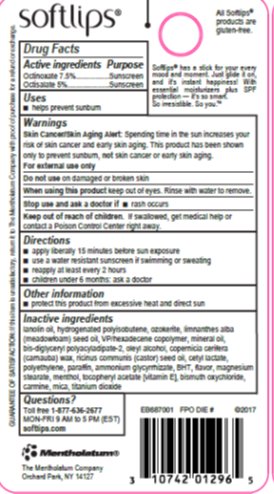
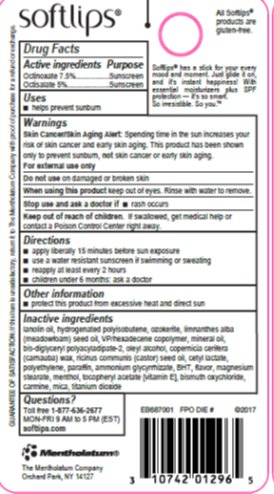
Label: SOFTLIPS PEARL- octinoxate, octisalate stick
- NDC Code(s): 10742-8573-1
- Packager: The Mentholatum Company
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated February 7, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredients
- Purpose
- Uses
- Warnings
- Directions
- Other information
-
Inactive ingredients
lanolin oil, hydrogenated polyisobutene, ozokerite, limnanthes alba (meadowfoam) seed oil, VP/hexadecene copolymer, mineral oil, bis-diglyceryl polyacyladipate-2, oleyl alcohol, copernicia cerifera (carnauba) wax, ricinus communis (castor) seed oil, cetyl lactate, polyethylene, paraffin, ammonium glycyrrhizate, BHT, flavor, magnesium stearate, menthol, tocopheryl acetate [vitamin E], bismuth oxychloride, carmine, mica, titanium dioxide
- Questions?
- Principal Display Panel
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
SOFTLIPS PEARL
octinoxate, octisalate stickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:10742-8573 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 75 mg in 1 g OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 50 mg in 1 g Inactive Ingredients Ingredient Name Strength LANOLIN OIL (UNII: OVV5IIJ58F) HYDROGENATED POLYBUTENE (1300 MW) (UNII: 7D1YQ9Y5EZ) CERESIN (UNII: Q1LS2UJO3A) MEADOWFOAM SEED OIL (UNII: 412ZHA4T4Y) VINYLPYRROLIDONE/HEXADECENE COPOLYMER (UNII: KFR5QEN0N9) MINERAL OIL (UNII: T5L8T28FGP) BIS-DIGLYCERYL POLYACYLADIPATE-2 (UNII: 6L246LAM9T) OLEYL ALCOHOL (UNII: 172F2WN8DV) CARNAUBA WAX (UNII: R12CBM0EIZ) CASTOR OIL (UNII: D5340Y2I9G) CETYL LACTATE (UNII: A7EVH2RK4O) PARAFFIN (UNII: I9O0E3H2ZE) AMMONIUM GLYCYRRHIZATE (UNII: 3VRD35U26C) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) MAGNESIUM STEARATE (UNII: 70097M6I30) MENTHOL, UNSPECIFIED FORM (UNII: L7T10EIP3A) .ALPHA.-TOCOPHEROL (UNII: H4N855PNZ1) BISMUTH OXYCHLORIDE (UNII: 4ZR792I587) CARMINIC ACID (UNII: CID8Z8N95N) MICA (UNII: V8A1AW0880) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:10742-8573-1 1 in 1 BLISTER PACK 02/01/2010 1 2 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 02/01/2010 Labeler - The Mentholatum Company (002105757) Registrant - The Mentholatum Company (002105757) Establishment Name Address ID/FEI Business Operations The Mentholatum Company 002105757 manufacture(10742-8573)