Label: ALCORTIN A- hydrocortisone acetate, aloe vera leaf and iodoquinol gel
-
Contains inactivated NDC Code(s)
NDC Code(s): 68040-705-02, 68040-705-13 - Packager: Primus Pharmaceuticals
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated October 30, 2009
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
-
DESCRIPTION
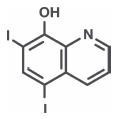
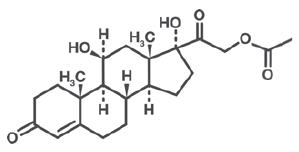
Each gram of Alcortin® A contains 2.0% (20 mg) Hydrocortisone Acetate and 1.0% (10 mg) Iodoquinol. Also contains 1.0% (10 mg) Aloe polysaccharide1. Other ingredients: Purified Water, Carbopol, Magnesium Aluminum Silicate, PPG-20 Methyl Glucose Ether, Aminomethyl Propanol, Propylene Glycol, Glycerine, Benzyl Alcohol, SD Alcohol 40 B, Biopeptide, Hydrochloric Acid, FD&C Blue # 1 and FD&C Yellow # 10.
- 1
- U.S. Patents
#6,436,679; #6,271,214; #6,133,440; #5,708,038; patent pending
-
CLINICAL PHARMACOLOGY
Hydrocortisone Acetate has anti-inflammatory, antipruritic and vasoconstrictive properties. While the mechanism of anti-inflammatory activity is unclear, there is evidence to suggest that a recognizable correlation exists between vasoconstrictor potency and therapeutic efficacy in humans. Iodoquinol has both antifungal and antibacterial properties.
Pharmacokinetics
The extent of percutaneous absorption of topical steroids is determined by many factors including the vehicle, the integrity of the epidermal barrier and the use of occlusive dressings. Hydrocortisone acetate can be absorbed from normal intact skin. Inflammation and/or other inflammatory disease processes in the skin increase percutaneous absorption. Occlusive dressings substantially increase the percutaneous absorption of topical corticosteroids. Once absorbed through the skin, hydrocortisone acetate is metabolized in the liver and most body tissue to hydrogenated and degraded forms such as tetrahydrocortisone and tetrahydrocortisol. These are excreted in the urine, mainly conjugated as glucuronides, together with a very small proportion of unchanged hydrocortisone acetate. There are no data available regarding the percutaneous absorption of iodoquinol; however, following oral administration, 3-5% of the dose was recovered in the urine as a glucuronide.
-
INDICATIONS AND USAGE
Based on a review of a related drug by the National Research Council and subsequent FDA classification for that drug, the indications are as follows: "Possibly" Effective: Contact or atopic dermatitis; impetiginized eczema; nummular eczema; endogenous chronic infectious dermatitis; stasis dermatitis; pyoderma; nuchal eczema and chronic eczematoid otitis externa; acne urticata; localized or disseminated neurodermatitis; lichen simplex chronicus; anogenital pruritus (vulvae, scroti, ani); folliculitis; bacterial dermatoses; mycotic dermatoses such as tinea (capitis, cruris, corporis, pedis); monliasis; intertrigo. Final classification of the less-than-effective indications requires further investigation.
- CONTRAINDICATIONS
-
WARNINGS AND PRECAUTIONS
For external use only. Keep away from eyes. If irritation develops, the use of Alcortin A should be discontinued and appropriate therapy instituted. Staining of the skin, hair and fabrics may occur. Not intended for use on infants or under diapers or occlusive dressings. If extensive areas are treated or if the occlusive dressing technique is used, the possibility exists of increased systemic absorption of the corticosteroid, and suitable precautions should be taken. Children may absorb proportionally larger amounts of topical corticosteroids and thus be more susceptible to systemic toxicity. Parents of pediatric patients should be advised not to use tight-fitting diapers or plastic pants on a child being treated in the diaper area, as these garments may constitute occlusive dressings. Iodoquinol may be absorbed through the skin and interfere with thyroid function tests. If such tests are contemplated, wait at least one month after discontinuance of therapy to perform these tests. The ferric chloride test for phenylketonuria (PKU) can yield a false positive result if iodoquinol is present in the diaper or urine. Prolonged use may result in overgrowth of non-susceptible organisms requiring appropriate therapy. Keep out of reach of children. Burning, itching, irritation and dryness have been reported infrequently following the use of topical corticosteroids.
