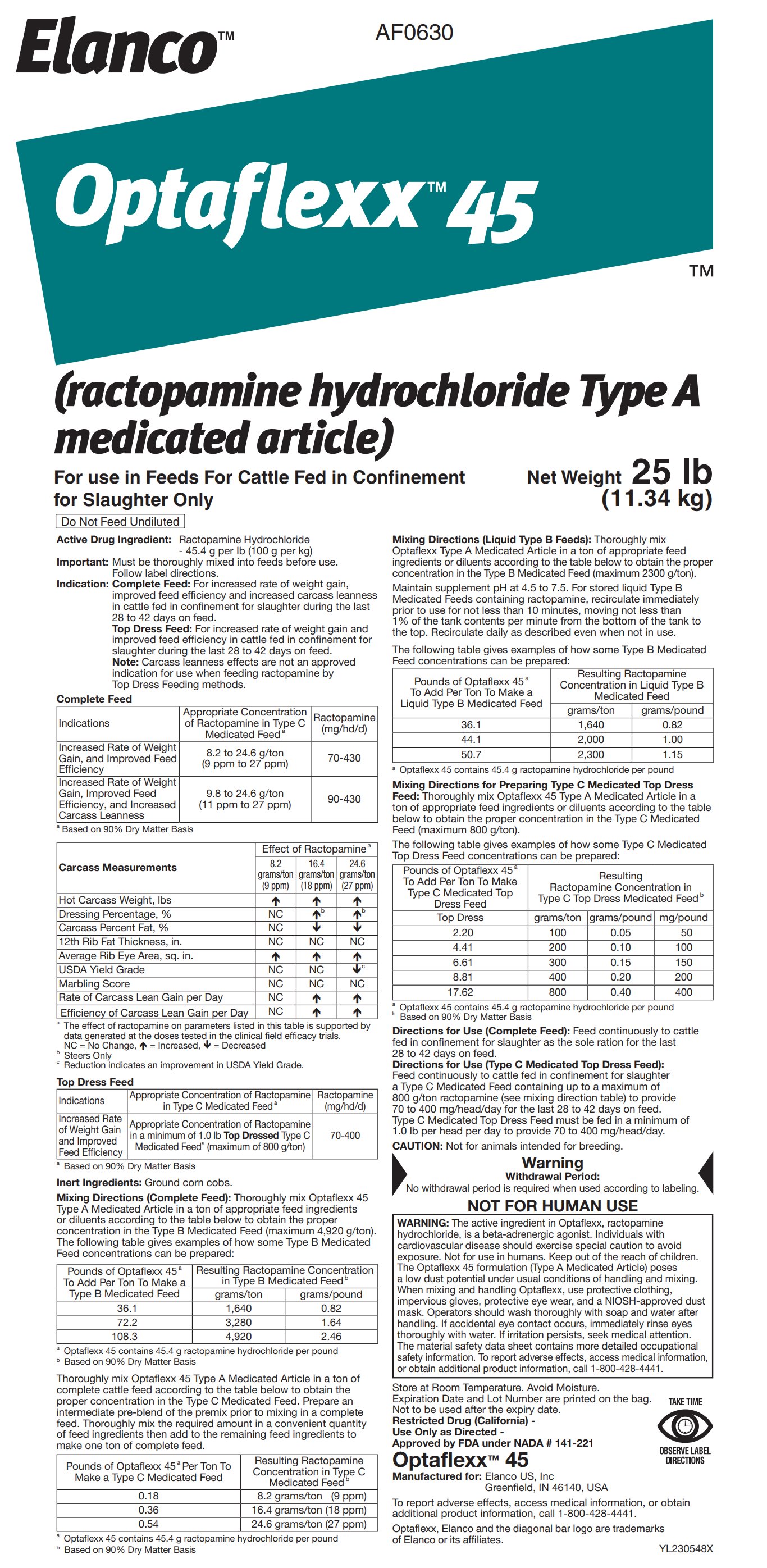
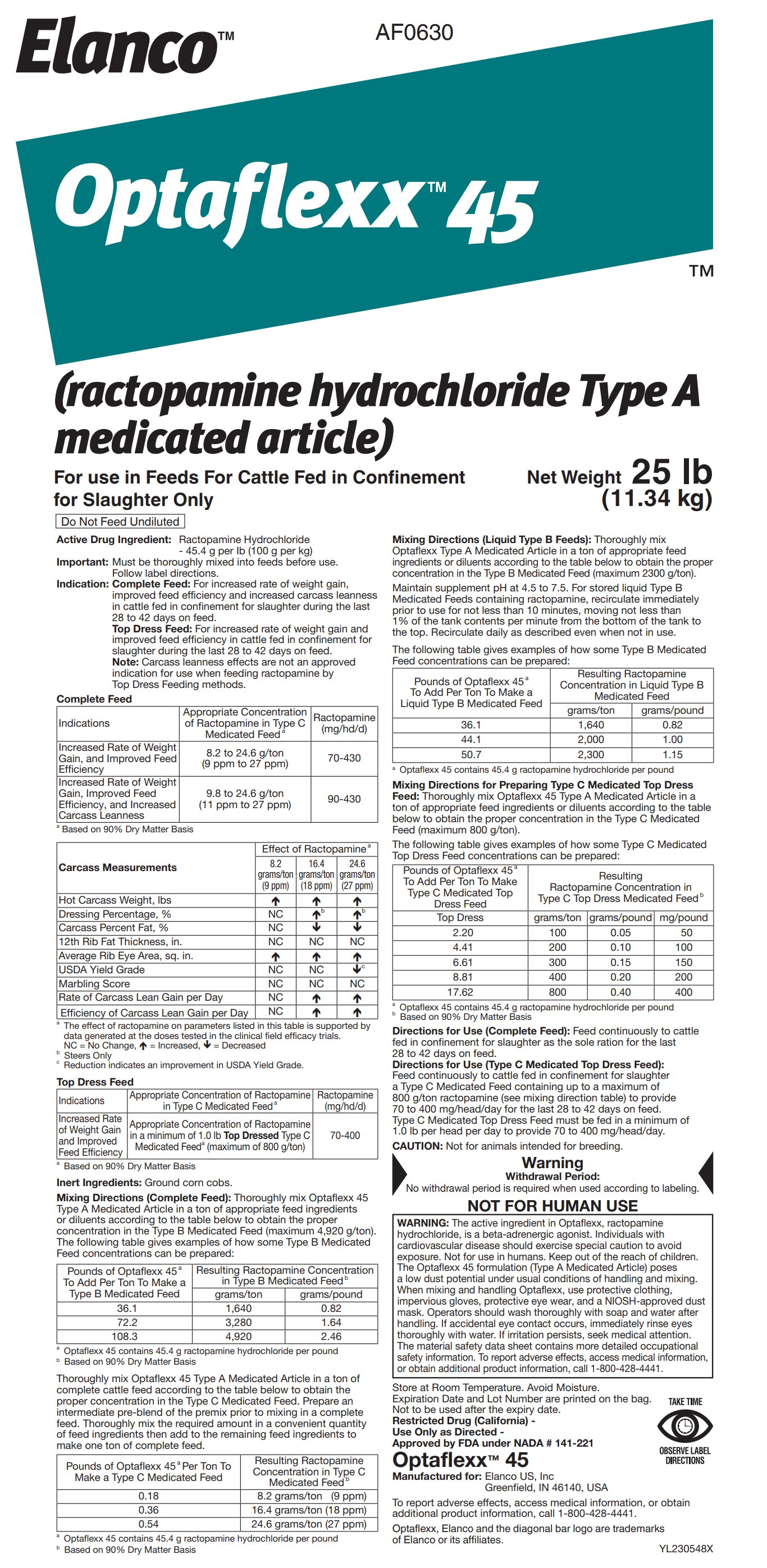
Label: OPTAFLEXX 45- ractopamine hydrochloride powder
- NDC Code(s): 58198-0630-9
- Packager: Elanco US Inc.
- Category: OTC TYPE A MEDICATED ARTICLE ANIMAL DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Animal Drug Application
Drug Label Information
Updated July 11, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active Drug Ingredient:
-
Indication:
Complete Feed: For increased rate of weight gain, improved feed efficiency and increased carcass leanness in cattle fed in confinement for slaughter during the last 28 to 42 days on feed.
