Label: PRASCEND- pergolide tablet
- NDC Code(s): 0010-4489-01, 0010-4489-02
- Packager: Boehringer Ingelheim Animal Health USA Inc.
- Category: PRESCRIPTION ANIMAL DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Animal Drug Application
Drug Label Information
Updated October 4, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Caution:
-
Description:
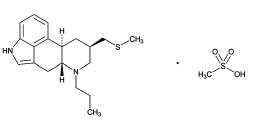
PRASCEND Tablets are rectangular light red colored, half-scored tablets containing 1 mg pergolide, as pergolide mesylate. Pergolide mesylate is a synthetic ergot derivative and is a potent dopamine receptor agonist. The chemical name of pergolide mesylate is 8β-[(Methylthio) methyl]-6-propylergoline monomethanesulfonate.
The chemical structure is:
- Indication:
-
Dosage and Administration:
Administer orally at a starting dose of 2 mcg/kg once daily. Dosage may be adjusted to effect, not to exceed 4 mcg/kg daily.
It has been reported that pergolide tablets may cause eye irritation, an irritating smell, or headache when PRASCEND Tablets are split or crushed. PRASCEND Tablets should not be crushed due to the potential for increased human exposure and care should be taken to minimize exposure when splitting tablets.
The tablets are scored and the calculated dosage should be provided to the nearest one-half tablet increment (see Table 1).
Table 1 Dosing Table
Dosage
Body weight
2 mcg/kg
4 mcg/kg
136 - 340 kg
(300 - 749 lb)
0.5 tablet
1 tablet
341 - 567 kg
(750 - 1,249 lb)
1 tablet
2 tablets
568 - 795 kg
(1,250 - 1,749 lb)
1.5 tablets
3 tablets
796 - 1,022 kg
(1,750 - 2,249 lb)
2 tablets
4 tablets
Dosing should be titrated according to individual response to therapy to achieve the lowest effective dose. Dose titration is based on improvement in clinical signs associated with Pituitary Pars Intermedia Dysfunction (PPID) and/or improvement or normalization of endocrine tests (for example, dexamethasone suppression test or endogenous ACTH test).
In some cases, adverse events were reported after a dose increase (see Post-Approval Experience).
If signs of dose intolerance develop, the dose should be decreased by half for 3 to 5 days and then titrated back up in 2 mcg/kg increments every 2 weeks until the desired effect is achieved.
- Contraindications:
-
Warnings:
Do not use in horses intended for human consumption.
Keep PRASCEND in a secure location out of reach of dogs, cats, and other animals to prevent accidental ingestion or overdose.
Dogs have eaten PRASCEND tablets that were placed in food intended for horses or dropped during administration of the tablets to the horses. Adverse reactions may occur if animals other than horses ingest PRASCEND tablets (see Post-Approval Experience).
-
Human Warnings:
Not for use in humans. Do not ingest the product. Keep this and all medications out of the reach of children. PRASCEND should not be administered by persons who have had adverse reactions to ergotamine or other ergot derivatives.
Pergolide, like other ergot derivatives, may cause emesis, dizziness, lethargy or low blood pressure.
Pregnant or lactating women should wear gloves when administering this product. It has been reported that pergolide tablets may cause eye irritation, an irritating smell, or headache when PRASCEND Tablets are split or crushed. PRASCEND Tablets should not be crushed due to the potential for increased human exposure and care should be taken to minimize exposure when splitting tablets. Store this product separately away from human medicinal products and handle this product with care to avoid accidental ingestion.
In case of accidental ingestion seek medical advice immediately and show the package leaflet or the label to the physician.
-
Precautions:
Treatment with PRASCEND may cause inappetence.
The use of PRASCEND in breeding, pregnant, or lactating horses has not been evaluated. The effects of pergolide mesylate on breeding, pregnant, or lactating horses are not known; however, the pharmacologic action of pergolide mesylate suggests that it may interfere with reproductive functions such as lactation. PRASCEND is approximately 90% associated with plasma proteins. Use caution if administering PRASCEND with other drugs that affect protein binding. Dopamine antagonists, such as neuroleptics (phenothiazines, domperidone) or metoclopramide, ordinarily should not be administered concurrently with PRASCEND (a dopamine agonist) since these agents may diminish the effectiveness of PRASCEND.
-
Adverse Reactions:
Pre-Approval Experience:
A total of 122 horses treated with PRASCEND Tablets for six months were included in a field study safety analysis.
