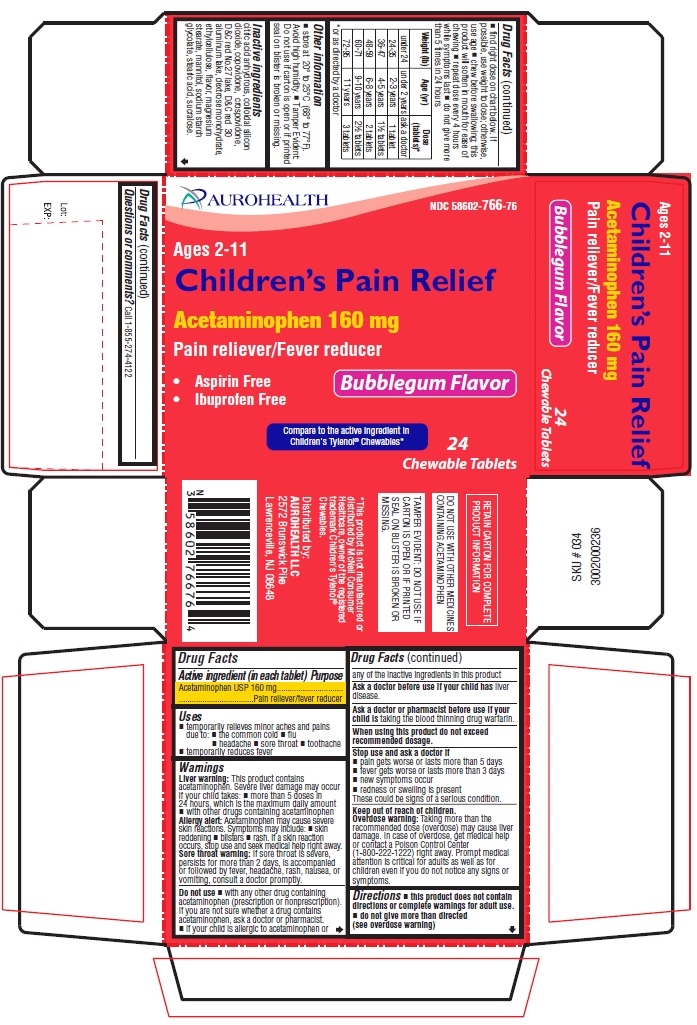
Label: CHILDRENS PAIN RELIEF- acetaminophen tablet, chewable
- NDC Code(s): 58602-766-76
- Packager: Aurohealth LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated November 29, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient (in each tablet)
- Purpose
- Uses
-
Warnings
-
Liver warning:This product contains acetaminophen. Severe liver damage may occur if your child takes:
- more than 5 doses in 24 hours, which is the maximum daily amount
- with other drugs containing acetaminophen
-
Allergy alert:Acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash.
If a skin reaction occurs, stop use and seek medical help right away.
- Sore throat warning:If sore throat is severe, persists for more than 2 days, is accompanied or followed by fever, headache, rash, nausea, or vomiting, consult a doctor promptly.
-
Liver warning:This product contains acetaminophen. Severe liver damage may occur if your child takes:
- Do not use
- Ask a doctor before use if your child has
- Ask a doctor or pharmacist before use if your child is
- When using this product
- Stop use and ask a doctor if
-
Keep out of reach of children.
Overdose warning:Taking more than the recommended dose (overdose) may cause liver damage. In case of overdose, get medical help or contact a Poison Control Center (1-800-222-1222) right away. Prompt medical attention is critical for adults as well as for children even if you do not notice any signs or symptoms.
-
Directions
- this product does not contain directions or complete warnings for adult use.
- do not give more than directed (see overdose warning)
- find right dose on chart below. If possible, use weight to dose; otherwise, use age
- chew before swallowing; this product will soften in mouth for ease of chewing
- repeat dose every 4 hours while symptoms last
- do not give more than 5 times in 24 hours
Weight (lb)
Age (yr)
Dose (tablets)*
* or as directed by a doctor under 24
under 2 years
ask a doctor
24-35
2-3 years
1 tablet
36-47
4-5 years
1½ tablets
48-59
6-8 years
2 tablets
60-71
9-10 years
2½ tablets
72-95
11 years
3 tablets
- Other information
-
Inactive ingredients
citric acid anhydrous, colloidal silicon dioxide, copovidone, crospovidone, D&C red No.27 lake, D&C red 30 aluminum lake, dextrose monohydrate, ethylcellulose, flavor, magnesium stearate, mannitol, sodium starch glycolate, stearic acid, sucralose.
Questions or comments?
Call 1-855-274-4122
* This product is not manufactured or distributed by McNeil Consumer Healthcare, owner of the registered trademark Children's Tylenol ®Chewables.
DO NOT USE WITH OTHER MEDICINES CONTAINING ACETAMINOPHEN
Distributed by:
AUROHEALTH LLC
2572 Brunswick Pike
Lawrenceville, NJ 08648 - PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 160 mg (24 Chewable Tablets)
-
INGREDIENTS AND APPEARANCE
CHILDRENS PAIN RELIEF
acetaminophen tablet, chewableProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:58602-766 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 160 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) COPOVIDONE K25-31 (UNII: D9C330MD8B) CROSPOVIDONE (120 .MU.M) (UNII: 68401960MK) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C RED NO. 30 (UNII: 2S42T2808B) DEXTROSE MONOHYDRATE (UNII: LX22YL083G) ETHYLCELLULOSE (4 MPA.S) (UNII: KC5472WRJK) MAGNESIUM STEARATE (UNII: 70097M6I30) MANNITOL (UNII: 3OWL53L36A) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) STEARIC ACID (UNII: 4ELV7Z65AP) SUCRALOSE (UNII: 96K6UQ3ZD4) Product Characteristics Color pink Score 2 pieces Shape ROUND Size 16mm Flavor BUBBLE GUM Imprint Code F8 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:58602-766-76 4 in 1 CARTON 09/13/2017 1 6 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M013 09/13/2017 Labeler - Aurohealth LLC (078728447) Establishment Name Address ID/FEI Business Operations APL HEALTHCARE LIMITED 650844777 manufacture(58602-766)