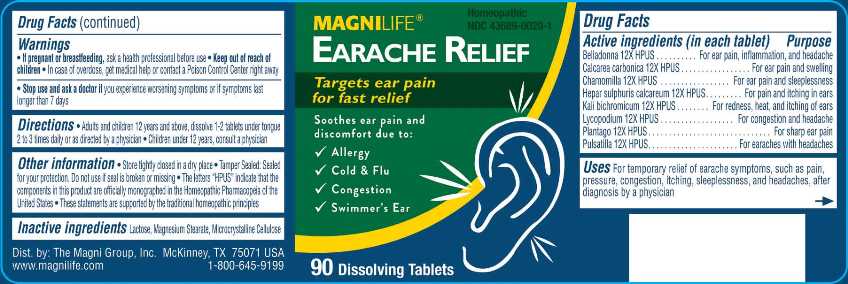
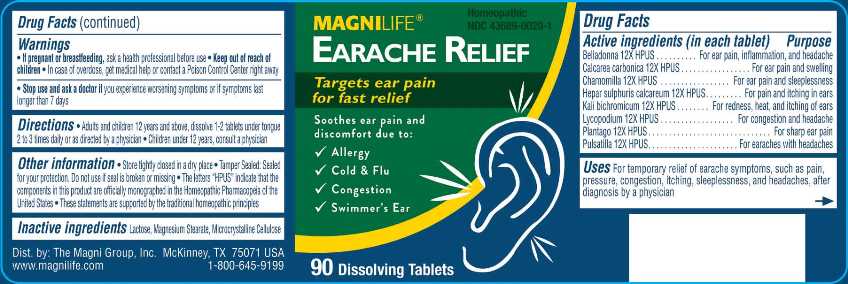
Label: EARACHE RELIEF (belladonna, calcarea carbonica, chamomilla, hepar sulphuris calcareum, kali bichromicum, lycopodium clavatum, plantago major, pulsatilla- vulgaris tablet
-
Contains inactivated NDC Code(s)
NDC Code(s): 43689-0020-1, 43689-0020-2 - Packager: The Magni Company
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated January 10, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENTS:
- USES:
- WARNINGS:
- KEEP OUT OF REACH OF CHILDREN:
-
OTHER INFORMATION:
• Store tightly closed in a cool, dry place (59° - 86°F)
• Tamper sealed: Sealed for your protection. Do not use if seal is broken or missing
• The letters "HPUS" indicate that the components in this product are officially monographed in the Homeopathic Pharmacopeia of the United
States• These statements are supported by the traditional homeopathic principles
- DIRECTIONS:
- INDICATIONS:
- INACTIVE INGREDIENTS:
- QUESTIONS:
- PACKAGE LABEL DISPLAY:
-
INGREDIENTS AND APPEARANCE
EARACHE RELIEF
belladonna, calcarea carbonica, chamomilla, hepar sulphuris calcareum, kali bichromicum, lycopodium clavatum, plantago major, pulsatilla (vulgaris) tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:43689-0020 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ATROPA BELLADONNA (UNII: WQZ3G9PF0H) (ATROPA BELLADONNA - UNII:WQZ3G9PF0H) ATROPA BELLADONNA 12 [hp_X] OYSTER SHELL CALCIUM CARBONATE, CRUDE (UNII: 2E32821G6I) (OYSTER SHELL CALCIUM CARBONATE, CRUDE - UNII:2E32821G6I) OYSTER SHELL CALCIUM CARBONATE, CRUDE 12 [hp_X] MATRICARIA CHAMOMILLA WHOLE (UNII: G0R4UBI2ZZ) (MATRICARIA CHAMOMILLA - UNII:G0R4UBI2ZZ) MATRICARIA CHAMOMILLA WHOLE 12 [hp_X] CALCIUM SULFIDE (UNII: 1MBW07J51Q) (CALCIUM SULFIDE - UNII:1MBW07J51Q) CALCIUM SULFIDE 12 [hp_X] POTASSIUM DICHROMATE (UNII: T4423S18FM) (DICHROMATE ION - UNII:9LKY4BFN2V) POTASSIUM DICHROMATE 12 [hp_X] LYCOPODIUM CLAVATUM SPORE (UNII: C88X29Y479) (LYCOPODIUM CLAVATUM SPORE - UNII:C88X29Y479) LYCOPODIUM CLAVATUM SPORE 12 [hp_X] PLANTAGO MAJOR WHOLE (UNII: W2469WNO6U) (PLANTAGO MAJOR - UNII:W2469WNO6U) PLANTAGO MAJOR WHOLE 12 [hp_X] PULSATILLA VULGARIS WHOLE (UNII: I76KB35JEV) (ANEMONE PULSATILLA - UNII:I76KB35JEV) PULSATILLA VULGARIS WHOLE 12 [hp_X] Inactive Ingredients Ingredient Name Strength LACTOSE (UNII: J2B2A4N98G) MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) Product Characteristics Color white Score no score Shape ROUND (Convex) Size 6mm Flavor Imprint Code ML Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:43689-0020-2 1 in 1 PACKAGE 08/24/2016 1 NDC:43689-0020-1 90 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 05/09/2016 Labeler - The Magni Company (113501902) Registrant - Apotheca Company (844330915) Establishment Name Address ID/FEI Business Operations Apotheca Company 844330915 manufacture(43689-0020) , api manufacture(43689-0020) , label(43689-0020) , pack(43689-0020)