Label: BACTOSHIELD CHG- chlorhexidine gluconate solution
-
Contains inactivated NDC Code(s)
NDC Code(s): 0116-1322-03, 0116-1322-08, 0116-1322-24, 0116-1322-41 - Packager: Xttrium Laboratories, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated November 5, 2019
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
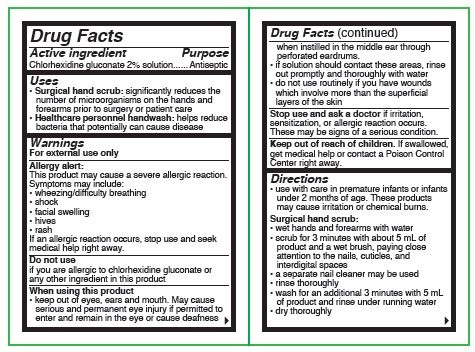
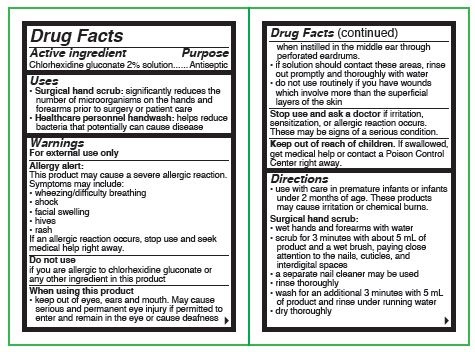
- Active ingredient
- Purposes
-
Uses
- surgical hand scrub: significantly reduces the number of microorganisms on the hands and forearms prior to surgery or patient care
- healthcare personnel handwash: helps reduce bacteria that potentially can cause disease
- patient preoperative skin preparation: preparation of the patient's skin prior to surgery
- skin wound and general skin cleansing
-
Warnings
For external use only
Allergy alert:
This product may cause a severe allergic reaction. Symptoms may include:
- wheezing/difficulty breathing
- shock
- facial swelling
- hives
- rash
If an allergic reaction occurs, stop use and seek medical help right away.
Do not use
- if you or the patient is allergic to chlorhexidine gluconate or any other ingredient in this product
- in contact with meninges
- in the genital area
- as a preoperative skin preparation of the head or face
When using this product
- keep out of eyes, ears and mouth. May cause serious and permanent eye injury if placed or kept in the eye during surgical procedures or may cause deafness when instilled in the middle ear through perforated eardrums
- if solution should contact these areas, rinse out promptly and thoroughly with water
- wounds which involve more than the superficial layers of the skin should not be routinely treated
- repeated general skin cleansing of large body areas should not be done except when advised by a health care provider
-
Directions
- use with care in premature infants or infants under 2 months of age. These products may cause irritation or chemical burns.
Surgical hand scrub:
- wet hands and forearms under running water for 30 seconds. Clean fingernails using a nailstick or similar cleaner.
- scrub for 1.5 minutes with about 8 mL of product with or without a wet brush paying close attention to the nails, cuticles, and skin between the fingers
- rinse thoroughly under the running water for 30 seconds
- wash for an additional 1.5 minutes with 8 mL of product and rinse under running water for 30 seconds
- dry thoroughly
Healthcare personnel handwash:
- wet hands with water
- dispense about 5 mL of product into cupped hands and wash in a vigorous manner for 15 seconds
- rinse and dry thoroughly
Patient preoperative skin preparation:
- apply product liberally to surgical site and swab for at least 2 minutes
- dry with a sterile towel
- repeat procedure for an additional 2 minutes and dry with a sterile towel
Skin wound and general skin cleansing:
- thoroughly rinse the area to e cleaned with water
- apply the minimum amount of product necessary to cover the skin or wound area and wash gently
- rinse again thoroughly
- Other information
- Inactive ingredients
- Questions or comments?
- OTHER SAFETY INFORMATION
-
PRINCIPAL DISPLAY PANEL
Bactoshield ® CHG 2%
NDC 11084-814-03
Surgical Scrub
Chlorhexidine Gluconate 2% Solution Antiseptic
Non-sterile Solution
Surgical Scrub
2% CHG
Antimicrobial
For Single-Use
Net Contents: 118 mL (4 fl oz)
1322-52G(319)
REORDER #1322-39
61345
2BACT04BTLLBLE




Bactoshield® CHG 2%
NDC 11084-814-24
Surgical Scrub
2% Solution Antiseptic
Chlorhexidine Gluconate
Surgical Scrub
2% CHG
Antimicrobial
Net Contents: 32 fl oz (946 mL)
REORDER #1322-24
61335
1322-21J(319)
2BACT32BTLLBLJ

Bactoshield ® CHG 2%
NDC 11084-814-08
Surgical Scrub
Chlorhexidine Gluconate 2% Solution Antiseptic
Surgical Scrub
2% CHG
Antimicrobial
Net Contents: 128 fl oz (1Gal)(3.78 Liters)
1322-06G(319)
REORDER #1322-08
62662
2BACT1GLBLB



NDC 11084-814-41
Bactoshield CHG 2%
Chlorhexidine Gluconate 2% Solution
Antiseptic
Healthcare Personnel Handwash
REORDER #: 1322-87
Manufactured for:
Deb USA, Inc.
Charlotte, NC 28217
1-866-783-0422
www.debmed.com
Contents: 33.8 fl oz (1.05 qt) (1 Liter)
61707
1322-86K (319)
SDSNATBKLBLA


NDC 11084-814-41
SDSNATFTLBLA

-
INGREDIENTS AND APPEARANCE
BACTOSHIELD CHG
chlorhexidine gluconate solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:0116-1322 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CHLORHEXIDINE GLUCONATE (UNII: MOR84MUD8E) (CHLORHEXIDINE - UNII:R4KO0DY52L) CHLORHEXIDINE GLUCONATE 20 mg in 1 mL Inactive Ingredients Ingredient Name Strength CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) COCO DIETHANOLAMIDE (UNII: 92005F972D) HYDROXYETHYL CELLULOSE (100 MPA.S AT 2%) (UNII: R33S7TK2EP) ISOPROPYL ALCOHOL (UNII: ND2M416302) LAURAMINE OXIDE (UNII: 4F6FC4MI8W) WATER (UNII: 059QF0KO0R) Product Characteristics Color yellow (Clear) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0116-1322-08 3708 mL in 1 BOTTLE; Type 0: Not a Combination Product 10/04/1999 2 NDC:0116-1322-24 946 mL in 1 BOTTLE; Type 0: Not a Combination Product 10/04/1999 3 NDC:0116-1322-03 118 mL in 1 BOTTLE; Type 0: Not a Combination Product 10/04/1999 4 NDC:0116-1322-41 1000 mL in 1 BOTTLE, DISPENSING; Type 0: Not a Combination Product 10/04/1999 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA019422 10/04/1999 Labeler - Xttrium Laboratories, Inc. (007470579) Registrant - Xttrium Laboratories, Inc. (007470579) Establishment Name Address ID/FEI Business Operations Xttrium Laboratories, Inc. 007470579 manufacture(0116-1322)