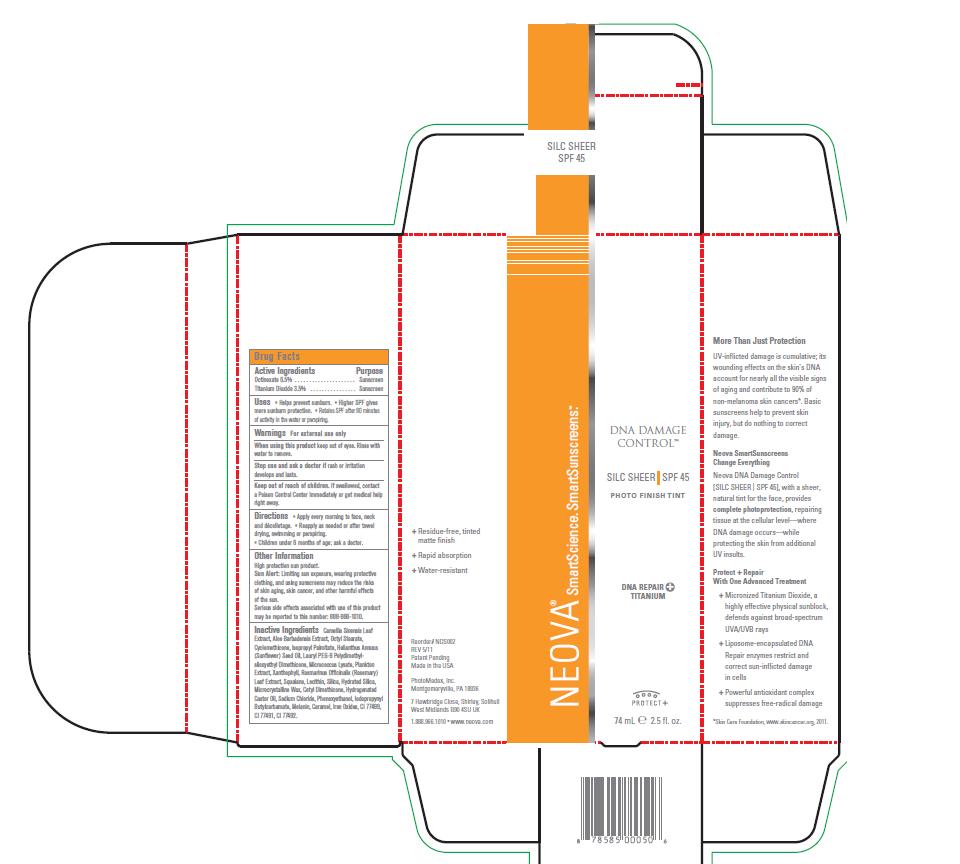
Label: NEOVA DNA DAMAGE CONTROL - SILC SHEER SPF 45- octinoxate, titanium dioxide emulsion
-
Contains inactivated NDC Code(s)
NDC Code(s): 62362-149-01, 62362-149-74 - Packager: PhotoMedex, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated January 6, 2012
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredients
- Purpose
- Uses
- Warnings
- When using this product:
- Stop use and ask a doctor if:
- Keep out of reach of children:
- Directions
-
Other Information
High protection sun product. Sun Alert: Limiting sun exposure, wearing protective clothing, and using sunscreens may reduce the risks of skin aging, skin cancer, and other harmful effects of the sun. Serious side effects associated with use of this product may be reported to this number: 888-966-1010.
-
Inactive Ingredients
Camellia Sinensis Leaf
Extract, Aloe Barbadensis Extract, Octyl Stearate,
Cyclomethicone, Isopropyl Palmitate, Helianthus Annuus
(Sunflower) Seed Oil, Lauryl PEG-9 Polydimethylsiloxyethyl
Dimethicone, Micrococcus Lysate, Plankton
Extract, Xanthophyll, Rosmarinus Officinalis (Rosemary)
Leaf Extract, Squalane, Lecithin, Silica, Hydrated Silica,
Microcrystalline Wax, Cetyl Dimethicone, Hydrogenated
Castor Oil, Sodium Chloride, Phenoxyethanol, Iodopropynyl
Butylcarbamate, Melanin, Caramel, Iron Oxides, CI 77499,
CI 77491, CI 77492. - Image of box and label
-
INGREDIENTS AND APPEARANCE
NEOVA DNA DAMAGE CONTROL - SILC SHEER SPF 45
octinoxate, titanium dioxide emulsionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:62362-149 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Octinoxate (UNII: 4Y5P7MUD51) (Octinoxate - UNII:4Y5P7MUD51) Octinoxate 6.5 mL in 100 mL Titanium Dioxide (UNII: 15FIX9V2JP) (Titanium Dioxide - UNII:15FIX9V2JP) Titanium Dioxide 3.5 mL in 100 mL Inactive Ingredients Ingredient Name Strength Green Tea Leaf (UNII: W2ZU1RY8B0) Aloe Vera Leaf (UNII: ZY81Z83H0X) Octyl Stearate (UNII: 772Y4UFC8B) Cyclomethicone (UNII: NMQ347994Z) Isopropyl Palmitate (UNII: 8CRQ2TH63M) Sunflower Oil (UNII: 3W1JG795YI) Lutein (UNII: X72A60C9MT) Rosemary (UNII: IJ67X351P9) Squalane (UNII: GW89575KF9) Egg Phospholipids (UNII: 1Z74184RGV) Silicon Dioxide (UNII: ETJ7Z6XBU4) Hydrated Silica (UNII: Y6O7T4G8P9) Microcrystalline Wax (UNII: XOF597Q3KY) Hydrogenated Castor Oil (UNII: ZF94AP8MEY) Sodium Chloride (UNII: 451W47IQ8X) Phenoxyethanol (UNII: HIE492ZZ3T) Iodopropynyl Butylcarbamate (UNII: 603P14DHEB) Caramel (UNII: T9D99G2B1R) Ferrosoferric Oxide (UNII: XM0M87F357) Ferric Oxide Red (UNII: 1K09F3G675) Ferric Oxide Yellow (UNII: EX438O2MRT) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:62362-149-01 1 in 1 BOX 1 NDC:62362-149-74 74 mL in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 01/06/2012 Labeler - PhotoMedex, Inc. (054503875) Establishment Name Address ID/FEI Business Operations PhotoMedex, Inc. 054503875 manufacture


 DNADControlSHEERLabel.jpg
DNADControlSHEERLabel.jpg