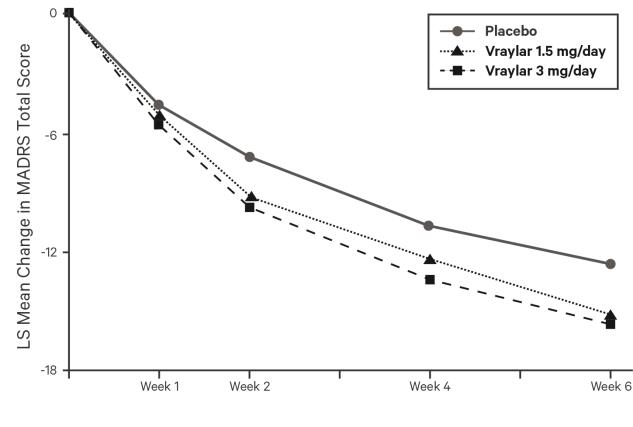
*LS Mean: least-squares mean
14.4 Adjunctive Treatment of Major Depressive Disorder
The efficacy of VRAYLAR as adjunctive therapy to antidepressants for the treatment of major depressive disorder (MDD) was evaluated in 2 trials in adult patients (mean age of 45 years, range 18 to 65 years; 72% were female; and 85% were Caucasian) who met DSM-IV-TR or DSM-5 criteria for MDD, with or without symptoms of anxiety, who had an inadequate response to 1 to 3 courses of prior antidepressant (ADT) therapy. Inadequate response during antidepressant treatment was defined as less than 50% improvement to antidepressant treatment of adequate dose and adequate duration.
In each study, the primary endpoint was change from baseline to Week 6 (Study 10) or Week 8 (Study 11) in the Montgomery-Asberg Depression Rating Scale (MADRS) total score, a 10-item clinician-rated scale used to assess the degree of depressive symptomatology, with 0 representing no symptoms and 60 representing worst symptoms.
Study 10: In a 6-week, placebo-controlled trial (N = 751) involving two fixed doses of VRAYLAR (1.5 mg per day or 3 mg per day) + ADT, VRAYLAR 1.5 mg + ADT was superior to placebo + ADT at end of Week 6 on the MADRS total score. The treatment effect in the VRAYLAR 3 mg per day + ADT group (vs. placebo + ADT) was not statistically significant.
Study 11: An 8-week, placebo-controlled trial (N = 808) involved flexible doses of VRAYLAR 1 to 2 mg per day + ADT or 2 to 4.5 mg per day + ADT. VRAYLAR 2 to 4.5 mg (mean dose was 2.6 mg) + ADT was superior to placebo + ADT at end of Week 8 on the MADRS total score. The treatment effect in the VRAYLAR 1 to 2 mg per day + ADT group (vs. placebo + ADT) was not statistically significant.
Results from the primary efficacy parameters for both trials (Studies 10 and 11) are shown below in Table 19. Figure 6 below shows the time course of response based on the primary efficacy measure (MADRS total score) in Study 10.
Table 19: Primary Analysis Results from Adjunctive Treatment of Major Depressive Disorder Trials Study Number Treatment Group (# ITT patients) Primary Efficacy Endpoint: MADRS Total Score Mean Baseline Score (SD) LS Mean Change from Baseline (SE) Placebo-subtracted Differencea (95% CI) Study 10 VRAYLAR (1.5 mg/day) + ADT* (n=250)
VRAYLAR (3 mg/day) + ADT
(n=252)
Placebo + ADT
(n=249)
32.8 (5.0)
32.7 (4.9)
31.9 (5.7)
-14.1 (0.7)
-13.1 (0.7)
-11.5 (0.7)
-2.5(-4.2, -0.9)
-1.5 (-3.2, 0.1)
Study 11 VRAYLAR (1 to 2 mg/day) + ADT (n=273) 29.0 (4.3)
-13.4 (0.5) -0.9 (-2.4, 0.6)
VRAYLAR (2 to 4.5 mg/day) + ADT*
(n=271)29.3 (4.1)
-14.6 (0.6)
-2.2 (-3.7, -0.6)
Placebo + ADT
(n=264)28.9 (4.3)
-12.5 (0.5) SD: standard deviation; SE: standard error; LS Mean: least-squares mean; CI: unadjusted confidence interval
* Dosages statistically significantly superior to placebo
a Difference (drug minus placebo) in least-squares mean change from baselineExamination of population subgroups based on age, sex, and race did not suggest any clear evidence of differential responsiveness.
