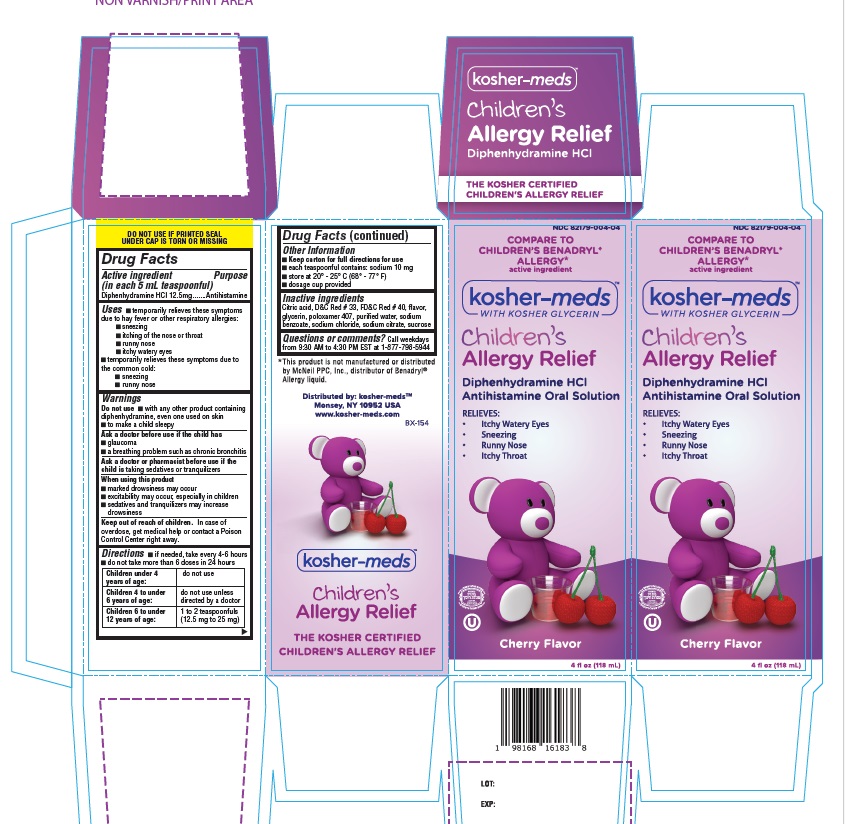
Label: KOSHER MEDS CHILDRENS ALLERGY RELIEF- diphenhydramine hcl solution
- NDC Code(s): 82179-004-04
- Packager: Mollec Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated May 15, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
- Purpose
- Uses
-
Warnings
Do not use
- with any other product containing diphenhydramine, even one used on skin
- to make a child sleepy
Ask a doctor before use if the child has
- glaucoma
- a breathing problem such as chronic bronchitis
Ask a doctor or pharmacist before use if the child istaking sedatives or tranquilizers
When using this product
- marked drowsiness may occur
- excitability may occur, especially in children
- sedatives and tranquilizers may increase drowsiness
Keep out of reach of children.In case of overdose, get medical help or contact a Poison Control Center right away.
- KEEP OUT OF REACH OF CHILDREN
- Directions
- Other Information
- Inactive ingredients
- Questions or comments?
- Product label
-
INGREDIENTS AND APPEARANCE
KOSHER MEDS CHILDRENS ALLERGY RELIEF
diphenhydramine hcl solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:82179-004 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DIPHENHYDRAMINE HYDROCHLORIDE (UNII: TC2D6JAD40) (DIPHENHYDRAMINE - UNII:8GTS82S83M) DIPHENHYDRAMINE HYDROCHLORIDE 12.5 mg in 5 mL Inactive Ingredients Ingredient Name Strength CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) FD&C RED NO. 40 (UNII: WZB9127XOA) GLYCERIN (UNII: PDC6A3C0OX) POLOXAMER 407 (UNII: TUF2IVW3M2) WATER (UNII: 059QF0KO0R) SODIUM BENZOATE (UNII: OJ245FE5EU) SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM CITRATE, UNSPECIFIED FORM (UNII: 1Q73Q2JULR) SUCROSE (UNII: C151H8M554) D&C RED NO. 33 (UNII: 9DBA0SBB0L) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:82179-004-04 1 in 1 CARTON 04/18/2024 1 118 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M013 04/18/2024 Labeler - Mollec Inc (040088196)