Label: NU-DERM SYSTEM NORMAL-OILY SKIN TRANSFORMATION- hydroquinone, homosalate, octisalate, and zinc oxide kit
- NDC Code(s): 62032-531-00
- Packager: OBAGI COSMECEUTICAL LLC
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated January 19, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
-
DESCRIPTION
Hydroquinone, USP 4% is 1, 4-benzenediol. The drug is freely soluble in water and in alcohol. Chemically, hydroquinone is designated as p-dihydroxybenzene; the empirical formula is C6H6O2; molecular weight is 110.11 g/mol. The chemical structure is in the diagram below.
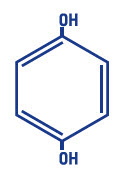
C6H6O2
Each gram of Obagi Nu-Derm® Clear contains:
ACTIVE: Hydroquinone, USP 4% (40 mg/g)
INACTIVES: ascorbic acid, BHT, butylparaben, cetyl alcohol, disodium EDTA, glycerin, lactic acid, methylparaben, propylparaben, saponins, sodium lauryl sulfate, sodium metabisulfite, stearyl alcohol, tocopheryl acetate, water (aqua)
Each gram of Obagi Nu-Derm Blender® contains:
ACTIVE: Hydroquinone, USP 4% (40 mg/g)
INACTIVES: ascorbic acid, BHT, cetyl alcohol, disodium EDTA, glycerin, lactic acid, methylparaben, phenyl trimethicone, PPG-2 myristyl ether propionate, propylparaben, saponins, sodium lauryl sulfate, sodium metabisulfite, TEA-salicylate, tocopheryl acetate, water (aqua)
Each gram of Obagi Nu-Derm® Sunfader® contains:
ACTIVES: Hydroquinone, USP 4% (40mg/g); Octinoxate, USP 7.5%; Oxybenzone, USP 5.5%
INACTIVES: ascorbic acid, BHT, butylparaben, cetyl alcohol, disodium EDTA, glycerin, methylparaben, propylparaben, saponins, sodium lauryl sulfate, sodium metabisulfite, stearyl alcohol, tocopheryl acetate, water (aqua)
-
CLINICAL PHARMACOLOGY
Topical application of hydroquinone produces a reversible depigmentation of the skin by inhibition of the enzymatic oxidation of tyrosine to 3, 4-dihydroxyphenylalanine (DOPA) and suppression of other melanocyte metabolic processes. Exposure to sunlight or ultraviolet light will cause repigmentation of the bleached areas, which may be prevented by the use of sunscreen agents such as those contained in Obagi Nu-Derm Sunfader® and Obagi Nu-Derm® Sun Shield Matte Broad Spectrum SPF 50.
- INDICATIONS AND USAGE
- CONTRAINDICATIONS
-
WARNINGS
Hydroquinone is a skin bleaching agent, which may produce unwanted cosmetic effects if not used as directed. The physician should be familiar with the contents of this insert before prescribing or dispensing this product.
Test for skin sensitivity before using by applying a small amount to an unbroken patch of skin and check within 24 hours. Some users of this product may experience a mild skin irritation. If skin irritation becomes severe, stop use and consult a doctor. Close patient supervision is recommended.
-
WARNINGS
Avoid contact with eyes, nose, mouth and lips. In case of accidental contact, patient should rinse thoroughly with water and contact a physician. Sunscreen use is an essential aspect of hydroquinone therapy because even minimal sunlight exposure sustains melanocytic activity.
Contains sodium metabisulfite, a sulfite that may cause allergic-type reactions including anaphylactic symptoms and life-threatening or less severe asthmatic episodes in certain susceptible people. The overall prevalence of sulfite sensitivity in the general population is unknown and probably low. Sulfite sensitivity is seen more frequently in asthmatic than in nonasthmatic people.