Table 2 Summary of the most common adverse reactions (N=122) Clinical sign
# Cases
Cases (%)
Decreased appetite
40
32.8
Lameness
22
18.0
Diarrhea/Loose stool
12
9.8
Colic
12
9.8
Lethargy
12
9.8
Abnormal Weight Loss
11
9.0
Laminitis*
10
8.2
Heart murmur
10
8.2
Death
8
6.6
Tooth disorder
8
6.6
Skin abscess
7
5.7
Musculoskeletal pain
6
4.9
Behavior change
6
4.9
*Three new cases and 7 pre-existing, recurring cases
Inappetence or decreased appetite occurred at one or more meals in 40 of 122 horses treated with PRASCEND. At the baseline evaluation 1.6% of owners reported a history of inappetence or decreased appetite as compared to the 32.8% of horses that experienced inappetence or decreased appetite during the study. Most cases of inappetence were transient and occurred during the first month of treatment; however, some horses experienced sporadic inappetence throughout the study. Two horses required a temporary reduction in dose due to inappetence during the first month of the study. Both horses returned to their original dose within 30 days.
Weight loss occurred in more than half of the horses in this study; however, weight loss that was considered abnormal was only reported in 11 horses.
Lethargy was reported in 9.8% of horses during the study, and was not reported in any horses at the baseline evaluation.
Behavioral changes were noted in 6 horses including aggression, kicking, agitation, nervous behavior and increased activity. One horse required a temporary reduction in dose due to energetic behavior during the first month of the study.
Eight horses died or were euthanized during the study due to worsening of pre-existing conditions (laminitis, dental disease, septic tenosynovitis) or colic (strangulating lipomas, large colon volvulus).
One mare was inadvertently enrolled in the study while pregnant and experienced dystocia resulting in the death of the foal.
Post-Approval Experience (2019):
The following adverse events are based on post approval adverse drug experience reporting for PRASCEND. Not all adverse events are reported. It is not always possible to reliably estimate the adverse event frequency or establish a causal relationship to product exposure using these data.
The following adverse events in horses are categorized in order of decreasing reporting frequency by body system and in decreasing order of reporting frequency within each body system:
General: anorexia, lethargy, weight loss
Gastrointestinal: diarrhea, abdominal pain/colic
Dermatological: alopecia, hyperhidrosis, dermatitis
Musculoskeletal: laminitis, muscle stiffness/soreness
Neurological: ataxia, seizure, muscle tremors
Behavioral: aggression (to other horses and humans), hyperactivity (anxiety, agitation), other behavioral changes (stud-like behavior, spooky, unpredictable, confused)
Clinical pathology: anemia, elevated liver enzymes, thrombocytopenia
The above adverse events were reported in some horses at starting dose levels, while in the others following a dose increase.
Death (including euthanasia) has been reported.
Adverse events have been reported in dogs following ingestion of tablets prepared for administration to horses.
To report suspected adverse reactions, to obtain a Safety Data Sheet (SDS), or for technical assistance, contact Boehringer Ingelheim Animal Health USA Inc. at 1-888-637-4251. For additional information about adverse drug experience reporting for animal drugs, contact the FDA at 1-888-FDA-VETS or online at http://www.fda.gov/reportanimalae.
-
Clinical Pharmacology:
Pergolide mesylate is a synthetic ergot derivative and is a potent dopamine receptor agonist. As with other dopamine agonists, pergolide inhibits the release of prolactin which suggests that it may interfere with lactation. In horses with PPID, pergolide is believed to exert its therapeutic effect by stimulating dopamine receptors, and has been shown to decrease the plasma levels of adrenocorticotropic hormone (ACTH), melanocyte stimulating hormone (MSH), and other pro-opiomelanocortin peptides. 1
Pharmacokinetic information in the horse is based on a study using single oral doses of 10 mcg/kg in six healthy mares between 3 and 17 years of age. 2 Pergolide was rapidly absorbed; the mean maximum concentration (Cmax) was 4.05±2.02 ng/mL with the median time to maximum concentration (Tmax) being 0.415 hours.
The area under the curve (AUC) was 14.08±7.46 hr∙ng/mL. The mean half life (T1/2) was 5.86±3.42 hours; the mean apparent oral clearance (CL/F) was 1204 mL/kg/hr; and the mean apparent volume of distribution (V/F) was 3082±1354 mL/kg.
-
Effectiveness:
An open-label, historical control, field study evaluated the effectiveness of PRASCEND for the control of clinical signs of PPID. A total of 122 horses with PPID were enrolled in the study, 113 of which were included in effectiveness evaluations. The success of each horse was based on results of endocrinology testing (dexamethasone suppression test or endogenous ACTH test) and/or improvement in clinical signs related to PPID (hirsutism, hyperhidrosis, polyuria/polydypsia, abnormal fat distribution, and/or muscle-wasting) on the Day 180 evaluation. Based on endocrine testing and investigators’ clinical assessment scores, 86 (76.1%) of the 113 evaluable cases were treatment successes.