Figure 6. LS Mean‡ Change from Baseline to Week 6 in MADRS Total Score in Adjunctive Treatment of Major Depressive Disorder (Study 10)
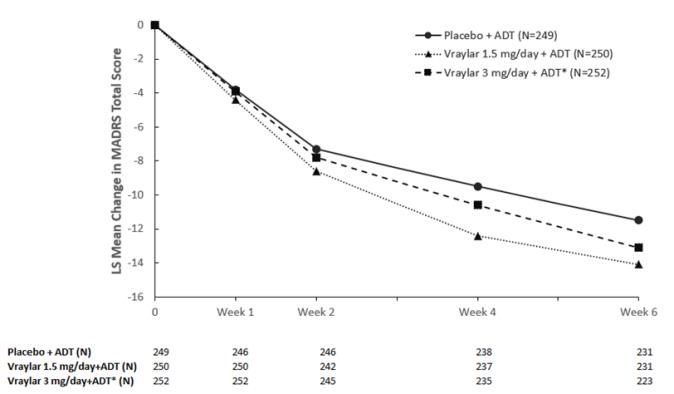
‡ LS Mean: least-squares mean
* Dose was not statistically significant.
- PANSS is a 30-item scale that measures positive symptoms of schizophrenia (7 items), negative symptoms of schizophrenia (7 items), and general psychopathology (16 items), each rated on a scale of 1 (absent) to 7 (extreme). The PANSS total score may range from 30 to 210 with the higher score reflecting greater severity.
-
16
HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
VRAYLAR (cariprazine) capsules are supplied as follows:
Capsule
StrengthImprint
CodesCapsule Color Package Configuration NDC Code 1.5 mg FL 1.5 White cap
and bodyBlister pack of 7 61874-115-17 Bottle of 30 61874-115-30 Bottle of 90 61874-115-90 Box of 20 (Hospital Unit Dose) 61874-115-20 3 mg FL 3 Green to blue-green cap and white body Bottle of 30 61874-130-30 Bottle of 90 61874-130-90 Box of 20 (Hospital Unit Dose) 61874-130-20 4.5 mg FL 4.5 Green to blue-green cap and body Bottle of 30 61874-145-30 Bottle of 90 61874-145-90 6 mg FL 6 Purple cap and
white bodyBottle of 30 61874-160-30 Bottle of 90 61874-160-90 (1) 1.5 mg,
(6) 3 mgFL 1.5,
FL 3Mixed Blister pack of 7 61874-170-08 -
17
PATIENT COUNSELING INFORMATION
Advise the patient or caregiver to read the FDA-approved patient labeling (Medication Guide).
Suicidal Thoughts and Behaviors
Advise patients and caregivers to look for the emergence of suicidal thoughts and behaviors, especially early during treatment and when the dosage is adjusted up or down and instruct them to report such symptoms to their healthcare provider [see Box Warning and Warnings and Precautions (5.2)].
Dosage and Administration
Advise patients that VRAYLAR can be taken with or without food. Counsel them on the importance of following dosage escalation instructions [see Dosage and Administration (2)].
Neuroleptic Malignant Syndrome (NMS)
Counsel patients about a potentially fatal adverse reaction, Neuroleptic Malignant Syndrome (NMS), that has been reported in association with administration of antipsychotic drugs. Advise patients, family members, or caregivers to contact the healthcare provider or to report to the emergency room if they experience signs and symptoms of NMS [see Warnings and Precautions (5.4)].
