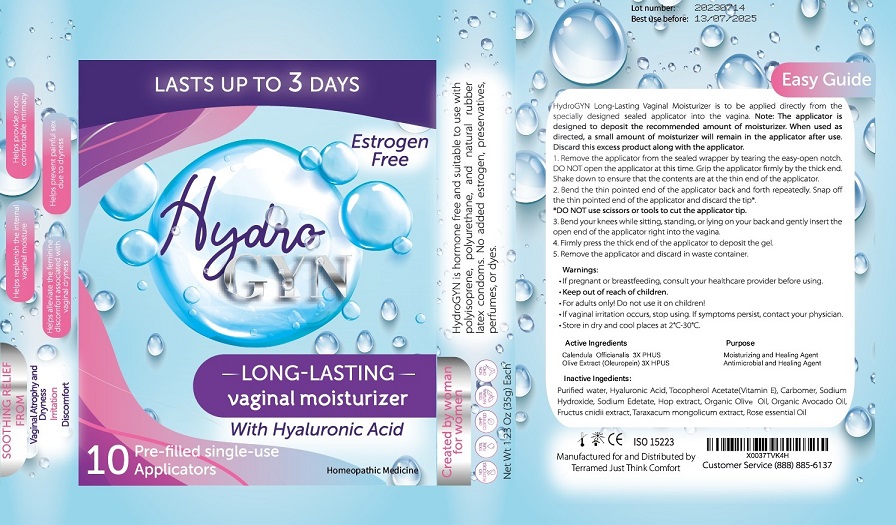
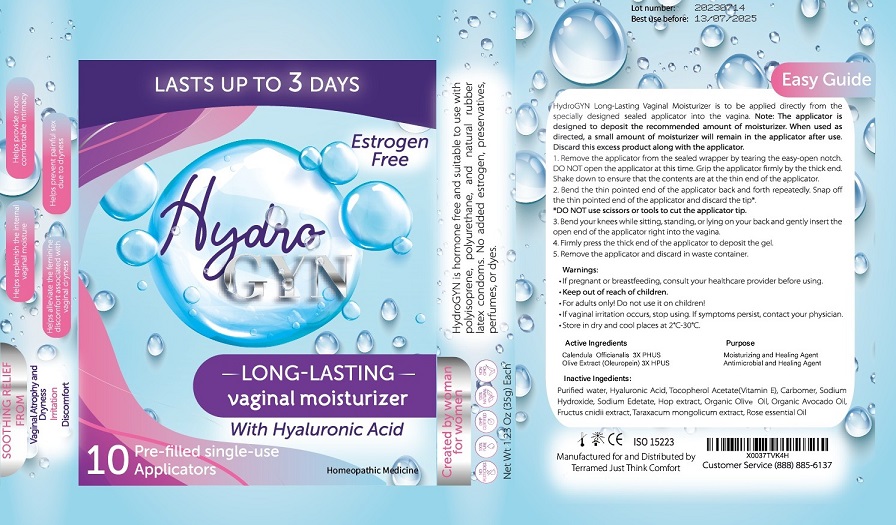
Label: TERRAMED JUST THINK COMFORT HYDRO- olive extract,calendula officinalis gel
- NDC Code(s): 83004-006-01
- Packager: Rida LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated October 26, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient Purpose
- PURPOSE
- Uses
-
Warnings
- For vaginal use only.
- If pregnant or breast-feeding ask a healthcare professional before use.
- Keep out of the reach of childrenIf swallowed, get medical help or contact Poison Control Center right away.
- For adults only. Do not use it on children.
- If vaginal irritation occurs, stop using. If symptoms persist, contact your physician.
- Store in dry and cool place at 2°-30°C
- KEEP OUT OF REACH OF CHILDREN
-
Directions
For individuals aged 12 and older:
- Remove the applicator from the sealed wrapper by tearing the easy-open notch. DO NOT open the applicator at this time. Grip the applicator firmly by the thick end. Shake down to ensure that the contents are at the thin end of the applicator.
- Bend the thin pointed end of the applicator back and forth repeatedly. Snap off the thin pointed end of the applicator and discard the tip.
- DO NOT use scissors or tools to cut the applicator tip
- Bend your knees while sitting, standing or lying on your back and gently insert the open end of the applicator right into the vagina.
- Firmly press the thick end of the applicator to deposit the gel.
- Remove the applicator and discard in waste container.
- Inactive Ingredients
- Other information
- Product label
-
INGREDIENTS AND APPEARANCE
TERRAMED JUST THINK COMFORT HYDRO
olive extract,calendula officinalis gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:83004-006 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OLEUROPEIN (UNII: 2O4553545L) (OLEUROPEIN - UNII:2O4553545L) OLEUROPEIN 3 [hp_X] in 100 mL CALENDULA OFFICINALIS FLOWERING TOP (UNII: 18E7415PXQ) (CALENDULA OFFICINALIS FLOWERING TOP - UNII:18E7415PXQ) CALENDULA OFFICINALIS FLOWERING TOP 3 [hp_X] in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) HYALURONATE SODIUM (UNII: YSE9PPT4TH) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) POVIDONE-IODINE (UNII: 85H0HZU99M) CARBOMER HOMOPOLYMER, UNSPECIFIED TYPE (UNII: 0A5MM307FC) SODIUM HYDROXIDE (UNII: 55X04QC32I) EDETATE SODIUM (UNII: MP1J8420LU) HOPS (UNII: 01G73H6H83) OLIVE OIL (UNII: 6UYK2W1W1E) AVOCADO OIL (UNII: 6VNO72PFC1) CNIDIUM MONNIERI FRUIT (UNII: V1IA3S3CUS) TARAXACUM MONGOLICUM WHOLE (UNII: 4E8QL88U6H) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:83004-006-01 10 in 1 BOX 11/11/2023 1 100 mL in 1 APPLICATOR; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 11/11/2023 Labeler - Rida LLC (004425803)