Carcinogenesis, Mutagenisis and Impairment of Fertility
Long term animal studies have not been performed to evaluate the carcinogenic potential of the effect on fertility of hydrocortisone or iodoquinol. In vitro studies to determine mutagenicity with hydrocortisone have revealed negative results. Mutagenicity studies have not been performed with iodoquinol.
-
ADVERSE REACTIONS
The following local adverse reactions are reported infrequently with topical corticosteroids. These reactions are listed in an approximate decreasing order of occurrence. Burning, itching, irritation, dryness, folliculitis, hypertrichosis, acneiform eruptions, hypopigmentation, perioral dermatitis, allergic contact dermatitis, maceration of the skin, secondary infections, skin atrophy, striae and miliaria.
- DOSAGE AND ADMINISTRATION
- HOW SUPPLIED
- SPL UNCLASSIFIED SECTION
-
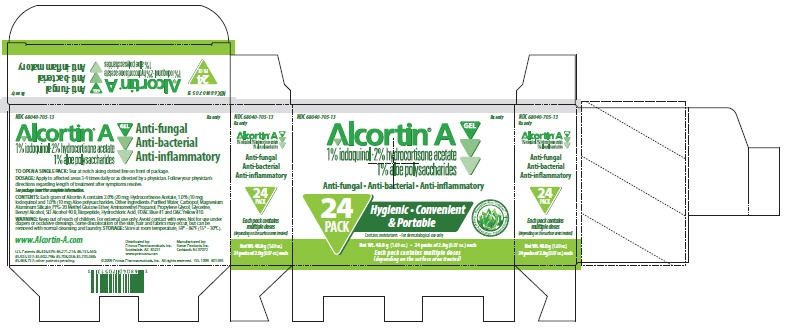
PRINCIPAL DISPLAY PANEL - 24 Pack Carton
NDC 68040-705-13
Rx onlyAlcortin® A Gel
1% iodoquinol•2% hydrocortisone acetate
1% aloe polysaccharidesAnti-fungal • Anti-bacterial • Anti-inflammatory
24
PACKHygienic • Convenient
& PortableContains moisturizers • For dermatological use only
Biopeptide Aloe Complex™
Deeper Penetration
Patented FormulaNet Wt. 48.0 g (1.69 oz.) • 24 packs of 2.0g (0.07 oz.) each
Each pack contains multiple doses
(depending on the surface area treated)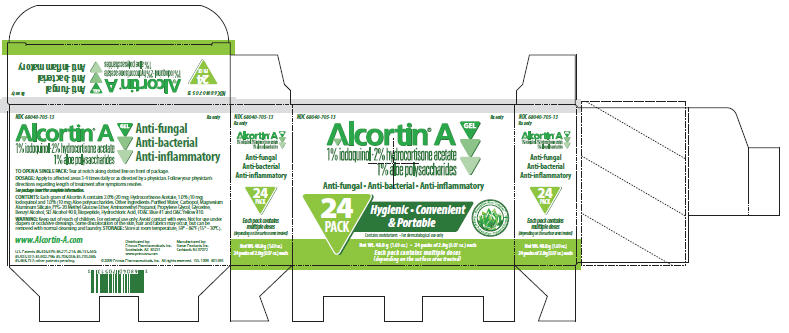
-
INGREDIENTS AND APPEARANCE
ALCORTIN A
hydrocortisone acetate, aloe vera leaf and iodoquinol gelProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:68040-705 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HYDROCORTISONE ACETATE (UNII: 3X7931PO74) (HYDROCORTISONE - UNII:WI4X0X7BPJ) HYDROCORTISONE ACETATE 20 mg in 1 g ALOE VERA LEAF (UNII: ZY81Z83H0X) (ALOE VERA LEAF - UNII:ZY81Z83H0X) ALOE VERA LEAF 10 mg in 1 g Iodoquinol (UNII: 63W7IE88K8) (Iodoquinol - UNII:63W7IE88K8) Iodoquinol 10 mg in 1 g Product Characteristics Color GREEN Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68040-705-13 24 in 1 BOX 1 NDC:68040-705-02 1 g in 1 PACKET Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date UNAPPROVED OTHER 01/01/2010 Labeler - Primus Pharmaceuticals (130834745) Registrant - Sonar Products, Inc (104283945) Establishment Name Address ID/FEI Business Operations Sonar Products, Inc 104283945 MANUFACTURE