Table 3 Proportion of Treatment Successes on Day 180
Percent success
Lower bound: one-sided 95% confidence interval
76.1% (86/113)
68.6%
Enrolled horses were diagnosed with PPID based on the presence of hirsutism and an abnormal pre-study endocrine test result. All horses were treated with 2 mcg/kg PRASCEND (to the nearest one-half tablet) orally once daily for the first three months. If the endocrine test result on Day 90 was normal or adequately improved, the horse continued on the same dose through Day 180. If the endocrine test result on Day 90 was abnormal, the dose increased to 4 mcg/kg given once daily through Day 180. Forty-seven (41.6%) of the 113 horses included in the effectiveness database required a dose increase at Day 90.
Improvement was noted in scores for all clinical sign categories and in mean results for endocrine tests.
Table 4 Percent of Animals with Improvement in Clinical Signs Relative to Baseline Scores Clinical sign
Day 90±7 (%)
Day 180±7 (%)
Hirsutism
32.7%
89.2%
Hyperhidrosis
27.4%
42.3%
Polyuria / polydypsia
31.0%
34.2%
Abnormal fat distribution
21.2%
33.3%
Muscle wasting
36.3%
46.0%
Table 5 Endocrine test results (mean values) Test
# Animals
Baseline
Day 90
Day 180
ACTH
(pg/mL)
20
73.53
51.12
45.08
DST**
(mcg/dL)
93
3.12
1.39
1.47
** Dexamethasone suppression test: Post dexamethasone cortisol concentration
-
Animal Safety:
In a six month target animal safety study healthy adult horses received PRASCEND administered orally, once daily, at doses of either 0 mcg/kg, 4 mcg/kg, 6 mcg/kg, or 8 mcg/kg (0X, 1X, 1.5X, or 2X the maximum recommended dose). There were eight healthy horses (four males and four females) in each treatment group. Doses were prepared by dissolving tablets in approximately 10 mL of a 50% sugar water solution.
PRASCEND treated groups had lower mean heart rates and higher mean temperatures than the control group. Horses in all treatment groups had minimum heart rates within the normal range and maximum temperatures below 101.5°F. One 1.5X horse experienced a mild episode of spasmodic colic on Day 3 that resolved after treatment with flunixin meglumine.
Mean red blood cell counts and hemoglobin values were lower in PRASCEND treated groups as compared to the control group. Other hematology parameters including hematocrit, white blood cells, absolute neutrophils, and absolute lymphocytes exhibited mild, transient decreases as compared to the control group. The hematology parameters generally decreased over the first 30 to 60 days after treatment initiation and then returned to values similar to pre-treatment levels. No treatment related alterations were identified on histopathology evaluation of bone marrow.
- Storage:
- How Supplied:
-
References:
1 Orth, D.N., Holscher, M.A., Wilson, M.G., et al. (1982) Equine Cushing’s Disease: Plasma Immunoreactive Proopiolipomelanocortin Peptide and Cortisol Levels Basally and in Response to Diagnostic Tests. Endocrinology. 110(4):1430-41
2 Wright A, Gehring R, Coetzee H (2008.) Pharmacokinetics of pergolide in normal mares. American College of Veterinary Internal Medicine Forum, Abstract #36, San Antonio, TX.
- SPL UNCLASSIFIED SECTION
- Principal Display Panel 10 – Tablet Blister Pack
-
Principal Display Panel - 60 – Tablet Display Carton
NDC 0010-4489-01
Prascend®
(pergolide tablets)
1 mg
Caution: Federal law restricts this drug to use by or on the order of a licensed veterinarian.
Dopamine receptor agonist for oral use in horses only
Net Contents: 60 tablets in blister packs containing 10 tablets each
Approved by FDA under NADA # 141-331
-
INGREDIENTS AND APPEARANCE
PRASCEND
pergolide tabletProduct Information Product Type PRESCRIPTION ANIMAL DRUG Item Code (Source) NDC:0010-4489 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PERGOLIDE MESYLATE (UNII: 55B9HQY616) (PERGOLIDE - UNII:24MJ822NZ9) PERGOLIDE 1 mg Product Characteristics Color RED (Light red) Score 2 pieces Shape RECTANGLE Size 12mm Flavor Imprint Code PRD Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0010-4489-01 6 in 1 CARTON 1 10 in 1 BLISTER PACK 2 NDC:0010-4489-02 16 in 1 CARTON 2 10 in 1 BLISTER PACK Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NADA NADA141331 10/25/2011 Labeler - Boehringer Ingelheim Animal Health USA Inc. (007134091)