Label: TRIESENCE- triamcinolone acetonide injection, suspension
- NDC Code(s): 0065-0543-01
- Packager: Alcon Laboratories, Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated August 4, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use TRIESENCE® (triamcinolone acetonide injectable suspension) 40 mg/mL safely and effectively. See full prescribing information for TRIESENCE® suspension.
TRIESENCE® (triamcinolone acetonide injectable suspension)
40 mg/mL
Initial U.S. Approval: 1957INDICATIONS AND USAGE
DOSAGE AND ADMINISTRATION
• Initial recommended dose for all indications except visualization: 4 mg (100 microliters of 40 mg/mL suspension) with subsequent dosage as needed over the course of treatment. (2.1)
• Recommended dose for visualization: 1 to 4 mg (25 to 100 microliters of 40 mg/mL suspension) administered intravitreally. (2.2)
DOSAGE FORMS AND STRENGTHS
Single use 1 mL vial containing 40 mg/mL of triamcinolone acetonide suspension. (3)
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
• TRIESENCE® suspension should not be administered intravenously. (5.1)
• Ophthalmic effects: May include cataracts, infections, and glaucoma. Monitor intraocular pressure. (5.1)
• Hypothalamic-pituitary-adrenal (HPA) axis suppression, Cushing's syndrome and hyperglycemia: Monitor patients for these conditions and taper doses gradually. (5.2)
• Infections: Increased susceptibility to new infection and increased risk of exacerbation, dissemination, or reactivation of latent infection. (5.3)
• Elevated blood pressure, salt and water retention, and hypokalemia: Monitor blood pressure and sodium, potassium serum levels. (5.4)
• GI perforation: Increased risk in patients with certain GI disorders. (5.5)
• Behavioral and mood disturbances: May include euphoria, insomnia, mood swings, personality changes, severe depression, and psychosis. (5.6)
• Decreases in bone density: Monitor bone density in patients receiving long term corticosteroid therapy. (5.7)
• Live or live attenuated vaccines: Do not administer to patients receiving immunosuppressive doses of corticosteroids. (5.8)
• Negative effects on growth and development: Monitor pediatric patients on long-term corticosteroid therapy. (5.9)
• Use in pregnancy: Fetal harm can occur with first trimester use. (5.10)
• Weight gain: May cause increased appetite. (5.11)
To report SUSPECTED ADVERSE REACTIONS, contact Alcon Laboratories, Inc. at 1-800-757-9195 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
• Anticoagulant agents: May enhance or diminish anticoagulant effects. Monitor coagulation indices. (7)
• Antidiabetic agents: May increase blood glucose concentrations. Dose adjustments of antidiabetic agents may be required. (7)
• CYP 3A4 inducers and inhibitors: May respectively increase or decrease clearance of corticosteroids necessitating dose adjustment. (7)
• NSAIDS including aspirin and salicylates: Increased risk of gastrointestinal side effects. (7)
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 12/2016
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
1.1 Ophthalmic Diseases
1.2 Visualization during Vitrectomy
2 DOSAGE AND ADMINISTRATION
2.1 Dosage for Treatment of Ophthalmic Diseases
2.2 Dosage for Visualization during Vitrectomy
2.3 Preparation for Administration
2.4 Administration
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Ophthalmic Effects
5.2 Alterations in Endocrine Function
5.3 Increased Risks Related to Infections
5.4 Alterations in Cardiovascular/Renal Function
5.5 Use in Patients with Gastrointestinal Disorders
5.6 Behavioral and Mood Disturbances
5.7 Decrease in Bone Density
5.8 Vaccination
5.9 Effect on Growth and Development
5.10 Use in Pregnancy
5.11 Weight Gain
5.12 Neuromuscular Effects
5.13 Kaposi's Sarcoma
6 ADVERSE REACTIONS
7 DRUG INTERACTIONS
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.3 Nursing Mothers
8.4 Pediatric Use
8.5 Geriatric Use
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
13.2 Animal Toxicology and/or Pharmacology
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
2.1 Dosage for Treatment of Ophthalmic Diseases
The initial recommended dose of TRIESENCE® suspension is 4 mg (100 microliters of 40 mg/mL suspension) with subsequent dosage as needed over the course of treatment.
