Label: VETERINARY LACTATED RINGERS- sodium chloride, sodium lactate, potassium chloride, calcium chloride injection, solution
- NDC Code(s): 0338-0092-02
- Packager: Baxter Healthcare Corporation
- Category: PRESCRIPTION ANIMAL DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated January 1, 2019
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
DESCRIPTION
Lactated Ringer’s Injection, USP is a sterile, nonpyrogenic solution for fluid and electrolyte replenishment in single dose containers for
intravenous administration. It contains no antimicrobial agents. Discard unused portion. Composition, osmolarity, pH, ionic concentration and caloric content are shown in Table 1.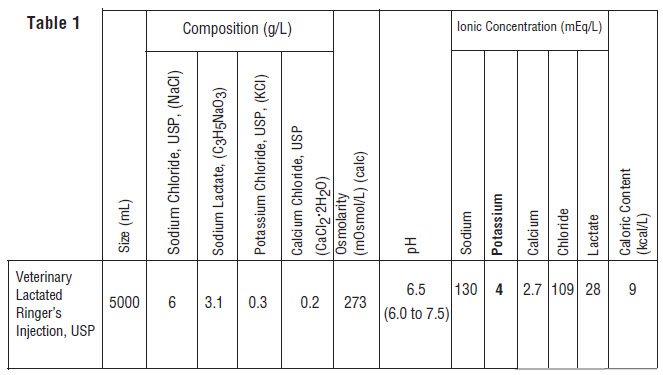
The plastic container is fabricated from a specially formulated polyvinyl chloride. The amount of water that can permeate from inside the container into the overwrap is insufficient to affect the solution significantly. Solutions in contact with the plastic container can leach out certain of its chemical components in very small amounts within the expiration period, e.g., di-2-ethylhexyl phthalate (DEHP), up to 5 parts per million. However, the safety of the plastic has been confirmed in tests in animals according to USP biological tests for plastic containers as well as by tissue culture toxicity studies.
-
CLINICAL PHARMACOLOGY
Lactated Ringer’s Injection, USP has value as a source of water and
electrolytes. It is capable of inducing diuresis depending on the clinical condition of the patient.Lactated Ringer’s Injection, USP produces a metabolic alkalinizing effect. Lactate ions are metabolized ultimately to carbon dioxide and water, which requires the consumption of hydrogen cations.
- INDICATIONS & USAGE
- CONTRAINDICATIONS
-
WARNINGS
Lactated Ringer’s Injection, USP should be used with great care, if at all, in patients with congestive heart failure, severe renal insufficiency, and in clinical states in which there exists edema with sodium retention.
Lactated Ringer’s Injection, USP should be used with great care, if at all, in patients with hyperkalemia, severe renal failure, and in conditions in which potassium retention is present.
Lactated Ringer’s Injection, USP should be used with great care in patients with metabolic or respiratory alkalosis. The administration of lactate ions should be done with great care in those conditions in which there is an increased level or an impaired utilization of these ions, such as severe hepatic insufficiency.Lactated Ringer’s Injection, USP should not be administered simultaneously with blood through the same administration set because of the likelihood of coagulation.
The intravenous administration of Lactated Ringer’s Injection, USP can cause fluid and/or solute overloading resulting in dilution of serum
electrolyte concentrations, overhydration, congested states, or pulmonary edema. The risk of dilutional states is inversely proportional to the electrolyte concentrations of the injections. The risk of solute overload causing congested states with peripheral and pulmonary edema is directly proportional to the electrolyte concentrations of the injections.
In patients with diminished renal function, administration of Lactated Ringer’s Injection, USP may result in sodium or potassium retention.
Lactated Ringer’s Injection, USP is not for use in the treatment of lactic acidosis. -
ADVERSE REACTIONS
Reactions which may occur because of the solution or the technique of administration include febrile response, infection at the site of injection, venous thrombosis or phlebitis extending from the site of injection, extravasation, and hypervolemia. If an adverse reaction does occur, discontinue the infusion, evaluate the patient, institute appropriate therapeutic countermeasures, and save the remainder of the fluid for examination if deemed necessary.