Top Dress Feed: For increased rate of weight gain and improved feed efficiency in cattle fed in confinement for slaughter during the last 28 to 42 days on feed.
Note: Carcass leanness effects are not an approved indication for use when feeding ractopamine by Top Dress Feeding methods.
Complete Feed
a Based on 90% Dry Matter Basis Indications
Appropriate Concentration of Ractopamine in Type C Medicated Feed a
Ractopamine
(mg/hd/d)Increased Rate of Weight Gain, and Improved Feed Efficiency
8.2 to 24.6 g/ton
(9 ppm to 27 ppm)70-430
Increased Rate of Weight Gain, Improved Feed Efficiency, and Increased Carcass Leanness
9.8 to 24.6 g/ton
(11 ppm to 27 ppm)90-430
a The effect of ractopamine on parameters listed in this table is supported by data generated at the doses tested in the clinical field efficacy trials. NC = No Change, ↑ = Increased, ↓ = Decreased b Steers Only c Reduction indicates an improvement in USDA Yield Grade. Carcass Measurements
Effect of Ractopaminea
8.2 grams/ton
(9 ppm)16.4 grams/ton
(18 ppm)24.6 grams/ton
(27 ppm)Hot Carcass Weight, lbs
↑
↑
↑
Dressing Percentage, %
NC
↑b
↑b
Carcass Percent Fat, %
NC
↓
↓
12th Rib Fat Thickness, in.
NC
NC
NC
Average Rib Eye Area, sq. in.
↑
↑
↑
USDA Yield Grade
NC
NC
↓c
Marbling Score
NC
NC
NC
Rate of Carcass Lean Gain per Day
NC
↑
↑
Efficiency of Carcass Lean Gain per Day
NC
↑
↑
Top Dress Feed
a Based on 90% Dry Matter Basis Indications
Appropriate Concentration of Ractopamine in Type C Medicated Feed a
Ractopamine
(mg/hd/d)Increased Rate of Weight Gain and Improved Feed Efficiency
Appropriate Concentration of Ractopamine in a minimum of 1.0 lb Top Dressed Type C Medicated Feeda (maximum of 800 g/ton)
70-400
Inert Ingredients: Ground corn cobs.
Mixing Directions (Complete Feed):
Thoroughly mix Optaflexx 45 Type A Medicated Article in a ton of appropriate feed ingredients or diluents according to the table below to obtain the proper concentration in the Type B Medicated Feed (maximum 4,920 g/ton). The following table gives examples of how some Type B Medicated Feed concentrations can be prepared:
a Optaflexx 45 contains 45.4 g ractopamine hydrochloride per pound b Based on 90% Dry Matter Basis Pounds of Optaflexx 45 a To Add Per Ton To Make a
Type B Medicated FeedResulting Ractopamine Concentration in Type B Medicated Feed b
grams/ton
grams/pound
36.1
1,640
0.82
72.2
3,280
1.64
108.3
4,920
2.46
Thoroughly mix Optaflexx 45 Type A Medicated Article in a ton of complete cattle feed according to the table below to obtain the proper concentration in the Type C Medicated Feed. Prepare an intermediate pre-blend of the premix prior to mixing in a complete feed. Thoroughly mix the required amount in a convenient quantity of feed ingredients then add to the remaining feed ingredients to make one ton of complete feed.
a Optaflexx 45 contains 45.4 g ractopamine hydrochloride per pound b Based on 90% Dry Matter Basis Pounds of Optaflexx 45 a Per Ton To Make a Type C Medicated Feed
Resulting Ractopamine Concentration in Type C Medicated Feed b
0.18
8.2 grams/ton (9 ppm)
0.36
16.4 grams/ton (18 ppm)
0.54
24.6 grams/ton (27 ppm)
Mixing Directions (Liquid Type B Feeds):
Thoroughly mix Optaflexx Type A Medicated Article in a ton of appropriate feed ingredients or diluents according to the table below to obtain the proper concentration in the Type B Medicated Feed (maximum 2300 g/ton).
Maintain supplement pH at 4.5 to 7.5. For stored liquid Type B Medicated Feeds containing ractopamine, recirculate immediately prior to use for not less than 10 minutes, moving not less than 1% of the tank contents per minute from the bottom of the tank to the top. Recirculate daily as described even when not in use.
