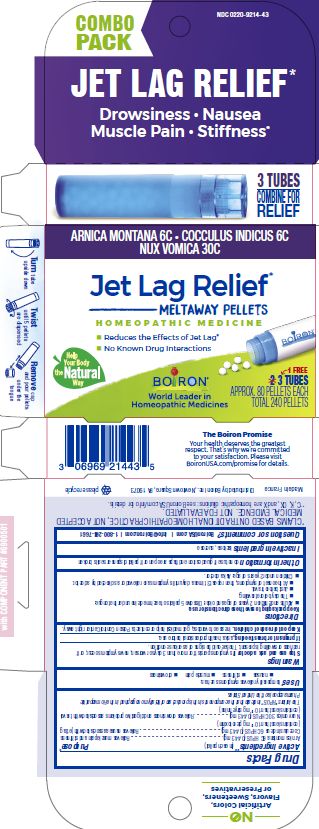
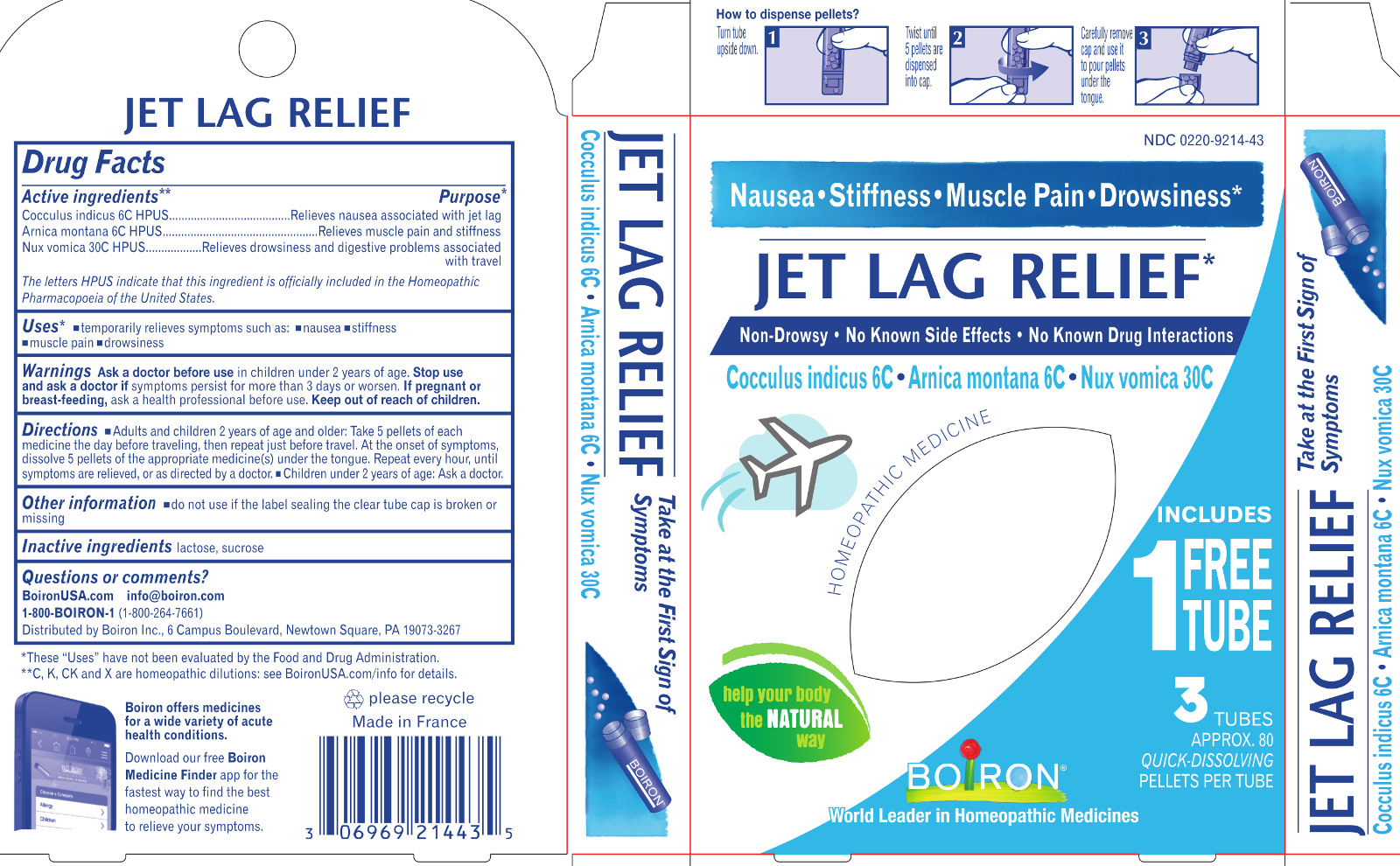
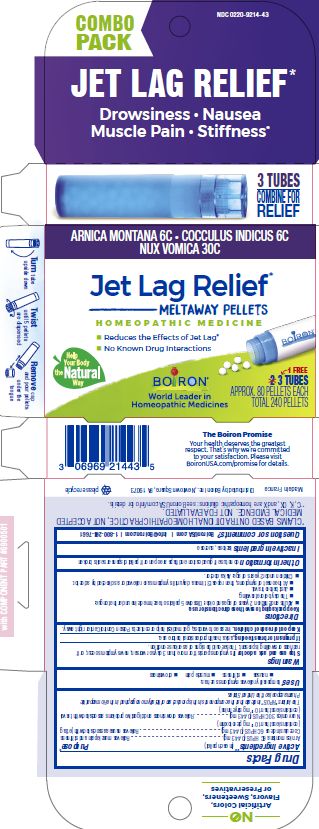
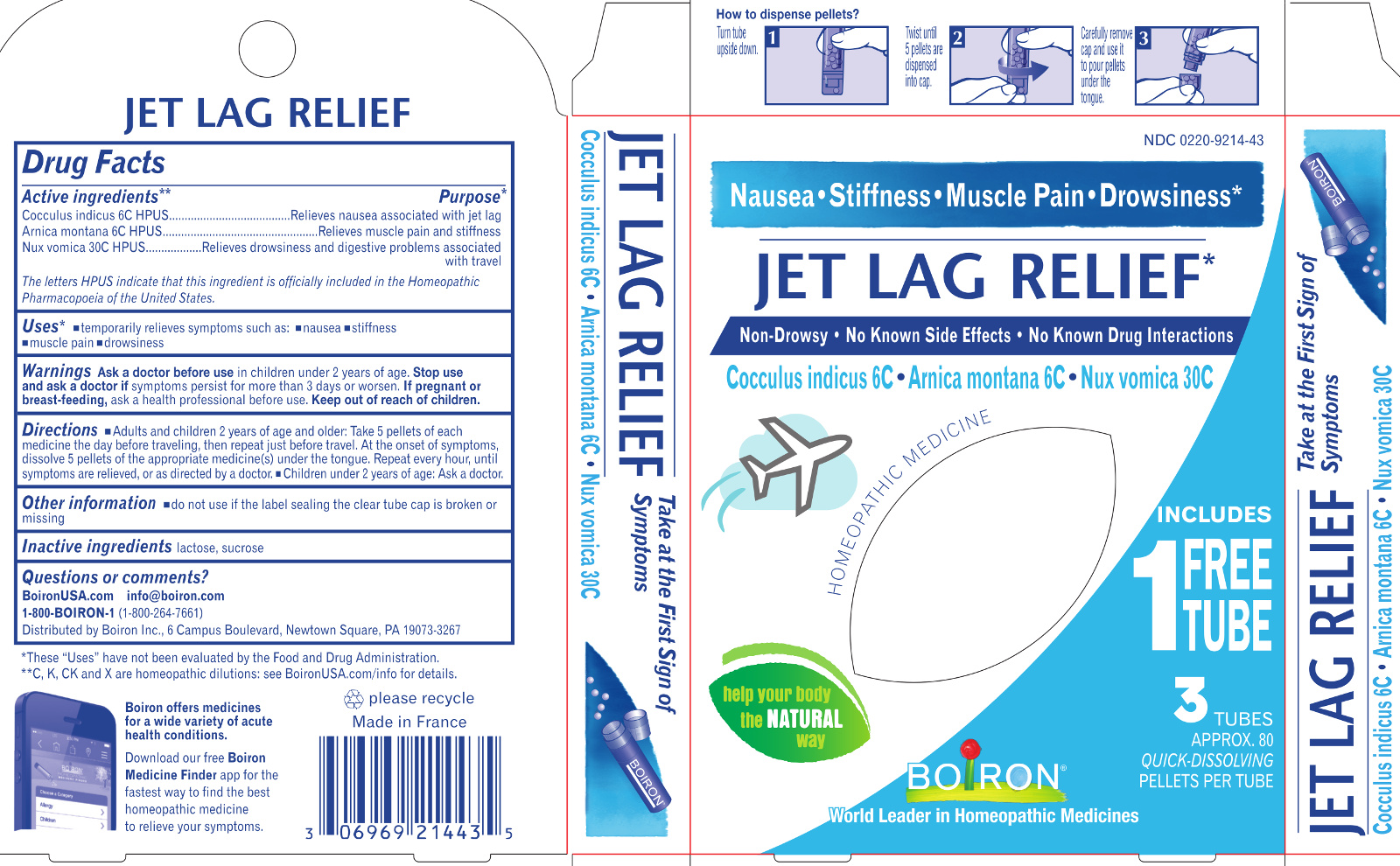
Label: JET LAG RELIEF- anamirta cocculus seed, arnica montana, strychnos nux-vomica whole pellet
- NDC Code(s): 0220-9214-43
- Packager: Laboratoires Boiron
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated October 4, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
ACTIVE INGREDIENT
Active Ingredients** (in each pellet)
Cocculus indicus 6C HPUS (0.443 mg)
Arnica montana 6C HPUS (0.443 mg) (contains less than 10 -12 mg picrotoxinin)
Nux Vomica 30C HPUS (0.443 mg) (contains less than 10 -12 mg strychnine)
The letters "HPUS" indicate that this ingredient is officially included in the Homeopathic Pharmacopoeia of the United States. - PURPOSE
- INDICATIONS & USAGE
- WARNINGS
-
DOSAGE & ADMINISTRATION
Directions
Keep packaging to save these directions for use.
Adults and children 2 years of age and older: Dissolve 5 pellets of each medicine under the tongue:
- The day before traveling.
- Just before travel.
- At the onset of symptoms, then repeat 3 times a day until symptoms are relieved or as directed by a doctor.
Children under 2 years of age: Ask a doctor.
- Inactive ingredients
-
SPL UNCLASSIFIED SECTION
do not use if glued carton and flaps are open or if pellet dispenser seal is broken
Jet Lag Relief*
Drowsiness Nausea Muscle Pain Stiffness*
Reduces the effects of Jet Lag*
No Know Drug Interactions
3 Tubes combine for relief
3 Tubes
Approx. 80 Pellets Each
Total 240 Pellets
Turn tube upside down Twist until 5 pellets are dispensed Remove cap and pour pellets under the tongue
*CLAIMS BASED ON TRADITIONAL HOMEOPATHIC PRACTICE, NOT ACCEPTED MEDICAL EVIDENCE. NOT FDA EVALUATED.
**C,K,CK, and X are homeopathic dilutions: see BoironUSA.com/info for details. - Questions, Comments?
- Principle Display Panel - Jet Lag Relief
-
INGREDIENTS AND APPEARANCE
JET LAG RELIEF
anamirta cocculus seed, arnica montana, strychnos nux-vomica whole pelletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:0220-9214 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ANAMIRTA COCCULUS SEED (UNII: 810258W28U) (ANAMIRTA COCCULUS SEED - UNII:810258W28U) ANAMIRTA COCCULUS SEED 6 [hp_C] ARNICA MONTANA (UNII: O80TY208ZW) (ARNICA MONTANA - UNII:O80TY208ZW) ARNICA MONTANA 6 [hp_C] STRYCHNOS NUX-VOMICA WHOLE (UNII: AB2NX17POS) (STRYCHNOS NUX-VOMICA WHOLE - UNII:AB2NX17POS) STRYCHNOS NUX-VOMICA WHOLE 30 [hp_C] Inactive Ingredients Ingredient Name Strength SUCROSE (UNII: C151H8M554) LACTOSE (UNII: J2B2A4N98G) Product Characteristics Color white Score Shape ROUND Size 4mm Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0220-9214-43 3 in 1 PACKAGE 01/01/2017 1 80 in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 01/01/2017 Labeler - Laboratoires Boiron (282560473) Registrant - Boiron Inc. (014892269) Establishment Name Address ID/FEI Business Operations Boiron 282560473 manufacture(0220-9214)