-
PRECAUTIONS
(also see WARNINGS):
Treatment should be limited to relatively small areas of the body at one time since some patients experience a transient skin reddening and a mild burning sensation, which does not preclude treatment.
PREGNANCY CATEGORY C
Animal reproduction studies have not been conducted with topical hydroquinone. It is also not known whether hydroquinone can cause fetal harm when used topically on a pregnant woman or affect reproductive capacity. It is not known to what degree, if any, topical hydroquinone is absorbed systemically. Topical hydroquinone should be used on pregnant women only when clearly indicated.
-
ADVERSE REACTIONS
No systemic adverse reactions have been reported. Occasional hypersensitivity (localized contact dermatitis) may occur, in which case the product should be discontinued and the physician notified immediately.
To report SUSPECTED ADVERSE REACTIONS, contact Obagi Cosmeceuticals LLC, at 1-800-636-7546 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
-
DOSAGE AND ADMINISTRATION
A thin application should be applied once or twice daily or as directed by a physician. If no improvement is seen after three (3) months of treatment, use of this product should be discontinued. Sun exposure should be limited by using a sunscreen agent or protective clothing to cover bleached skin when using and after using this product in order to prevent darkening from reoccurring.
- HOW SUPPLIED
-
SPL UNCLASSIFIED SECTION
 1-800-636-7546
1-800-636-7546
Manufactured for:
Obagi Cosmeceuticals LLC,
Long Beach, CA 90806All products/brand names, whether designated by notice or not (®/TM), are trademarks of Obagi Cosmeceuticals LLC and/or its affiliates.
©2019 Obagi Cosmeceuticals LLC.
All rights reserved. www.obagi.comRevised 01/2019 9458404
Obagi Nu-Derm® Clear (Hydroquinone, USP 4%)
Skin Bleaching CreamObagi Nu-Derm Blender® (Hydroquinone, USP 4%)
Skin Bleaching CreamObagi Nu-Derm Sunfader®
(Hydroquinone, USP 4%; Octinoxate, USP 7.5%; Oxybenzone, USP 5.5%) Skin Bleaching Cream with Sunscreens - SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
-
Uses
- helps prevent sunburn
- If used as directed with other sun protection measures (see Directions), decreases the risk of skin cancer and early skin aging caused by the sun
- Warnings
-
Directions
- apply liberally 15 minutes before sun exposure
- use a water resistant sunscreen if swimming or sweating
- reapply at least every 2 hours
-
Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
- limit time in the sun, especially from 10 a.m.–2 p.m.
- wear long-sleeved shirts, pants, hats, and sunglasses
- children under 6 months: Ask a doctor
- Other information
-
Inactive ingredients
Water (Aqua), C15-19 Alkane, Octyldodecyl Neopentanoate, Polymethylsilsesquioxane, Sorbitan Olivate, Silica, Polyglyceryl-6 Polyrininoleate, Sodium Chloride, Xanthan Gum, Glycerin, Hydroxyacetophenone, Disodium EDTA, 1,2-Hexanediol, Caprylyl Glycol, Sodium Hydroxide, Triethoxycaprylsilane, Polyhydroxystearic Acid, Disteardimonium Hectorite, Polyglyceryl-2 Isostearate, Euphorbia Cerifera (candelilla) Wax, Beeswax, Dimethicone
- Questions or comments?