Tardive Dyskinesia
Counsel patients on the signs and symptoms of tardive dyskinesia and to contact their health care provider if these abnormal movements occur [see Warnings and Precautions (5.5)].
Late-Occurring Adverse Reactions
Counsel patients that adverse reactions may not appear until several weeks after the initiation of VRAYLAR treatment [see Warnings and Precautions (5.6)].
Metabolic Changes (Hyperglycemia and Diabetes Mellitus, Dyslipidemia, and Weight Gain)
Educate patients about the risk of metabolic changes, how to recognize symptoms of hyperglycemia and diabetes mellitus, and the need for specific monitoring, including blood glucose, lipids, and weight [see Warnings and Precautions (5.7)].
Leukopenia/Neutropenia
Advise patients with a pre-existing low WBC or a history of drug-induced leukopenia/neutropenia that they should have their CBC monitored while taking VRAYLAR [see Warnings and Precautions (5.8)].
Orthostatic Hypotension and Syncope
Counsel patients on the risk of orthostatic hypotension and syncope, especially early in treatment, and also at times of re-initiating treatment or increases in dose [see Warnings and Precautions (5.9)].
Interference with Cognitive and Motor Performance
Caution patients about performing activities requiring mental alertness, such as operating hazardous machinery or operating a motor vehicle, until they are reasonably certain that VRAYLAR therapy does not affect them adversely [see Warnings and Precautions (5.12)].
Heat Exposure and Dehydration
Educate patients regarding appropriate care in avoiding overheating and dehydration [see Warnings and Precautions (5.13)].
Concomitant Medications
Advise patients to notify their physicians if they are taking, or plan to take, any prescription or over-the-counter drugs since there is a potential for interactions [see Drug Interactions (7.1)].
Pregnancy
Advise patients that third trimester use of VRAYLAR may cause extrapyramidal and/or withdrawal symptoms in a neonate. Advise patients to notify their healthcare provider with a known or suspected pregnancy [see Use in Specific Populations (8.1)].
Pregnancy Registry
Advise patients that there is a pregnancy exposure registry that monitors pregnancy outcomes in women exposed to VRAYLAR during pregnancy [see Use in Specific Populations (8.1)].
Licensed from Gedeon Richter Plc.
Manufactured by:
Forest Laboratories Ireland Limited
Dublin, IE.Distributed by:
AbbVie Inc.
North Chicago, IL 60064, USAVRAYLAR and its design are trademarks of Allergan Pharmaceuticals International Limited, an AbbVie company.
© 2024 AbbVie. All rights reserved.
v7.0USPI115
-
MEDICATION GUIDE
MEDICATION GUIDE
VRAYLAR® (VRAY-lar)
(cariprazine)
capsulesWhat is the most important information I should know about VRAYLAR?
VRAYLAR may cause serious side effects, including:
-
Increased risk of death in elderly people with dementia related psychosis. Medicines like VRAYLAR can raise the risk of death in elderly who have lost touch with reality (psychosis) due to confusion and memory loss (dementia). VRAYLAR is not approved for the treatment of patients with dementia-related psychosis.
-
Increased risk of suicidal thoughts and actions. VRAYLAR and antidepressant medicines may increase suicidal thoughts or actions in some children and young adults especially within the first few months of treatment or when the dose is changed.
○ Depression and other mental illnesses are the most important causes of suicidal thoughts and actions.
○ Pay close attention to any changes, especially sudden changes in mood, behaviors, thoughts, or feelings. This is very important when VRAYLAR or the antidepressant medicine is started or when the dose is changed.
○ Call the healthcare provider right away to report new or sudden changes in mood, behavior, thoughts, or feelings, or if you develop suicidal thoughts or actions.
○ Keep all follow-up visits with the healthcare provider as scheduled. Call the healthcare provider between visits as needed, especially if you have concerns about symptoms.