2.2 Dosage for Visualization during Vitrectomy
The recommended dose of TRIESENCE® suspension is 1 to 4 mg (25 to 100 microliters of 40 mg/mL suspension) administered intravitreally.
2.3 Preparation for Administration
STRICT ASEPTIC TECHNIQUE IS MANDATORY. The vial should be vigorously shaken for 10 seconds before use to ensure a uniform suspension. Prior to withdrawal, the suspension should be inspected for clumping or granular appearance (agglomeration). An agglomerated product results from exposure to freezing temperatures and should not be used. After withdrawal, TRIESENCE® suspension should be injected without delay to prevent settling in the syringe. Careful technique should be employed to avoid the possibility of entering a blood vessel or introducing organisms that can cause infection.
2.4 Administration
The injection procedure should be carried out under controlled aseptic conditions, which include the use of sterile gloves, a sterile drape, and a sterile eyelid speculum (or equivalent). Adequate anesthesia and a broad-spectrum microbicide should be given prior to the injection.
Following the injection, patients should be monitored for elevation in intraocular pressure and for endophthalmitis. Monitoring may consist of a check for perfusion of the optic nerve head immediately after the injection, tonometry within 30 minutes following the injection, and biomicroscopy between two and seven days following the injection. Patients should be instructed to report any symptoms suggestive of endophthalmitis without delay.
Each vial should only be used for the treatment of a single eye. If the contralateral eye requires treatment, a new vial should be used and the sterile field, syringe, gloves, drapes, eyelid speculum, and injection needles should be changed before TRIESENCE® suspension is administered to the other eye.
- 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
Corticosteroids are contraindicated in patients with systemic fungal infections.
Triamcinolone is contraindicated in patients who are hypersensitive to corticosteroids or any components of this product. Rare instances of anaphylactoid reactions have occurred in patients receiving corticosteroid therapy. [See Adverse Reactions (6)].
-
5 WARNINGS AND PRECAUTIONS
5.1 Ophthalmic Effects
TRIESENCE® suspension should not be administered intravenously. Strict aseptic technique is mandatory.
Risk of infection
Corticosteroids may mask some signs of infection, and new infections may appear during their use. There may be decreased resistance and inability to localize infection when corticosteroids are used. Corticosteroids may enhance the establishment of secondary ocular infections due to fungi or viruses. If an infection occurs during corticosteroid therapy, it should be promptly controlled by suitable antimicrobial therapy.
See also Increased Risks Related to Infection (5.3).
Elevated Intraocular Pressure
Increases in intraocular pressure associated with triamcinolone acetonide injection have been observed in 20-60% of patients. This may lead to glaucoma with possible damage to the optic nerve. Effects on intraocular pressure may last up to 6 months following injection and are usually managed by topical glaucoma therapy. A small percentage of patients may require aggressive non-topical treatment. Intraocular pressure as well as perfusion of the optic nerve head should be monitored and managed appropriately.
Endophthalmitis
The rate of infectious culture positive endophthalmitis is 0.5%. Proper aseptic techniques should always be used when administering triamcinolone acetonide. In addition, patients should be monitored following the injection to permit early treatment should an infection occur.
Cataracts
Use of corticosteroids may produce cataracts, particularly posterior subcapsular cataracts.
Patients with Ocular Herpes Simplex
Corticosteroids should be used cautiously in patients with ocular herpes simplex because of possible corneal perforation. Corticosteroids should not be used in active ocular herpes simplex.
5.2 Alterations in Endocrine Function
Hypothalamic-pituitary-adrenal (HPA) axis suppression, Cushing's syndrome, and hyperglycemia. Monitor patients for these conditions with chronic use.
