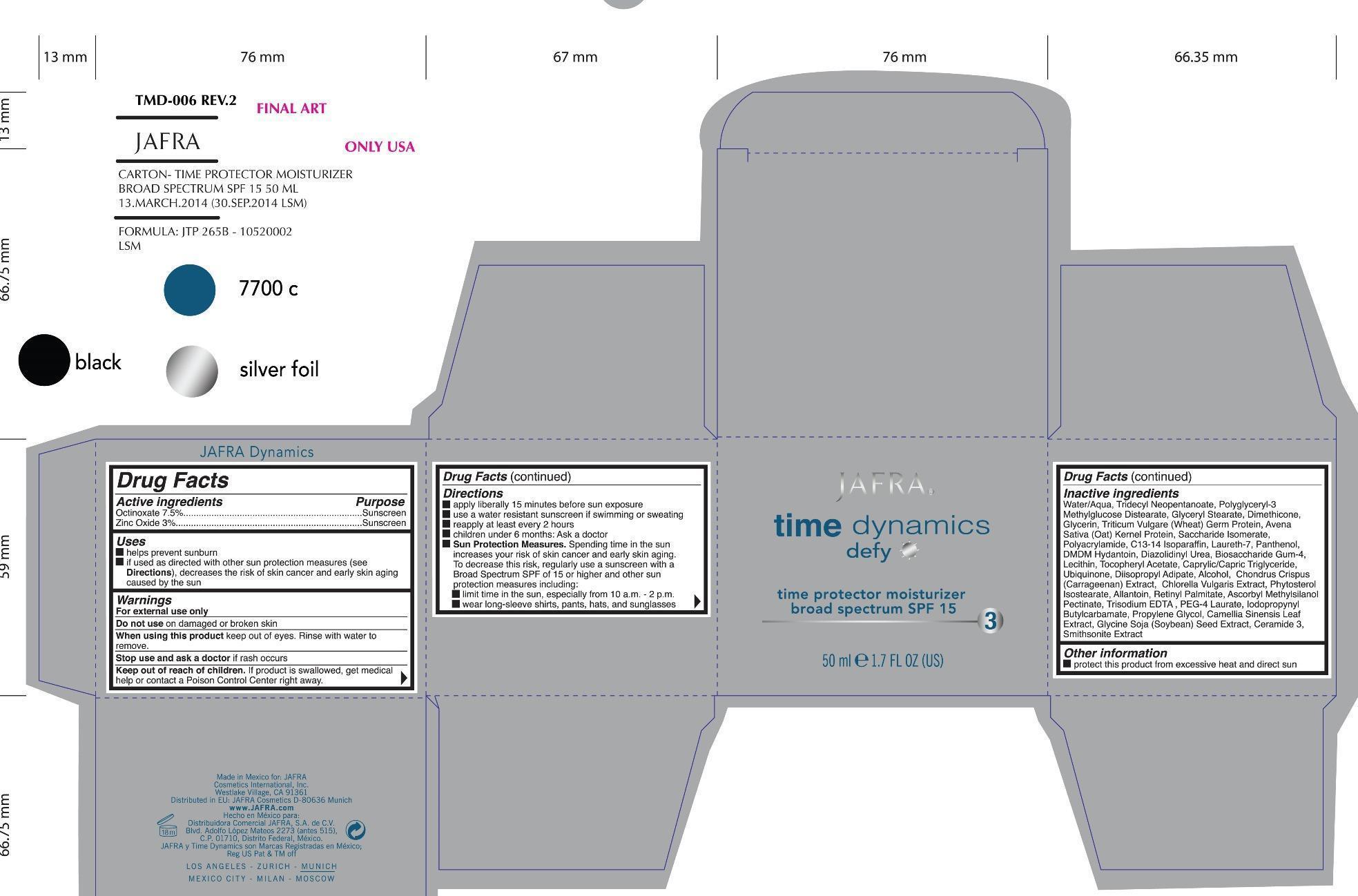
Label: DEFY TIME PROTECTOR MOISTURIZER BROAD SPECTRUM SPF 15 JAFRA TIME DYNAMICS- octinoxate, zinc oxide cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 68828-201-01, 68828-201-02 - Packager: Jafra Cosmetics International
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated May 21, 2019
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- KEEP OUT OF REACH OF CHILDREN
- INDICATIONS & USAGE
- WARNINGS
-
DOSAGE & ADMINISTRATION
Directions
- apply liberally 15 minutes before sun exposure
- use a water resistant sunscreen if swimming or sweating
- reapply at least every 2 hours
- children under 6 months: Ask a doctor
- Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum of SPF 15 or higher and other sun protection measures including:
limit time in the sun, especially from 10a.m - 2p.m.