Call a healthcare provider right away if you or your family member has any of the following symptoms, especially if they are new, worse, or worry you:- thoughts about suicide or dying
- attempts to commit suicide
- new or worse depression
- new or worse anxiety
- feeling very agitated or restless
- panic attacks
- trouble sleeping (insomnia)
- new or worse irritability
- acting aggressive, being angry, or violent
- acting on dangerous impulses
- an extreme increase in activity and talking (mania)
- other unusual changes in behavior or mood
What is VRAYLAR?
VRAYLAR is a prescription medicine used in adults:
- to treat schizophrenia
- for short-term (acute) treatment of manic or mixed episodes that happen with bipolar I disorder
- to treat depressive episodes that happen with bipolar I disorder (bipolar depression)
- along with antidepressant medicines to treat major depressive disorder (MDD)
Do not take VRAYLAR if you are allergic to cariprazine. See the end of this Medication Guide for a complete list of ingredients in VRAYLAR. Before taking VRAYLAR, tell your healthcare provider about all of your medical conditions, including if you:
- have or have had heart problems or a stroke
- have or have had low or high blood pressure
- have or have had diabetes or high blood sugar, or a family history of diabetes or high blood sugar. Your healthcare provider should check your blood sugar before you start and during treatment with VRAYLAR.
- have or have had high levels of total cholesterol, LDL cholesterol, or triglycerides or low levels of HDL cholesterol.
- have or had seizures (convulsions)
- have or have had kidney or liver problems
- have or had a low white blood cell count
- are pregnant or plan to become pregnant. VRAYLAR may harm your unborn baby. Taking VRAYLAR during your third trimester of pregnancy may cause your baby to have abnormal muscle movements or withdrawal symptoms after birth. Talk to your healthcare provider about the risk to your unborn baby if you take VRAYLAR during pregnancy.
○ Tell your healthcare provider if you become pregnant or think you are pregnant during treatment with VRAYLAR.
○ If you become pregnant during treatment with VRAYLAR, talk to your healthcare provider about registering with the National Pregnancy Registry for Atypical Antipsychotics. You can register by calling 1-866-961-2388 or go to http://womensmentalhealth.org/clinical-and-research-programs/pregnancyregistry/.
- are breastfeeding or plan to breastfeed. It is not known if VRAYLAR passes into your breast milk. Talk to your healthcare provider about the best way to feed your baby during treatment with VRAYLAR.
VRAYLAR and other medicines may affect each other causing possible serious side effects. VRAYLAR may affect the way other medicines work, and other medicines may affect how VRAYLAR works.
Your healthcare provider can tell you if it is safe to take VRAYLAR with your other medicines. Do not start or stop any medicines while taking VRAYLAR without talking to your healthcare provider first.
Know the medicines you take. Keep a list of your medicines to show your healthcare provider and pharmacist when you get a new medicine.How should I take VRAYLAR? - Take VRAYLAR exactly as your healthcare provider tells you to take it. Do not change the dose or stop taking VRAYLAR without first talking to your healthcare provider.
- Take VRAYLAR 1 time each day with or without food.
- If you take too much VRAYLAR, call your healthcare provider or Poison Control Center at 1-800-222-1222 or go to the nearest hospital emergency room, right away.
What should I avoid while taking VRAYLAR? - Do not drive, operate machinery, or do other dangerous activities until you know how VRAYLAR affects you. VRAYLAR may make you drowsy.
- Do not become too hot or dehydrated during treatment with VRAYLAR.
○ Do not exercise too much.
○ In hot weather, stay inside in a cool place if possible.
○ Stay out of the sun.
○ Do not wear too much clothing or heavy clothing.
○ Drink plenty of water.
What are the possible side effects of VRAYLAR?
VRAYLAR may cause serious side effects, including:-
See “What is the most important information I should know about VRAYLAR?”