The following table gives examples of how some Type B Medicated Feed concentrations can be prepared:
a Optaflexx 45 contains 45.4 g ractopamine hydrochloride per pound Pounds of Optaflexx 45 a To Add Per Ton To Make a Liquid Type B Medicated Feed
Resulting Ractopamine Concentration in Liquid Type B Medicated Feed
grams/ton
grams/pound
36.1
1,640
0.82
44.1
2,000
1.00
50.7
2,300
1.15
Mixing Directions for Preparing Type C Medicated Top Dress Feed:
Thoroughly mix Optaflexx 45 Type A Medicated Article in a ton of appropriate feed ingredients or diluents according to the table below to obtain the proper concentration in the Type C Medicated Feed (maximum 800 g/ton).
The following table gives examples of how some Type C Medicated Top Dress Feed concentrations can be prepared:
a Optaflexx 45 contains 45.4 g ractopamine hydrochloride per pound b Based on 90% Dry Matter Basis Pounds of Optaflexx 45 a To Add Per Ton To Make Type C Medicated Top Dress Feed
Resulting Ractopamine Concentration in Type C Top Dress Medicated Feed b
Top Dress
grams/ton
grams/pound
mg/pound
2.20
100
0.05
50
4.41
200
0.10
100
6.61
300
0.15
150
8.81
400
0.20
200
17.62
800
0.40
400
- Directions for Use (Complete Feed):
-
Directions for Use (Type C Medicated Top Dress Feed):
Feed continuously to cattle fed in confinement for slaughter a Type C Medicated Feed containing up to a maximum of 800 g/ton ractopamine (see mixing direction table) to provide 70 to 400 mg/head/ day for the last 28 to 42 days on feed. Type C Medicated Top Dress Feed must be fed in a minimum of 1.0 lb per head per day to provide 70 to 400 mg/head/day.
CAUTION: Not for animals intended for breeding.
- Warning
-
BOXED WARNING
(What is this?)
WARNING: The active ingredient in Optaflexx, ractopamine hydrochloride, is a beta-adrenergic agonist. Individuals with cardiovascular disease should exercise special caution to avoid exposure. Not for use in humans. Keep out of the reach of children. The Optaflexx 45 formulation (Type A Medicated Article) poses a low dust potential under usual conditions of handling and mixing. When mixing and handling Optaflexx, use protective clothing, impervious gloves, protective eye wear, and a NIOSH-approved dust mask. Operators should wash thoroughly with soap and water after handling. If accidental eye contact occurs, immediately rinse eyes thoroughly with water. If irritation persists, seek medical attention. The material safety data sheet contains more detailed occupational safety information. To report adverse effects, access medical information, or obtain additional product information, call 1-800-428-4441.
-
STORAGE AND HANDLING
Store at Room Temperature. Avoid Moisture.
Expiration Date and Lot Number are printed on the bag.
Not to be used after the expiry date.
Restricted Drug (California) -
Use Only as Directed -
Approved by FDA under NADA # 141-221
Optaflexx™ 45
Manufactured for: Elanco US, Inc
Greenfield, IN 46140, USA
To report adverse effects, access medical information, or obtain additional product information, call 1-800-428-4441.
Optaflexx, Elanco and the diagonal bar logo are trademarks of Elanco or its affiliates.
BG230548X
- Principal Display Panel – 25 lb Bag Label
-
INGREDIENTS AND APPEARANCE
OPTAFLEXX 45
ractopamine hydrochloride powderProduct Information Product Type OTC TYPE A MEDICATED ARTICLE ANIMAL DRUG Item Code (Source) NDC:58198-0630 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ractopamine hydrochloride (UNII: 309G9J93TP) (ractopamine - UNII:57370OZ3P1) ractopamine hydrochloride 45.4 g in 0.45 kg Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:58198-0630-9 11.34 kg in 1 BAG Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NADA NADA141221 06/03/2010 Labeler - Elanco US Inc. (966985624) Establishment Name Address ID/FEI Business Operations Evonik Corporation 130890994 API MANUFACTURE Establishment Name Address ID/FEI Business Operations TriRx Speke Limited 228138655 MANUFACTURE, PACK, LABEL