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - Kit Carton
-
INGREDIENTS AND APPEARANCE
NU-DERM SYSTEM NORMAL-OILY SKIN TRANSFORMATION
hydroquinone, homosalate, octisalate, and zinc oxide kitProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:62032-531 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:62032-531-00 1 in 1 CARTON 12/02/2019 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 BOTTLE, PLASTIC 57 g Part 2 1 BOTTLE, PLASTIC 198 mL Part 3 1 BOTTLE, PLASTIC 198 mL Part 4 1 BOTTLE, PLASTIC 57 g Part 5 1 BOTTLE, PLASTIC 48 g Part 6 1 BOTTLE, PLASTIC 57 g Part 7 1 TUBE 85 g Part 1 of 7 OBAGI NU-DERM EXFODERM FORTE
face and neck (excluding shaving preparations) lotionProduct Information Route of Administration TOPICAL Other Ingredients Ingredient Kind Ingredient Name Quantity INGR WATER (UNII: 059QF0KO0R) INGR GLYCERIN (UNII: PDC6A3C0OX) INGR DIMETHICONE (UNII: 92RU3N3Y1O) INGR METHYLPARABEN (UNII: A2I8C7HI9T) INGR PROPYLPARABEN (UNII: Z8IX2SC1OH) INGR POLYSORBATE 60 (UNII: CAL22UVI4M) INGR TROLAMINE (UNII: 9O3K93S3TK) INGR MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) INGR CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) INGR CETYL ALCOHOL (UNII: 936JST6JCN) INGR LACTIC ACID, UNSPECIFIED FORM (UNII: 33X04XA5AT) INGR EMU OIL (UNII: 344821WD61) INGR STEARIC ACID (UNII: 4ELV7Z65AP) INGR STEARYL ALCOHOL (UNII: 2KR89I4H1Y) INGR Glycolic Acid (UNII: 0WT12SX38S) Product Characteristics color WHITE C48325 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 57 g in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date COSMETIC 01/01/1988 Part 2 of 7 OBAGI NU-DERM FOAMING
cleansing (cold creams, cleansing lotions, liquids, and pads) gelProduct Information Route of Administration TOPICAL Other Ingredients Ingredient Kind Ingredient Name Quantity INGR WATER (UNII: 059QF0KO0R) INGR PHENOXYETHANOL (UNII: HIE492ZZ3T) INGR METHYLPARABEN (UNII: A2I8C7HI9T) INGR PROPYLPARABEN (UNII: Z8IX2SC1OH) INGR BUTYLPARABEN (UNII: 3QPI1U3FV8) INGR ETHYLPARABEN (UNII: 14255EXE39) INGR ISOBUTYLPARABEN (UNII: 0QQJ25X58G) INGR FD&C YELLOW NO. 5 (UNII: I753WB2F1M) INGR SODIUM LAUROYL OAT AMINO ACIDS (UNII: FSW2K9B9N5) INGR COCAMIDOPROPYL BETAINE (UNII: 5OCF3O11KX) INGR SODIUM LAURETH-3 SULFATE (UNII: BPV390UAP0) INGR ALOE VERA LEAF (UNII: ZY81Z83H0X) INGR SODIUM CHLORIDE (UNII: 451W47IQ8X) INGR MEDICAGO SATIVA WHOLE (UNII: DJO934BRBD) INGR CHAMOMILE (UNII: FGL3685T2X) INGR XANTHAN GUM (UNII: TTV12P4NEE) INGR D&C RED NO. 33 (UNII: 9DBA0SBB0L) Product Characteristics color RED C48326 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 198 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date COSMETIC 01/01/1988 Part 3 of 7 OBAGI NU-DERM TONER