-
Stroke (cerebrovascular problems) in elderly people with dementia-related psychosis that can lead to death.
- Neuroleptic malignant syndrome (NMS) is a serious condition that can lead to death. Call your healthcare provider or go to the nearest hospital emergency room right away if you have some or all of the following signs and symptoms of NMS:
○ high fever ○ stiff muscles ○ confusion ○ increased sweating ○ changes in your breathing, heart rate, and blood pressure -
Uncontrolled body movements (tardive dyskinesia). VRAYLAR may cause movements that you cannot control in your face, tongue, or other body parts. Tardive dyskinesia may not go away, even if you stop taking VRAYLAR. Tardive dyskinesia may also start after you stop taking VRAYLAR.
-
Late occurring side effects. VRAYLAR stays in your body for a long time. Some side effects may not happen right away and can start a few weeks after you start taking VRAYLAR, or if your dose of VRAYLAR increases. Your healthcare provider should monitor you for side effects for several weeks after you start and after any increase in your dose of VRAYLAR.
-
Problems with your metabolism such as:
○ high blood sugar (hyperglycemia) and diabetes. Increases in blood sugar can happen in some people who take VRAYLAR. Extremely high blood sugar can lead to coma or death. Your healthcare provider should check your blood sugar before you start, or soon after you start VRAYLAR, and then regularly during long-term treatment with VRAYLAR.
• feel very thirsty • need to urinate more than usual
• feel very hungry • feel weak or tired
• feel sick to your stomach • feel confused, or your breath smells fruity
-
increased fat levels (cholesterol and triglycerides) in your blood. Your healthcare provider should check the fat levels in your blood before you start, or soon after you start VRAYLAR, and then periodically during treatment with VRAYLAR.
- weight gain. You and your healthcare provider should check your weight before you start and often during treatment with VRAYLAR.
-
Low white blood cell count. Your healthcare provider may do blood tests during the first few months of treatment with VRAYLAR.
-
Decreased blood pressure (orthostatic hypotension). You may feel lightheaded or faint when you rise too quickly from a sitting or lying position.
-
Falls. VRAYLAR may make you sleepy or dizzy, may cause a decrease in your blood pressure when changing position (orthostatic hypotension), and can slow your thinking and motor skills which may lead to falls that can cause fractures or other injuries.
-
Seizures (convulsions).
-
Sleepiness, drowsiness, feeling tired, difficulty thinking and doing normal activities. See “What should I avoid while taking VRAYLAR?”
-
Problems controlling your body temperature so that you feel too warm. See “What should I avoid while taking VRAYLAR?”
- Difficulty swallowing that can cause food or liquid to get into your lungs.
These are not all the possible side effects of VRAYLAR.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.How should I store VRAYLAR?
- Store VRAYLAR at room temperature, between 68°F to 77°F (20°C to 25°C).
General information about the safe and effective use of VRAYLAR.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use VRAYLAR for a condition for which it was not prescribed. Do not give VRAYLAR to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about VRAYLAR that is written for healthcare professionals.What are the ingredients in VRAYLAR?
Active ingredient: cariprazine
Inactive ingredients: gelatin, magnesium stearate, pregelatinized starch, shellac, and titanium dioxide.
Colorants include: black iron oxide, FD&C Blue 1, FD&C Red 3, FD&C Red 40, or yellow iron oxide.
Manufactured by: Forest Laboratories Ireland Limited, Dublin, IE.
Distributed by: AbbVie Inc. North Chicago, IL 60064, USA
©2024 AbbVie. All rights reserved.
VRAYLAR and its design are trademarks of Allergan Pharmaceuticals International Limited, an AbbVie company.
For more information, go to www.VRAYLAR.com or call 1-800-678-1605.This Medication Guide has been approved by the U.S. Food and Drug Administration. Revised 11/2024
v7.0MG115
-
Increased risk of death in elderly people with dementia related psychosis. Medicines like VRAYLAR can raise the risk of death in elderly who have lost touch with reality (psychosis) due to confusion and memory loss (dementia). VRAYLAR is not approved for the treatment of patients with dementia-related psychosis.
