Label: CYTRA-K CRYSTALS- potassium citrate and citric acid granule, for solution
-
Contains inactivated NDC Code(s)
NDC Code(s): 60258-005-01 - Packager: Cypress Pharmaceutical, Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated September 20, 2016
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
-
DESCRIPTION
CYTRA-K CRYSTALS (potassium citrate and citric acid for oral solution) is a pleasant-tasting oral systemic alkalizer containing potassium citrate and citric acid in a sugar-free base.
Each packet of CYTRA-K CRYSTALS, when reconstituted, provides the following amounts of active ingredients : Potassium Citrate Monohydrate 3300 mg Citric Acid Monohydrate 1002 mg and provides 30 mEq potassium, equivalent to 30 mEq bicarbonate (HCO3). -
INACTIVE INGREDIENTS
Sodium saccharin, fruit punch flavor, and FD&C Red Dye #3.
Potassium Citrate Monohydrate has the chemical name: 1,2,3-Propanetricarboxylic acid, 2-hydroxy-,tripotassium salt, monohydrate. Its chemical structure is as follows:
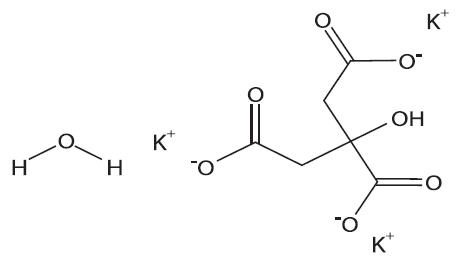
C6H5K3O7 ∙ H2O M.W. 324.41
Citric Acid Monohydrate has the chemical name: 1,2,3-Propanetricarboxylic acid, 2-hydroxy-, monohydrate. Its chemical structure is as follows:
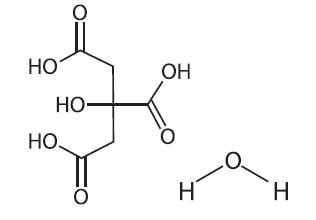
C6H8O7 ∙ H2O M.W. 210.14
-
CLINICAL PHARMACOLOGY
Potassium citrate is absorbed and metabolized to potassium bicarbonate, thus acting as a systemic alkalizer. The effects are essentially those of chlorides before absorption and those of bicarbonates subsequently. Oxidation is virtually complete so that less than 5% of the potassium citrate is excreted in the urine unchanged.
-
INDICATIONS AND USAGE
CYTRA-K CRYSTALS is an effective alkalinizing agent useful in those conditions where long-term maintenance of an alkaline urine is desirable, such as in patients with uric acid and cystine calculi of the urinary tract, especially when the administration of sodium salts is undesirable or contraindicated. In addition, it is a valuable adjuvant when administered with uricosuric agents in gout therapy, since urates tend to crystallize out of an acid urine. It is also effective in correcting the acidosis of certain renal tubular disorders where the administration of potassium citrate may be preferable. CYTRA-K CRYSTALS is highly concentrated, and when administered after meals and before bedtime, allows one to maintain an alkaline urinary pH around the clock, usually without the necessity of a 2 A.M. dose. CYTRA-K CRYSTALS alkalinizes the urine without producing a systemic alkalosis in recommended dosage. It is highly palatable, pleasant tasting, and tolerable, even when administered for long periods. Potassium citrate does not neutralize the gastric juice or disturb digestion.
- CONTRAINDICATIONS
-
WARNINGS AND PRECAUTIONS
There have been several reports, published and unpublished, concerning nonspecific small-bowel lesions consisting of stenosis, with or without ulceration, associated with the administration of enteric-coated thiazides with potassium salts. These lesions may occur with enteric-coated potassium tablets alone or when they are used with nonenteric-coated thiazides, or certain other oral diuretics. These small-bowel lesions have caused obstruction, hemorrhage, and perforation. Surgery was frequently required and deaths have occurred. Based on a large survey of physicians and hospitals, both United States and foreign, the incidence of these lesions is low, and a causal relationship in man has not been definitely established. Available information tends to implicate enteric-coated potassium salts, although lesions of this type also occur spontaneously. Therefore, coated potassium-containing formulations should be administered only when indicated, and should be discontinued immediately if abdominal pain, distention, nausea, vomiting, or gastrointestinal bleeding occur. Large doses may cause hyperkalemia and alkalosis, especially in the presence of renal disease. Concurrent administration of potassium-containing medication, potassium-sparing diuretics, angiotensin-converting enzyme (ACE) inhibitors, or cardiac glycosides may lead to toxicity. Do not exceed recommended dosage. Discontinue use if adverse reactions occur.
