Label: CLOPHED- chlophedianol hydrochloride, guaifenesin, phenylephrine hydrochloride liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 49963-630-04, 49963-630-15 - Packager: Portal Pharmaceutical
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated December 30, 2010
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
-
Uses
temporarily relieves these symptoms due to the common cold, hay fever (allergic rhinitis) or other upper respiratory allergies:
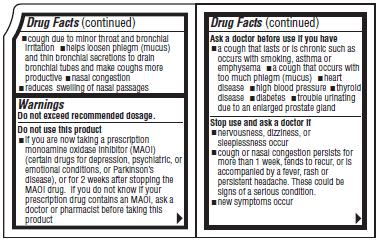
- cough due to minor throat and bronchial irritation
- helps loosen phelgm (mucus) and thin bronchial secretions to drain bronchial tubes and make coughs more productive
- nasal congestion
- reduces swelling of the nasal passages
-
Warnings
Do not exceed recommended dosage.
Do not use this product
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product
Ask a doctor before use if you have
- a cough that lasts or is chronic such as occurs with smoking, asthma or emphysema
- a cough that occurs with too much phlegm (mucus)
- heart disease
- high blood pressure
- thyroid disease
- diabetes
- trouble urinating due to an enlarged prostate gland
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product
-
Directions
Do not exceed recommended dosage.
Adults and children 12 years of age and over:
2 teaspoonfuls every 6 to 8 hours, not to exceed 8 teaspoonfuls in a 24 hour period
Children 6 to under 12 years of age:
1 teaspoonful every 6 to 8 hours, not to exceed 4 teaspoonfuls in a 24 hour period
Children under 6 years of age
Consult a doctor
- Other information
- Inactive ingredients
- Questions? Comments?
-
Product Packaging:
Packaging below represents the labeling currently used:
Principal display panel and side panel for 118 mL label:
NDC 49963-630-04
Clophed
Each teaspoonful (5 mL) for oral administration contains:
Chlophedianol HCl.....................................12.5 mg
Guaifenesin...............................................100 mg
Phenylephrine HCl.........................................5 mg
Cough Suppressant/Expectorant/Nasal Decongestant
Alcohol Free/Dye Free/Gluten Free/Sugar Free
4 fl oz (118 mL)
Tamper evident by foil seal under cap. Do not use if foil seal is broken or missing.
Supplied in a tight, light-resistant container with a child-resistant cap.
See package insert for full prescribing information.
Manufactured for:
Portal Pharmaceutical
Mayaguez, PR 00680 Rev. 10/09



-
INGREDIENTS AND APPEARANCE
CLOPHED
chlophedianol hydrochloride, guaifenesin, phenylephrine hydrochloride liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:49963-630 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Chlophedianol Hydrochloride (UNII: 69QQ58998Y) (Chlophedianol - UNII:42C50P12AP) Chlophedianol Hydrochloride 12.5 mg in 5 mL Guaifenesin (UNII: 495W7451VQ) (Guaifenesin - UNII:495W7451VQ) Guaifenesin 100 mg in 5 mL Phenylephrine Hydrochloride (UNII: 04JA59TNSJ) (Phenylephrine - UNII:1WS297W6MV) Phenylephrine Hydrochloride 5 mg in 5 mL Inactive Ingredients Ingredient Name Strength Citric Acid (UNII: 2968PHW8QP) Glycerin (UNII: PDC6A3C0OX) Propylene Glycol (UNII: 6DC9Q167V3) Water (UNII: 059QF0KO0R) Sodium Citrate (UNII: 1Q73Q2JULR) Saccharin Sodium (UNII: SB8ZUX40TY) Sorbitol (UNII: 506T60A25R) Product Characteristics Color Score Shape Size Flavor ORANGE, VANILLA Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:49963-630-04 118 mL in 1 BOTTLE 2 NDC:49963-630-15 15 mL in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part341 05/28/2010 Labeler - Portal Pharmaceutical (831005199)