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
VRAYLAR
cariprazine capsule, gelatin coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:61874-115 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CARIPRAZINE (UNII: F6RJL8B278) (CARIPRAZINE - UNII:F6RJL8B278) CARIPRAZINE 1.5 mg Inactive Ingredients Ingredient Name Strength GELATIN, UNSPECIFIED (UNII: 2G86QN327L) MAGNESIUM STEARATE (UNII: 70097M6I30) STARCH, CORN (UNII: O8232NY3SJ) SHELLAC (UNII: 46N107B71O) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERROSOFERRIC OXIDE (UNII: XM0M87F357) Product Characteristics Color white (WHITE) , white (WHITE) Score no score Shape CAPSULE (CAPSULE) Size 14mm Flavor Imprint Code FL;1;5 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:61874-115-30 30 in 1 BOTTLE; Type 0: Not a Combination Product 03/01/2016 2 NDC:61874-115-90 90 in 1 BOTTLE; Type 0: Not a Combination Product 03/01/2016 05/24/2019 3 NDC:61874-115-20 2 in 1 CARTON 03/01/2016 3 NDC:61874-115-11 10 in 1 BLISTER PACK; Type 0: Not a Combination Product 4 NDC:61874-115-17 1 in 1 CARTON 03/01/2016 4 7 in 1 BLISTER PACK; Type 0: Not a Combination Product 5 NDC:61874-115-07 7 in 1 BLISTER PACK; Type 0: Not a Combination Product 03/01/2016 6 NDC:61874-115-31 5 in 1 TRAY 03/01/2016 03/01/2016 6 1 in 1 CARTON 6 30 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA204370 09/17/2015 VRAYLAR
cariprazine capsule, gelatin coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:61874-130 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CARIPRAZINE (UNII: F6RJL8B278) (CARIPRAZINE - UNII:F6RJL8B278) CARIPRAZINE 3 mg Inactive Ingredients Ingredient Name Strength GELATIN, UNSPECIFIED (UNII: 2G86QN327L) MAGNESIUM STEARATE (UNII: 70097M6I30) STARCH, CORN (UNII: O8232NY3SJ) SHELLAC (UNII: 46N107B71O) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERROSOFERRIC OXIDE (UNII: XM0M87F357) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FD&C RED NO. 40 (UNII: WZB9127XOA) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) Product Characteristics Color green (blue green) , white (WHITE) Score no score Shape CAPSULE (CAPSULE) Size 14mm Flavor Imprint Code FL;3 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:61874-130-30 30 in 1 BOTTLE; Type 0: Not a Combination Product 03/01/2016 2 NDC:61874-130-90 90 in 1 BOTTLE; Type 0: Not a Combination Product 03/01/2016 05/24/2019 3 NDC:61874-130-20 2 in 1 CARTON 03/01/2016 3 NDC:61874-130-11 10 in 1 BLISTER PACK; Type 0: Not a Combination Product 4 NDC:61874-130-07 7 in 1 BLISTER PACK; Type 0: Not a Combination Product 03/01/2016 5 NDC:61874-130-31 5 in 1 TRAY 03/01/2016 03/01/2016 5 1 in 1 CARTON 5 30 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA204370 09/17/2015 VRAYLAR
cariprazine capsule, gelatin coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:61874-145 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CARIPRAZINE (UNII: F6RJL8B278) (CARIPRAZINE - UNII:F6RJL8B278) CARIPRAZINE 4.5 mg Inactive Ingredients Ingredient Name Strength GELATIN, UNSPECIFIED (UNII: 2G86QN327L) MAGNESIUM STEARATE (UNII: 70097M6I30) STARCH, CORN (UNII: O8232NY3SJ) SHELLAC (UNII: 46N107B71O) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FD&C RED NO. 40 (UNII: WZB9127XOA) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) Product Characteristics Color green (blue green) Score no score Shape CAPSULE (CAPSULE) Size 14mm Flavor Imprint Code FL;4;5 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:61874-145-30 30 in 1 BOTTLE; Type 0: Not a Combination Product 03/01/2016 2 NDC:61874-145-90 90 in 1 BOTTLE; Type 0: Not a Combination Product 03/01/2016 05/24/2019 3 NDC:61874-145-07 7 in 1 BLISTER PACK; Type 0: Not a Combination Product 03/01/2016 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA204370 09/17/2015 VRAYLAR
