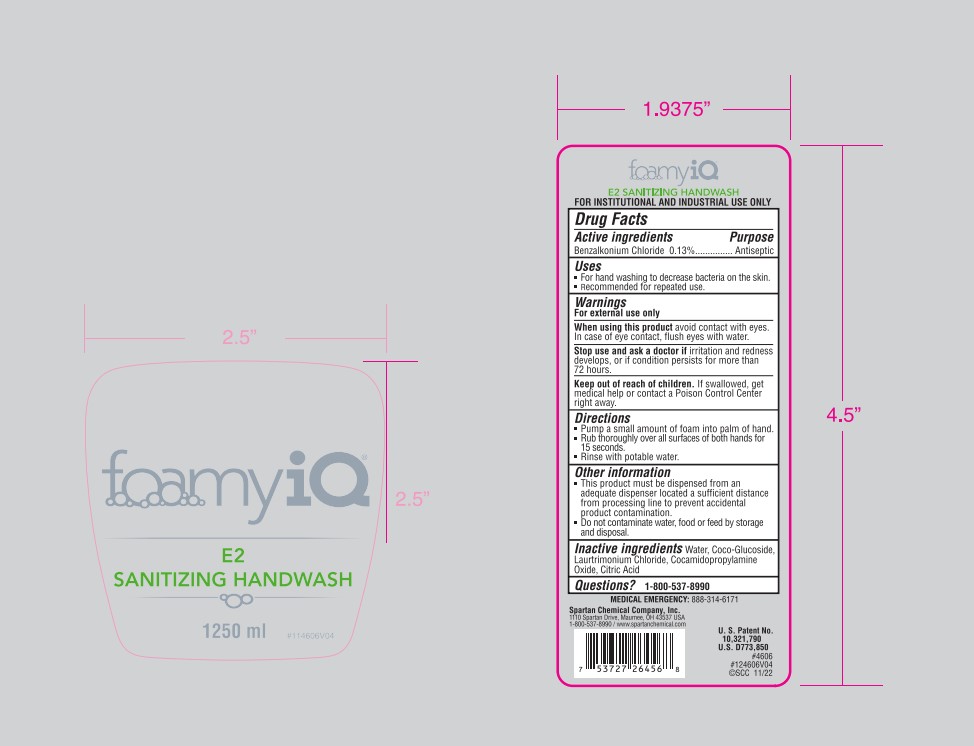
Label: FOAMYIQ E2 SANITIZING HANDWASH- benzalkonium chloride soap
- NDC Code(s): 64009-200-06
- Packager: Spartan Chemical Company
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated December 19, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active Ingredients
- Purpose
- Uses
- Warnings
- Directions
- Other Information
- Inactive Ingredients
- Questions?
- Principal Display Panel - 1.25 L Container Label
-
INGREDIENTS AND APPEARANCE
FOAMYIQ E2 SANITIZING HANDWASH
benzalkonium chloride soapProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:64009-200 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) (BENZALKONIUM - UNII:7N6JUD5X6Y) BENZALKONIUM CHLORIDE 1.3 g in 1 L Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) COCO GLUCOSIDE (UNII: ICS790225B) LAURTRIMONIUM CHLORIDE (UNII: A81MSI0FIC) COCAMIDOPROPYLAMINE OXIDE (UNII: M4SL82J7HK) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:64009-200-06 1.25 L in 1 CONTAINER; Type 0: Not a Combination Product 12/03/2019 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug 505G(a)(3) 12/03/2019 Labeler - Spartan Chemical Company (005036728) Establishment Name Address ID/FEI Business Operations Spartan Chemical Company 005036728 manufacture(64009-200)