cleansing (cold creams, cleansing lotions, liquids, and pads) liquidProduct Information Route of Administration TOPICAL Other Ingredients Ingredient Kind Ingredient Name Quantity INGR WATER (UNII: 059QF0KO0R) INGR GLYCERIN (UNII: PDC6A3C0OX) INGR HAMAMELIS VIRGINIANA TOP WATER (UNII: NT00Y05A2V) INGR SODIUM PYRROLIDONE CARBOXYLATE (UNII: 469OTG57A2) INGR DMDM HYDANTOIN (UNII: BYR0546TOW) INGR IODOPROPYNYL BUTYLCARBAMATE (UNII: 603P14DHEB) INGR POTASSIUM ALUM (UNII: 1L24V9R23S) INGR PANTHENOL (UNII: WV9CM0O67Z) INGR SAGE (UNII: 065C5D077J) INGR CALENDULA OFFICINALIS FLOWER (UNII: P0M7O4Y7YD) INGR POLYSORBATE 80 (UNII: 6OZP39ZG8H) INGR ALLANTOIN (UNII: 344S277G0Z) INGR ALOE VERA LEAF (UNII: ZY81Z83H0X) INGR FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Product Characteristics color BLUE C48333 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 198 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date COSMETIC 01/02/1988 Part 4 of 7 NU-DERM CLEAR SKIN BLEACHING AND CORRECTOR
hydroquinone creamProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HYDROQUINONE (UNII: XV74C1N1AE) (HYDROQUINONE - UNII:XV74C1N1AE) HYDROQUINONE 40 mg in 1 g Inactive Ingredients Ingredient Name Strength EDETATE DISODIUM (UNII: 7FLD91C86K) BUTYLPARABEN (UNII: 3QPI1U3FV8) STEARYL ALCOHOL (UNII: 2KR89I4H1Y) SODIUM LAURYL SULFATE (UNII: 368GB5141J) CETYL ALCOHOL (UNII: 936JST6JCN) GLYCERIN (UNII: PDC6A3C0OX) LACTIC ACID, UNSPECIFIED FORM (UNII: 33X04XA5AT) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) ASCORBIC ACID (UNII: PQ6CK8PD0R) SODIUM METABISULFITE (UNII: 4VON5FNS3C) WATER (UNII: 059QF0KO0R) METHYLPARABEN (UNII: A2I8C7HI9T) PROPYLPARABEN (UNII: Z8IX2SC1OH) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) Product Characteristics Color WHITE Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 57 g in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date UNAPPROVED DRUG OTHER 01/01/1988 Part 5 of 7 OBAGI NU-DERM HYDRATE FACIAL MOISTURIZER
face and neck (excluding shaving preparations)Product Information Route of Administration TOPICAL Other Ingredients Ingredient Kind Ingredient Name Quantity INGR WATER (UNII: 059QF0KO0R) INGR GLYCERIN (UNII: PDC6A3C0OX) INGR MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) INGR SODIUM HYDROXIDE (UNII: 55X04QC32I) INGR CAESALPINIA SPINOSA RESIN (UNII: WL3883U2PO) INGR SHEA BUTTER (UNII: K49155WL9Y) INGR DIMETHICONE/VINYL DIMETHICONE CROSSPOLYMER (SOFT PARTICLE) (UNII: 9E4CO0W6C5) INGR CYCLOMETHICONE 5 (UNII: 0THT5PCI0R) INGR CETYL ALCOHOL (UNII: 936JST6JCN) INGR SACCHARIDE ISOMERATE (UNII: W8K377W98I) INGR DIMETHICONE (UNII: 92RU3N3Y1O) INGR TOCOPHEROL (UNII: R0ZB2556P8) INGR LAURETH-12 (UNII: OAH19558U1) INGR PHENOXYETHANOL (UNII: HIE492ZZ3T) INGR ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) INGR STEARIC ACID (UNII: 4ELV7Z65AP) INGR AVOCADO OIL (UNII: 6VNO72PFC1) INGR SOY STEROL (UNII: PL360EPO9J) INGR CAPRYLYL GLYCOL (UNII: 00YIU5438U) INGR LEVOMENOL (UNII: 24WE03BX2T) INGR HEXYLENE GLYCOL (UNII: KEH0A3F75J) INGR TETRAHYDRODIFERULOYLMETHANE (UNII: 00U0645U03) INGR PANTHENOL (UNII: WV9CM0O67Z) INGR MANGIFERA INDICA SEED BUTTER (UNII: 4OXD9M35X2) INGR SODIUM STEAROYL GLUTAMATE (UNII: 65A9F4P024) INGR CARBOMER HOMOPOLYMER, UNSPECIFIED TYPE (UNII: 0A5MM307FC) INGR ALLANTOIN (UNII: 344S277G0Z) INGR GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) Product Characteristics color WHITE C48325 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 48 g in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date COSMETIC 11/07/2012 Part 6 of 7 NU-DERM BLENDER SKIN LIGHTENER AND BLENDING