cariprazine capsule, gelatin coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:61874-160 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CARIPRAZINE (UNII: F6RJL8B278) (CARIPRAZINE - UNII:F6RJL8B278) CARIPRAZINE 6 mg Inactive Ingredients Ingredient Name Strength GELATIN, UNSPECIFIED (UNII: 2G86QN327L) MAGNESIUM STEARATE (UNII: 70097M6I30) STARCH, CORN (UNII: O8232NY3SJ) SHELLAC (UNII: 46N107B71O) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERROSOFERRIC OXIDE (UNII: XM0M87F357) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FD&C RED NO. 3 (UNII: PN2ZH5LOQY) Product Characteristics Color purple (PURPLE) , white (WHITE) Score no score Shape CAPSULE (CAPSULE) Size 16mm Flavor Imprint Code FL;6 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:61874-160-30 30 in 1 BOTTLE; Type 0: Not a Combination Product 03/01/2016 2 NDC:61874-160-90 90 in 1 BOTTLE; Type 0: Not a Combination Product 03/01/2016 05/24/2019 3 NDC:61874-160-07 7 in 1 BLISTER PACK; Type 0: Not a Combination Product 03/01/2016 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA204370 09/17/2015 VRAYLAR
cariprazine kitProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:61874-170 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:61874-170-08 1 in 1 CARTON 03/01/2016 05/31/2024 1 1 in 1 BLISTER PACK; Type 0: Not a Combination Product Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 BLISTER PACK 1 Part 2 1 BLISTER PACK 6 Part 1 of 2 VRAYLAR
cariprazine capsule, gelatin coatedProduct Information Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CARIPRAZINE (UNII: F6RJL8B278) (CARIPRAZINE - UNII:F6RJL8B278) CARIPRAZINE 1.5 mg Inactive Ingredients Ingredient Name Strength GELATIN, UNSPECIFIED (UNII: 2G86QN327L) MAGNESIUM STEARATE (UNII: 70097M6I30) STARCH, CORN (UNII: O8232NY3SJ) SHELLAC (UNII: 46N107B71O) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERROSOFERRIC OXIDE (UNII: XM0M87F357) Product Characteristics Color white (WHITE) , white (WHITE) Score no score Shape CAPSULE (CAPSULE) Size 14mm Flavor Imprint Code FL;1;5 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 1 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA204370 09/17/2015 Part 2 of 2 VRAYLAR
cariprazine capsule, gelatin coatedProduct Information Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CARIPRAZINE (UNII: F6RJL8B278) (CARIPRAZINE - UNII:F6RJL8B278) CARIPRAZINE 3 mg Inactive Ingredients Ingredient Name Strength GELATIN, UNSPECIFIED (UNII: 2G86QN327L) MAGNESIUM STEARATE (UNII: 70097M6I30) STARCH, CORN (UNII: O8232NY3SJ) SHELLAC (UNII: 46N107B71O) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERROSOFERRIC OXIDE (UNII: XM0M87F357) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FD&C RED NO. 40 (UNII: WZB9127XOA) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) Product Characteristics Color green (blue green) , white (WHITE) Score no score Shape CAPSULE (CAPSULE) Size 14mm Flavor Imprint Code FL;3 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 6 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA204370 09/17/2015 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA204370 09/17/2015 Labeler - Allergan, Inc. (144796497)