Label: PREPARATION H SOOTHING RELIEF CLEANSING AND COOLING WIPES- witch hazel cloth
- NDC Code(s): 0573-0553-60, 0573-0553-95
- Packager: Haleon US Holdings LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated March 2, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredient
- Purpose
- Uses
- Warnings
-
Directions
- adults: unfold wipe and cleanse the area by gently wiping, patting or blotting. If necessary, repeat until all matter is removed from the area.
- use up to 6 times daily or after each bowel movement and before applying topical hemorrhoidal treatments, and then discard
- children under 12 years of age: consult a doctor
- Other information
- Inactive ingredients
- Questions or comments?
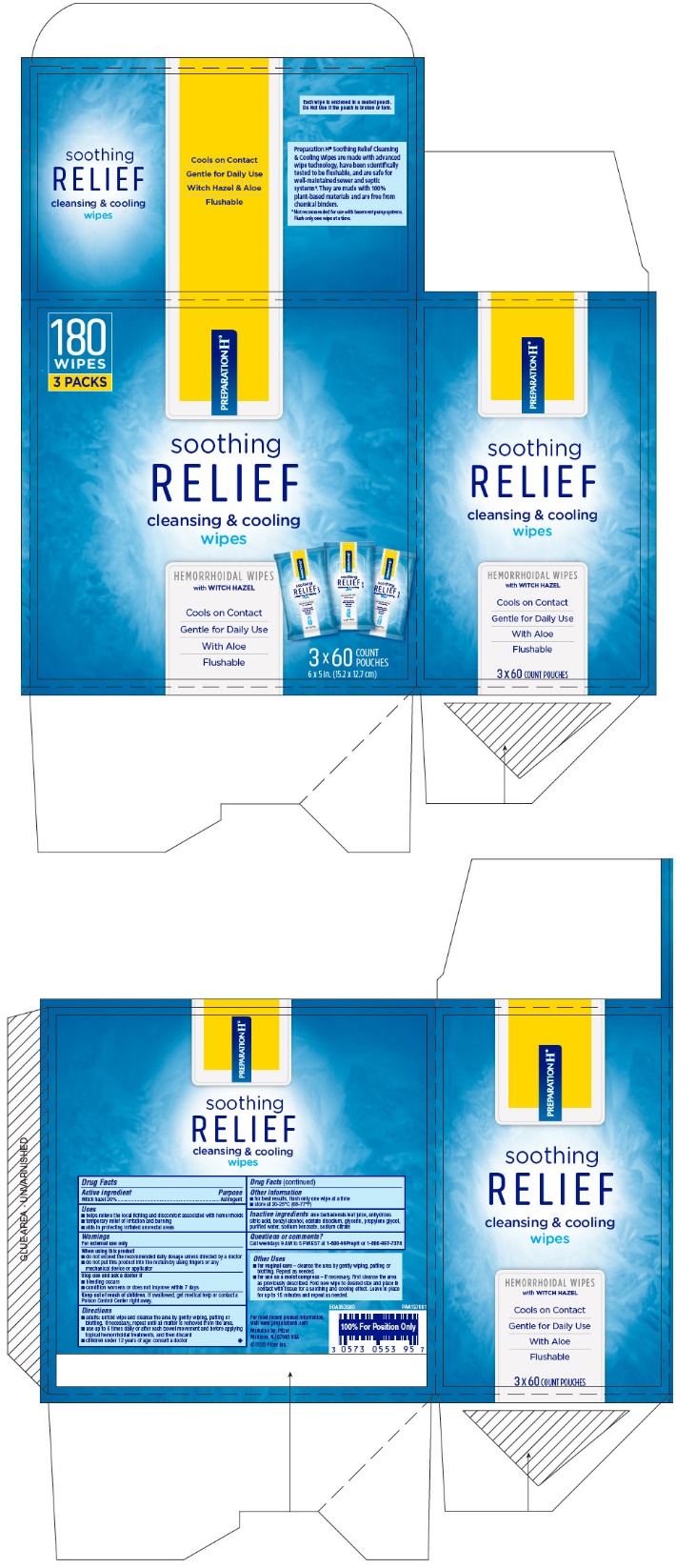
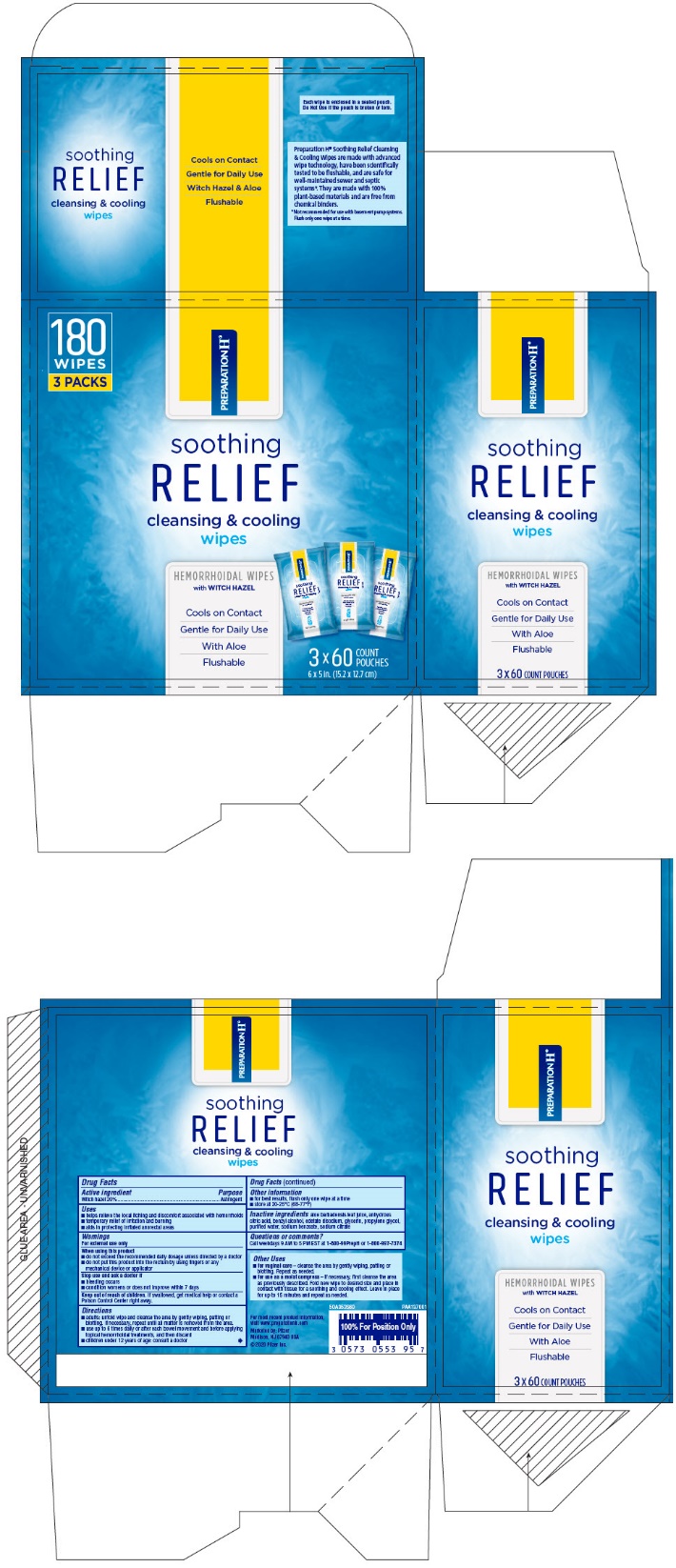
- PRINCIPAL DISPLAY PANEL - 60 Wipe Pouch Carton
- PRINCIPAL DISPLAY PANEL - 60 Wipe Pouch
-
INGREDIENTS AND APPEARANCE
PREPARATION H SOOTHING RELIEF CLEANSING AND COOLING WIPES
witch hazel clothProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:0573-0553 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength WITCH HAZEL (UNII: 101I4J0U34) (WITCH HAZEL - UNII:101I4J0U34) WITCH HAZEL 200 mg Inactive Ingredients Ingredient Name Strength ALOE VERA LEAF (UNII: ZY81Z83H0X) ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) GLYCERIN (UNII: PDC6A3C0OX) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SODIUM CITRATE, UNSPECIFIED FORM (UNII: 1Q73Q2JULR) WATER (UNII: 059QF0KO0R) BENZYL ALCOHOL (UNII: LKG8494WBH) EDETATE DISODIUM (UNII: 7FLD91C86K) SODIUM BENZOATE (UNII: OJ245FE5EU) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0573-0553-95 3 in 1 CARTON 03/04/2020 1 60 in 1 POUCH; Type 0: Not a Combination Product 2 NDC:0573-0553-60 60 in 1 POUCH; Type 0: Not a Combination Product 03/04/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M015 03/04/2020 Labeler - Haleon US Holdings LLC (079944263)