-
PRECAUTIONS
Clinical evaluation and periodic laboratory determinations are necessary to monitor changes in fluid balance, electrolyte concentrations, and acid base balance during prolonged parenteral therapy or whenever the condition of the patient warrants such evaluation.
Lactated Ringer’s Injection, USP must be used with caution. Excess administration may result in metabolic alkalosis.
Caution must be exercised in the administration of Lactated Ringer’s Injection, USP to patients receiving corticosteroids or corticotropin.
Do not administer unless solution is clear and seal is intact.
Reactions which may occur because of the solution or the technique of administration include febrile response, infection at the site of injection, venous thrombosis or phlebitis extending from the site of injection, extravasation, and hypervolemia.
If an adverse reaction does occur, discontinue the infusion, evaluate the patient, institute appropriate therapeutic countermeasures, and save the remainder of the fluid for examination if deemed necessary. -
DOSAGE & ADMINISTRATION
As directed by a veterinarian. Dosage is dependent upon the age, weight and clinical condition of the patient as well as laboratory determinations.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit.
All injections in plastic containers are intended for intravenous administration using sterile equipment.
Additives may be incompatible. Complete information is not available. Those additives known to be incompatible should not be used. Consult with pharmacist, if available. If, in the informed judgment of the veterinarian, it is deemed advisable to introduce additives, use aseptic technique. Mix thoroughly when additives have been introduced. Do not store solutions containing additives. - OVERDOSAGE
-
HOW SUPPLIED
Lactated Ringer’s Injection, USP in plastic container is available as follows:
Size (mL) Code NDC
5000 2B8219 NDC 0338-0092-02
Exposure of pharmaceutical products to heat should be minimized. Avoid excessive heat. It is recommended the product be stored at room temperature (25°C); brief exposure up to 40°C does not adversely affect the product. -
Directions for use of plastic container
To Open
Tear overwrap down side at slit and remove solution container. Visually inspect the container. If the outlet port protector is damaged, detached, or not present, discard container as solution path sterility may be impaired. Some opacity of the plastic due to moisture absorption during the sterilization process may be observed. This is normal and does not affect the solution quality or safety. The opacity will diminish gradually. Check for minute leaks by squeezing inner bag firmly. If leaks are found, discard solution as sterility may be impaired. If supplemental medication is desired, follow directions below.Preparation for Administration
- Suspend container from eyelet support.
- Remove plastic protector from outlet port at bottom of container.
- Attach administration set. Refer to complete directions accompanying set.
To Add Medication
WARNING: Additives may be incompatible.
To add medication before solution administration1. Prepare medication site.
2. Using syringe with 19 to 22 gauge needle, puncture medication port and inject.
3. Mix solution and medication thoroughly. For high density medication such as potassium chloride, squeeze ports while ports are upright and mix thoroughly.
To add medication during solution administration
1. Close clamp on the set.
2. Prepare medication site.
3. Using syringe with 19 to 22 gauge needle, puncture resealable
medication port and inject.