wear long-sleeve shirts, pants, hats, and sunglasses
-
INACTIVE INGREDIENT
Inactive Ingredients
Water/Aqua, Tridecyl Neopentanoate, Polyglyceryl-3 Methylglucose Distearate, Glyceryl Stearate, Dimethicone, Glycerin, Triticum Vulgare (Wheat) Germ Protein, Avena Sativa (Oat) Kernel Protein,Saccharide lsomerate, Polyacrylamide, C13-141soparaffin, Laureth-7, Panthenol, DMDM Hydantoin, Diazolidinyl Urea, Biosaccharide Gum-4, Lecithin,Tocopheryl Acetate, Caprylic/Capric Triglylceride, Ubiquinone, Diisopropyl Adipate, Alcohol, Chondrus Crispus (Carrageenan) Extract, Chlorella Vulgaris Extract, Phytosterol lsostearate, Allantoin, Retinyl Palmitate, Ascorbyl Methylsilanol Pectinate, Trisodium EDTA, PEG-4 Laurate, lodopropynyl Bulylcarbamate, Propylene Glycol, Camellia Sinensis Leal Extract, Glycine Soja (Soybean) Seed Extract, Ceramide 3, Smithsonite Extract
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
DEFY TIME PROTECTOR MOISTURIZER BROAD SPECTRUM SPF 15 JAFRA TIME DYNAMICS
octinoxate, zinc oxide creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:68828-201 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 7.5 g in 100 mL ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 3 g in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) TRIDECYL NEOPENTANOATE (UNII: 3Z8H1DA7J5) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) DIMETHICONE (UNII: 92RU3N3Y1O) GLYCERIN (UNII: PDC6A3C0OX) WHEAT GERM (UNII: YR3G369F5A) SACCHARIDE ISOMERATE (UNII: W8K377W98I) POLYACRYLAMIDE (1500 MW) (UNII: 5D6TC4BRWV) C13-14 ISOPARAFFIN (UNII: E4F12ROE70) LAURETH-7 (UNII: Z95S6G8201) PANTHENOL (UNII: WV9CM0O67Z) DMDM HYDANTOIN (UNII: BYR0546TOW) DIAZOLIDINYL UREA (UNII: H5RIZ3MPW4) BIOSACCHARIDE GUM-4 (UNII: 9XRL057X90) LECITHIN, SOYBEAN (UNII: 1DI56QDM62) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) UBIDECARENONE (UNII: EJ27X76M46) DIISOPROPYL ADIPATE (UNII: P7E6YFV72X) ALCOHOL (UNII: 3K9958V90M) CHONDRUS CRISPUS CARRAGEENAN (UNII: UE856F2T78) CHLORELLA VULGARIS (UNII: RYQ4R60M02) ALLANTOIN (UNII: 344S277G0Z) VITAMIN A PALMITATE (UNII: 1D1K0N0VVC) ASCORBYL METHYLSILANOL PECTINATE (UNII: M3PF9237YC) EDETATE TRISODIUM (UNII: 420IP921MB) PEG-4 LAURATE (UNII: AYF4VM3N1Z) IODOPROPYNYL BUTYLCARBAMATE (UNII: 603P14DHEB) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) GREEN TEA LEAF (UNII: W2ZU1RY8B0) SOYBEAN (UNII: L7HT8F1ZOD) CERAMIDE 3 (UNII: 4370DF050B) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68828-201-02 1 in 1 CARTON 11/25/2014 1 NDC:68828-201-01 50 mL in 1 JAR; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 11/25/2014 Labeler - Jafra Cosmetics International (041676479) Registrant - Jafra Cosmetics International (041676479) Establishment Name Address ID/FEI Business Operations Jafra Manufacturing S.A. de C.V. 814732061 manufacture(68828-201)