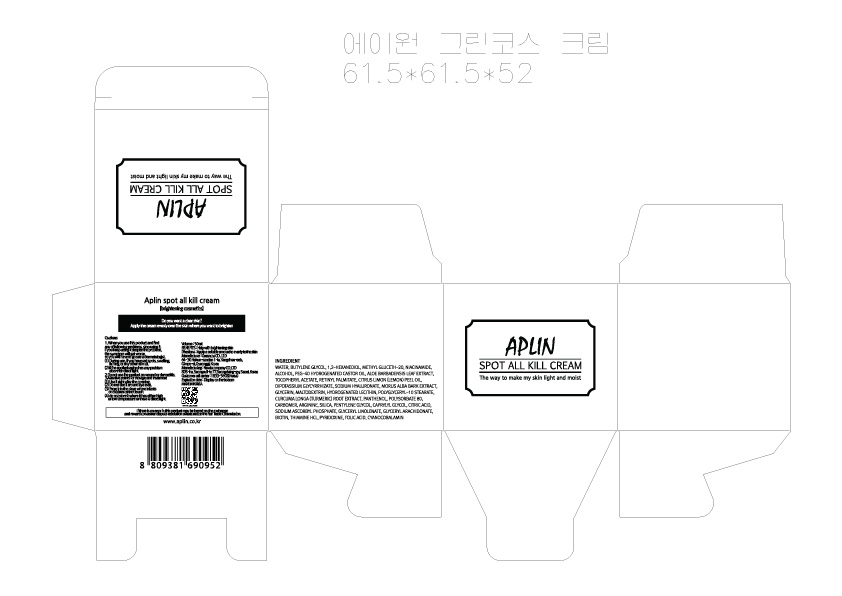
Label: APLIN SPOT ALL KILL- niacinamide cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 71079-0001-1 - Packager: Newizcompany
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated November 17, 2016
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- INACTIVE INGREDIENT
- PURPOSE
- KEEP OUT OF REACH OF CHILDREN
- INDICATIONS & USAGE
-
WARNINGS
Cautions :
1. When you use this product and find any of following problems, stop using it. if you keep using it despite the problem, the symptom will get worse, so you will have to go see a dermatologist.
(1) During use, if you have red spots, swelling, itching, or any other stimuli.
(2) if the applied region has any problem above for direct light.
2.Do not use the product on wound or dermatitis. 3.Attentive points for storage and treatment :
(1)Use il right after the opening.
(2)Do not use it around eye area.
(3)Store it in the place where infants or children cannot touch. (4)do not store it where it has either high or low temperature or there is direct light.
- DOSAGE & ADMINISTRATION
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
APLIN SPOT ALL KILL
niacinamide creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:71079-0001 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength NIACINAMIDE (UNII: 25X51I8RD4) (NIACINAMIDE - UNII:25X51I8RD4) NIACINAMIDE 2 g in 100 mL Inactive Ingredients Ingredient Name Strength GLYCERIN (UNII: PDC6A3C0OX) ALOE VERA FLOWER (UNII: 575DY8C1ER) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:71079-0001-1 50 mL in 1 JAR; Type 0: Not a Combination Product 09/01/2016 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 10/01/2015 Labeler - Newizcompany (689851607) Registrant - Newizcompany (689851607) Establishment Name Address ID/FEI Business Operations Newizcompany 689851607 manufacture(71079-0001)