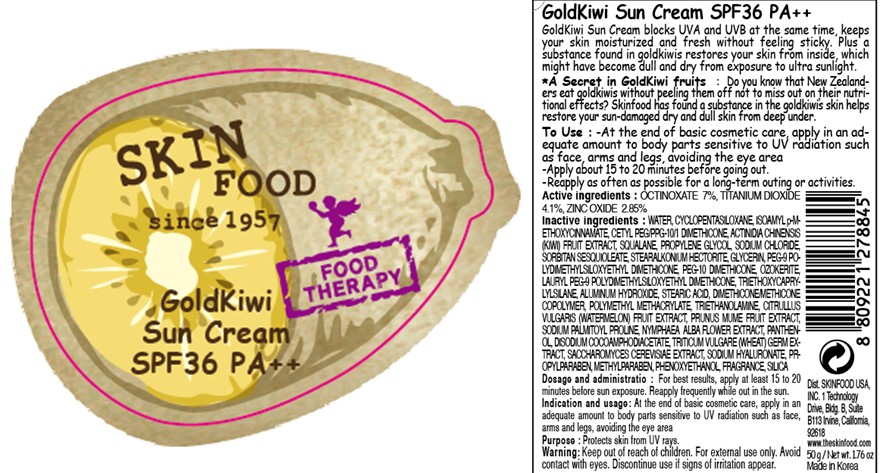
Label: GOLDKIWI SUN SPF 36 PA PLUS PLUS- octinoxate cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 76214-034-01 - Packager: SKINFOOD CO., LTD.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated October 6, 2011
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
-
INACTIVE INGREDIENT
Inactive ingredients:
WATER, CYCLOPENTASILOXANE, ISOAMYL p-METHOXYCINNAMATE, CETYL PEG/PPG-10/1 DIMETHICONE, ACTINIDIA CHINENSIS (KIWI) FRUIT EXTRACT, SQUALANE, PROPYLENE GLYCOL, SODIUM CHLORIDE, SORBITAN SESQUIOLEATE, STEARALKONIUM HECTORITE, GLYCERIN, PEG-9 POLYDIMETHYLSILOXYETHYL DIMETHICONE, PEG-10 DIMETHICONE, OZOKERITE, LAURYL PEG-9 POLYDIMETHYLSILOXYETHYL DIMETHICONE, TRIETHOXYCAPRYLYLSILANE, ALUMINUM HYDROXIDE, STEARIC ACID, DIMETHICONE/METHICONE COPOLYMER, POLYMETHYL METHACRYLATE, TRIETHANOLAMINE, CITRULLUS VULGARIS (WATERMELON) FRUIT EXTRACT, PRUNUS MUME FRUIT EXTRACT, SODIUM PALMITOYL PROLINE, NYMPHAEA ALBA FLOWER EXTRACT, PANTHENOL, DISODIUM COCOAMPHODIACETATE, TRITICUM VULGARE (WHEAT) GERM EXTRACT, SACCHAROMYCES CEREVISIAE EXTRACT, SODIUM HYALURONATE, PROPYLPARABEN, METHYLPARABEN, PHENOXYETHANOL, FRAGRANCE, SILICA - PURPOSE
- WARNINGS
- KEEP OUT OF REACH OF CHILDREN
- INDICATIONS & USAGE
- DOSAGE & ADMINISTRATION
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
GOLDKIWI SUN SPF 36 PA PLUS PLUS
octinoxate creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:76214-034 Route of Administration CUTANEOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 3.5 g in 50 g TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM - UNII:D1JT611TNE) TITANIUM DIOXIDE 2.05 g in 50 g ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC CATION - UNII:13S1S8SF37) ZINC OXIDE 1.42 g in 50 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) CYCLOMETHICONE 5 (UNII: 0THT5PCI0R) KIWI FRUIT (UNII: 71ES77LGJC) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SQUALANE (UNII: GW89575KF9) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SODIUM CHLORIDE (UNII: 451W47IQ8X) SORBITAN SESQUIOLEATE (UNII: 0W8RRI5W5A) GLYCERIN (UNII: PDC6A3C0OX) TRIETHOXYCAPRYLYLSILANE (UNII: LDC331P08E) ALUMINUM HYDROXIDE (UNII: 5QB0T2IUN0) STEARIC ACID (UNII: 4ELV7Z65AP) METHYLPARABEN (UNII: A2I8C7HI9T) PHENOXYETHANOL (UNII: HIE492ZZ3T) PROPYLPARABEN (UNII: Z8IX2SC1OH) TROLAMINE (UNII: 9O3K93S3TK) WATERMELON (UNII: 231473QB6R) PRUNUS MUME FRUIT (UNII: 639190I8CU) SODIUM PALMITOYL PROLINE (UNII: 64L053FRFO) NYMPHAEA ALBA FLOWER (UNII: 40KQ7Q535O) PANTHENOL (UNII: WV9CM0O67Z) DISODIUM COCOAMPHODIACETATE (UNII: 18L9G3U51M) WHEAT GERM (UNII: YR3G369F5A) SACCHAROMYCES CEREVISIAE (UNII: 978D8U419H) HYALURONATE SODIUM (UNII: YSE9PPT4TH) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:76214-034-01 50 g in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 03/01/2011 Labeler - SKINFOOD CO., LTD. (690324173) Registrant - SKINFOOD CO., LTD. (690324173) Establishment Name Address ID/FEI Business Operations SKINFOOD CO., LTD. 690324173 manufacture