4. Remove container from IV pole and/or turn to an upright position.
5. Evacuate both ports by squeezing them while container is in the upright position.
6. Mix solution and medication thoroughly.
7. Return container to in use position and continue administration.
CAUTION: Federal (USA) law restricts this drug to use by or on the order of a licensed veterinarian. - SPL UNCLASSIFIED SECTION
-
PRINCIPAL DISPLAY PANEL

Container Label
Veterinary Lactated
Ringer’s Injection USP5000 mL
EACH 100 ML CONTAINS 600 MG SODIUM CHLORIDE USP
310 MG SODIUM LACTATE 30 MG POTASSIUM CHLORIDE USP
20 MG CALCIUM CHLORIDE USP PH 6.5 (6.0 TO 7.5) MEQ/L
SODIUM 130 POTASSIUM 4 CALCIUM 2.7 CHLORIDE 109
LACTATE 28 OSMOLARITY 273 MOSMOL/L (CALC)
STERILE NONPYROGENIC SINGLE DOSE CONTAINER CONTAINS
NO ANTIMICROBIALAGENTS USE SOLUTION PROMPTLY
FOLLOWING INITIALENTRY NOT FOR USE INTHE TREATMENT
OF LACTIC ACIDOSIS ADDITIVES MAY BE INCOMPATIBLE
CONSULT WITH PHARMACIST IF AVAILABLE WHEN INTRODUCING
ADDITIVES USE ASEPTIC TECHNIQUE MIX THOROUGHLY DISCARD
UNUSED PORTION DOSAGE INTRAVENOUSLY AS DIRECTED BY
A VETERINARIAN SEE PACKAGE INSERT CAUTIONS SQUEEZE
AND INSPECT INNER BAG WHICH MAINTAINS PRODUCT
STERILITY DISCARD IF LEAKS ARE FOUND DO NOT ADMINISTER
SIMULTANEOUSLY WITH BLOOD DO NOT USE UNLESS SOLUTION
IS CLEAR AND SEALIS INTACT STORE UNIT IN MOISTURE
BARRIER OVERWRAP AT ROOM TEMPERATURE (25°C/77°F)
UNTIL READY TO USE AVOID EXCESSIVE HEATFOR ANIMAL USE ONLY
CAUTION FEDERAL(USA) LAW RESTRICTS THIS DRUG TO USE BY
OR ON THE ORDER OF A LICENSED VETERINARIAN
BAXTER IS A REGISTERED TRADEMARK OF BAXTER INTERNATIONAL, INC.FOR CUSTOMER SERVICE CALL
800 933 0303BAXTER HEALTHCARE CORPORATION
DEERFIELD, IL 60015 USAMADE IN USA
NDC 0338-0092-02
2B8219
07-25-00-0419Baxter Logo
4500
4000
3500
3000
2500
2000
1500
1000
500
-
INGREDIENTS AND APPEARANCE
VETERINARY LACTATED RINGERS
sodium chloride, sodium lactate, potassium chloride, calcium chloride injection, solutionProduct Information Product Type PRESCRIPTION ANIMAL DRUG Item Code (Source) NDC:0338-0092 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) (CHLORIDE ION - UNII:Q32ZN48698, SODIUM CATION - UNII:LYR4M0NH37) SODIUM CHLORIDE 600 mg in 100 mL SODIUM LACTATE (UNII: TU7HW0W0QT) (LACTIC ACID - UNII:33X04XA5AT, SODIUM CATION - UNII:LYR4M0NH37) SODIUM LACTATE 310 mg in 100 mL POTASSIUM CHLORIDE (UNII: 660YQ98I10) (POTASSIUM CATION - UNII:295O53K152, CHLORIDE ION - UNII:Q32ZN48698) POTASSIUM CHLORIDE 30 mg in 100 mL CALCIUM CHLORIDE (UNII: M4I0D6VV5M) (CALCIUM CATION - UNII:2M83C4R6ZB, CHLORIDE ION - UNII:Q32ZN48698) CALCIUM CHLORIDE 20 mg in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0338-0092-02 2 in 1 CARTON 1 5000 mL in 1 BAG Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 01/01/2019 Labeler - Baxter Healthcare Corporation (005083209) Establishment Name Address ID/FEI Business Operations Baxter Healthcare Corporation 059140764 analysis, label, manufacture, pack, sterilize, api manufacture Establishment Name Address ID/FEI Business Operations Baxter Healthcare Corporation 194684502 analysis Establishment Name Address ID/FEI Business Operations Baxter, S.A. de C.V. 810432484 analysis, label, manufacture, pack, sterilize