Corticosteroids can produce reversible HPA axis suppression with the potential for glucocorticosteroid insufficiency after withdrawal of treatment. Drug induced secondary adrenocortical insufficiency may be minimized by gradual reduction of dosage. This type of relative insufficiency may persist for months after discontinuation of therapy; therefore, in any situation of stress occurring during that period, hormone therapy should be reinstituted.
Metabolic clearance of corticosteroids is decreased in hypothyroid patients and increased in hyperthyroid patients. Changes in thyroid status of the patient may necessitate adjustment in dosage.
5.3 Increased Risks Related to Infections
Corticosteroids may increase the risks related to infections with any pathogen, including viral, bacterial, fungal, protozoan, or helminthic infections. The degree to which the dose, route and duration of corticosteroid administration correlates with the specific risks of infection is not well characterized; however, with increasing doses of corticosteroids, the rate of occurrence of infectious complications increases.
Corticosteroids may mask some signs of infection and may reduce resistance to new infections.
Corticosteroids may exacerbate infections and increase risk of disseminated infection. The use of corticosteroids in active tuberculosis should be restricted to those cases of fulminating or disseminated tuberculosis in which the corticosteroid is used for the management of the disease in conjunction with an appropriate antituberculous regimen.
Chickenpox and measles can have a more serious or even fatal course in non-immune children or adults on corticosteroids. In children or adults who have not had these diseases, particular care should be taken to avoid exposure. If a patient is exposed to chickenpox, prophylaxis with varicella zoster immune globulin (VZIG) may be indicated. If patient is exposed to measles, prophylaxis with pooled intramuscular immunoglobulin (IG) may be indicated. If chickenpox develops, treatment with antiviral agents may be considered.
Corticosteroids should be used with great care in patients with known or suspected Strongyloides (threadworm) infestation. In such patients, corticosteroid-induced immunosuppression may lead to Strongyloides hyperinfection and dissemination with widespread larval migration, often accompanied by severe enterocolitis and potentially fatal gram-negative septicemia.
Corticosteroids may increase risk of reactivation or exacerbation of latent infection. If corticosteroids are indicated in patients with latent tuberculosis or tuberculin reactivity, close observation is necessary as reactivation of the disease may occur. During prolonged corticosteroid therapy, these patients should receive chemoprophylaxis.
Corticosteroids may activate latent amebiasis. Therefore, it is recommended that latent or active amebiasis be ruled out before initiating corticosteroid therapy in any patient who has spent time in the tropics or in any patient with unexplained diarrhea.
Corticosteroids should not be used in cerebral malaria.
5.4 Alterations in Cardiovascular/Renal Function
Corticosteroids can cause elevation of blood pressure, salt and water retention, and increased excretion of potassium and calcium. These effects are less likely to occur with the synthetic derivatives except when used in large doses. Dietary salt restriction and potassium supplementation may be necessary. These agents should be used with caution in patients with hypertension, congestive heart failure, or renal insufficiency.
Literature reports suggest an association between use of corticosteroids and left ventricular free wall rupture after a recent myocardial infarction; therefore, therapy with corticosteroids should be used with caution in these patients.
5.5 Use in Patients with Gastrointestinal Disorders
There is an increased risk of gastrointestinal perforation in patients with certain GI disorders. Signs of GI perforation, such as peritoneal irritation, may be masked in patients receiving corticosteroids.
Corticosteroids should be used with caution if there is a probability of impending perforation, abscess or other pyogenic infections; diverticulitis; fresh intestinal anastomoses; and active or latent peptic ulcer.
5.6 Behavioral and Mood Disturbances
Corticosteroid use may be associated with central nervous system effects ranging from euphoria, insomnia, mood swings, personality changes, and severe depression, to frank psychotic manifestations. Also, existing emotional instability or psychotic tendencies may be aggravated by corticosteroids.