Should be used with caution by patients with low urinary output unless under the supervision of a physician. As with all liquids containing a high concentration of potassium, patients should be directed to dilute adequately with water to minimize the possibility of gastrointestinal injury associated with the oral ingestion of concentrated potassium salt preparations; and preferably, to take each dose after meals to avoid saline laxative effect.
-
ADVERSE REACTIONS
CYTRA-K CRYSTALS is generally well tolerated without any unpleasant side effects when given in recommended doses to patients with normal renal function and urinary output. However, as with any alkalinizing agent, caution must be used in certain patients with abnormal renal mechanisms to avoid development of hyperkalemia or alkalosis. Potassium intoxication causes listlessness, weakness, mental confusion, tingling of extremities, and other symptoms associated with a high concentration of potassium in the serum. Periodic determinations of serum electrolytes should be carried out in those patients with renal disease in order to avoid these complications. Hyperkalemia may exhibit the following electrocardiographic abnormalities: Disappearance of the P wave, widening and slurring of QRS complex, changes of the S-T segment, tall peaked T waves, etc.
-
OVERDOSAGE
The administration of oral potassium salts to persons with normal excretory mechanisms for potassium rarely causes serious hyperkalemia. However, if excretory mechanisms are impaired, hyperkalemia can result (see Contraindications and Warnings). Hyperkalemia, when detected, must be treated immediately because lethal levels can be reached in a few hours.
Treatment
Should hyperkalemia occur, treatment measures include the following:
(1) Elimination of foods or medications containing potassium. (2) The intravenous administration of 300 to 500 mL/hr of dextrose solution (10 to 25%), containing 10 units of insulin /20 gm dextrose. (3) The use of exchange resins, hemodialysis, or peritoneal dialysis. In treating hyperkalemia, it should be recalled that in patients who have been stabilized on digitalis, too rapid a lowering of the plasma potassium concentration can produce digitalis toxicity.
-
DOSAGE AND ADMINISTRATION
CYTRA-K CRYSTALS should be taken mixed in cool water or juice according to directions, followed by additional water or juice, if desired. Proper dilution may help prevent gastroin-testinal injury associated with the oral ingestion of concentrated potassium salt preparations.
- HOW SUPPLIED
-
SPL UNCLASSIFIED SECTION
KEEP THIS PRODUCT OUT OF THE REACH OF CHILDREN. IN CASE OF ACCIDENTAL OVERDOSAGE, SEEK PROFESSIONAL ASSISTANCE OR CONTACT A POISON CONTROL CENTER IMMEDIATELY.
Call your doctor for medical advice about side effects. You may report side effects to Cypress Pharmaceutical, Inc. at 1-800-793-2145 or FDA at 1-800-FDA-1088.
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 4.32 g Packet Box
-
INGREDIENTS AND APPEARANCE
CYTRA-K CRYSTALS
potassium citrate and citric acid granule, for solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:60258-005 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Potassium Citrate (UNII: EE90ONI6FF) (Anhydrous Citric Acid - UNII:XF417D3PSL) Potassium Citrate 3.3 g Citric Acid Monohydrate (UNII: 2968PHW8QP) (Anhydrous Citric Acid - UNII:XF417D3PSL) Anhydrous Citric Acid 1.002 g Inactive Ingredients Ingredient Name Strength Saccharin Sodium Anhydrous (UNII: I4807BK602) 33 mg Product Characteristics Color red (FD&C Red Dye #3) Score Shape Size Flavor FRUIT PUNCH Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:60258-005-01 100 in 1 BOX; Type 0: Not a Combination Product 05/01/2003 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 05/01/2003 Labeler - Cypress Pharmaceutical, Inc. (790248942) Registrant - Cypress Pharmaceutical, Inc. (790248942) Establishment Name Address ID/FEI Business Operations Tower Laboratories Ltd 001587203 manufacture(60258-005)