hydroquinone creamProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HYDROQUINONE (UNII: XV74C1N1AE) (HYDROQUINONE - UNII:XV74C1N1AE) HYDROQUINONE 40 mg in 1 g Inactive Ingredients Ingredient Name Strength EDETATE DISODIUM (UNII: 7FLD91C86K) PPG-2 MYRISTYL ETHER PROPIONATE (UNII: 88R97D8U8A) TROLAMINE SALICYLATE (UNII: H8O4040BHD) SODIUM LAURYL SULFATE (UNII: 368GB5141J) CETYL ALCOHOL (UNII: 936JST6JCN) GLYCERIN (UNII: PDC6A3C0OX) LACTIC ACID, UNSPECIFIED FORM (UNII: 33X04XA5AT) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) ASCORBIC ACID (UNII: PQ6CK8PD0R) SODIUM METABISULFITE (UNII: 4VON5FNS3C) WATER (UNII: 059QF0KO0R) METHYLPARABEN (UNII: A2I8C7HI9T) PROPYLPARABEN (UNII: Z8IX2SC1OH) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) PHENYL TRIMETHICONE (UNII: DR0K5NOJ4R) Product Characteristics Color WHITE Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 57 g in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date UNAPPROVED DRUG OTHER 01/01/1988 Part 7 of 7 NU-DERM SUN SHIELD MATTE BROAD SPECTRUM SPF 50 SUNSCREEN
homosalate, octisalate, and zinc oxide lotionProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HOMOSALATE (UNII: V06SV4M95S) (HOMOSALATE - UNII:V06SV4M95S) HOMOSALATE 100 mg in 1 g OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 50 mg in 1 g ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 165 mg in 1 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) GLYCERIN (UNII: PDC6A3C0OX) Hydroxyacetophenone (UNII: G1L3HT4CMH) XANTHAN GUM (UNII: TTV12P4NEE) Sodium Chloride (UNII: 451W47IQ8X) Sodium Hydroxide (UNII: 55X04QC32I) Octyldodecyl Neopentanoate (UNII: X8725R883T) Triethoxycaprylylsilane (UNII: LDC331P08E) C15-19 Alkane (UNII: CI87N1IM01) Disteardimonium Hectorite (UNII: X687XDK09L) POLYGLYCERYL-2 ISOSTEARATE (UNII: 7B8OE71MQC) Sorbitan Olivate (UNII: MDL271E3GR) DIMETHICONE (UNII: 92RU3N3Y1O) 1,2-HEXANEDIOL (UNII: TR046Y3K1G) CAPRYLYL GLYCOL (UNII: 00YIU5438U) EDETATE DISODIUM (UNII: 7FLD91C86K) CANDELILLA WAX (UNII: WL0328HX19) Yellow Wax (UNII: 2ZA36H0S2V) POLYMETHYLSILSESQUIOXANE (4.5 MICRONS) (UNII: 59Z907ZB69) Silicon Dioxide (UNII: ETJ7Z6XBU4) Product Characteristics Color WHITE Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 85 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 12/02/2019 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date UNAPPROVED DRUG OTHER 12/02/2019 Labeler - OBAGI COSMECEUTICAL LLC (790553353) Establishment Name Address ID/FEI Business Operations PURETEK CORPORATION 785961046 MANUFACTURE(62032-531) , LABEL(62032-531) , PACK(62032-531) Establishment Name Address ID/FEI Business Operations G.S. COSMECEUTICAL USA, INC. 017014734 MANUFACTURE(62032-531) Establishment Name Address ID/FEI Business Operations Swiss-American CDMO, LLC 080170933 MANUFACTURE(62032-531)