5.7 Decrease in Bone Density
Corticosteroids decrease bone formation and increase bone resorption both through their effect on calcium regulation (i.e., decreasing absorption and increasing excretion) and inhibition of osteoblast function. This, together with a decrease in the protein matrix of the bone secondary to an increase in protein catabolism, and reduced sex hormone production, may lead to inhibition of bone growth in children and adolescents and the development of osteoporosis at any age. Special consideration should be given to patients at increased risk of osteoporosis (i.e., postmenopausal women) before initiating corticosteroid therapy and bone density should be monitored in patients on long term corticosteroid therapy.
5.8 Vaccination
Administration of live or live attenuated vaccines is contraindicated in patients receiving immunosuppressive doses of corticosteroids. Killed or inactivated vaccines may be administered; however, the response to such vaccines cannot be predicted. Immunization procedures may be undertaken in patients who are receiving corticosteroids as replacement therapy, e.g., for Addison's disease.
While on corticosteroid therapy, patients should not be vaccinated against smallpox. Other immunization procedures should not be undertaken in patients who are on corticosteroids, especially on high dose, because of possible hazards of neurological complications and a lack of antibody response.
5.9 Effect on Growth and Development
Long-term use of corticosteroids can have negative effects on growth and development in children. Growth and development of pediatric patients on prolonged corticosteroid therapy should be carefully monitored.
5.10 Use in Pregnancy
Triamcinolone acetonide can cause fetal harm when administered to a pregnant woman. Human and animal studies suggest that use of corticosteroids during the first trimester of pregnancy is associated with an increased risk of orofacial clefts, intrauterine growth restriction and decreased birth weight. If this drug is used during pregnancy, or if the patient becomes pregnant while using this drug, the patient should be apprised of the potential hazard to the fetus. [See Use in Specific Populations (8.1)].
5.11 Weight Gain
Systemically administered corticosteroids may increase appetite and cause weight gain.
5.12 Neuromuscular Effects
Although controlled clinical trials have shown corticosteroids to be effective in speeding the resolution of acute exacerbations of multiple sclerosis, they do not show that they affect the ultimate outcome or natural history of the disease. The studies do show that relatively high doses of corticosteroids are necessary to demonstrate a significant effect.
An acute myopathy has been observed with the use of high doses of corticosteroids, most often occurring in patients with disorders of neuromuscular transmission (e.g., myasthenia gravis), or in patients receiving concomitant therapy with neuromuscular blocking drugs (e.g., pancuronium). This acute myopathy is generalized, may involve ocular and respiratory muscles, and may result in quadriparesis. Elevation of creatine kinase may occur. Clinical improvement or recovery after stopping corticosteroids may require weeks to years.
-
6 ADVERSE REACTIONS
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Adverse event data were collected from 300 published articles containing data from controlled and uncontrolled clinical trials which evaluated over 14,000 eyes treated with different concentrations of triamcinolone acetonide. The most common dose administered within these trials was triamcinolone acetonide 4 mg administered as primary or adjunctive therapy primarily as a single injection.
The most common reported adverse events following administration of triamcinolone acetonide were elevated intraocular pressure and cataract progression. These events have been reported to occur in 20-60% of patients.
Less common reactions occurring in up to 2% include endophthalmitis (infectious and non-infectious), hypopyon, injection site reactions (described as blurring and transient discomfort), glaucoma, vitreous floaters, and detachment of retinal pigment epithelium, optic disc vascular disorder, eye inflammation, conjunctival hemorrhage and visual acuity reduced. Cases of exophthalmos have also been reported.
Common adverse reactions for systemically administered corticosteroids include fluid retention, alteration in glucose tolerance, elevation in blood pressure, behavioral and mood changes, increased appetite and weight gain.
Other reactions reported to have occurred with the administration of corticosteroids include:
Allergic Reactions: Anaphylactoid reaction, anaphylaxis, angioedema
Cardiovascular: Bradycardia, cardiac arrest, cardiac arrhythmias, cardiac enlargement, circulatory collapse, congestive heart failure, fat embolism, hypertrophic cardiomyopathy in premature infants, myocardial rupture following recent myocardial infarction, pulmonary edema, syncope, tachycardia, thromboembolism, thrombophlebitis, vasculitis
Dermatologic: Acne, allergic dermatitis, cutaneous and subcutaneous atrophy, dry scalp, edema, facial erythema, hyper or hypopigmentation, impaired wound healing, increased sweating, petechiae and ecchymoses, rash, sterile abscess, striae, suppressed reactions to skin tests, thin fragile skin, thinning scalp hair, urticaria
Endocrine: Abnormal fat deposits, decreased carbohydrate tolerance, development of Cushingoid state, hirsutism, manifestations of latent diabetes mellitus and increased requirements for insulin or oral hypoglycemic agents in diabetics, menstrual irregularities, moon facies, secondary adrenocortical and pituitary unresponsiveness (particularly in times of stress, as in trauma, surgery or illness), suppression of growth in children
Fluid and Electrolyte Disturbances: Potassium loss, hypokalemic alkalosis, sodium retention
Gastrointestinal: Abdominal distention, elevation in serum liver enzymes levels (usually reversible upon discontinuation), hepatomegaly, hiccups, malaise, nausea, pancreatitis, peptic ulcer with possible perforation and hemorrhage, ulcerative esophagitis
Metabolic: Negative nitrogen balance due to protein catabolism
Musculoskeletal: Aseptic necrosis of femoral and humeral heads, charcot-like arthropathy, loss of muscle mass, muscle weakness, osteoporosis, pathologic fracture of long bones, steroid myopathy, tendon rupture, vertebral compression fractures
Neurological: Arachnoiditis, convulsions, depression, emotional instability, euphoria, headache, increased intracranial pressure with papilledema (pseudo-tumor cerebri) usually following discontinuation of treatment, insomnia, meningitis, neuritis, neuropathy, paraparesis/paraplegia, paresthesia, sensory disturbances, vertigo
Reproductive: Alteration in motility and number of spermatozoa.
-
7 DRUG INTERACTIONS
• Amphotericin B: There have been cases reported in which concomitant use of Amphotericin B and hydrocortisone was followed by cardiac enlargement and congestive heart failure. See Potassium depleting agents.
• Anticholinesterase agents: Concomitant use of anticholinesterase agents and corticosteroids may produce severe weakness in patients with myasthenia gravis. If possible, anticholinesterase agents should be withdrawn at least 24 hours before initiating corticosteroid therapy.
• Anticoagulant agents: Co-administration of corticosteroids and warfarin usually results in inhibition of response to warfarin, although there have been some conflicting reports. Therefore, coagulation indices should be monitored frequently to maintain the desired anticoagulant effect.
• Antidiabetic agents: Because corticosteroids may increase blood glucose concentrations, dosage adjustments of antidiabetic agents may be required.
• Antitubercular drugs: Serum concentrations of isoniazid may be decreased.
• CYP 3A4 inducers (e.g., barbiturates, phenytoin, carbamazepine, and rifampin): Drugs such as barbiturates, phenytoin, ephedrine, and rifampin, which induce hepatic microsomal drug metabolizing enzyme activity may enhance metabolism of corticosteroid and require that the dosage of corticosteroid be increased.
• CYP 3A4 inhibitors (e.g., ketoconazole, macrolide antibiotics): Ketoconazole has been reported to decrease the metabolism of certain corticosteroids by up to 60% leading to an increased risk of corticosteroid side effects.
• Cholestyramine: Cholestyramine may increase the clearance of corticosteroids.
• Cyclosporine: Increased activity of both cyclosporine and corticosteroids may occur when the two are used concurrently. Convulsions have been reported with concurrent use.
• Digitalis: Patients on digitalis glycosides may be at increased risk of arrhythmias due to hypokalemia.
• Estrogens, including oral contraceptives: Estrogens may decrease the hepatic metabolism of certain corticosteroids thereby increasing their effect.
• NSAIDS including aspirin and salicylates: Concomitant use of aspirin or other non-steroidal antiinflammatory agents and corticosteroids increases the risk of gastrointestinal side effects. Aspirin should be used cautiously in conjunction with corticosteroids in hypoprothrombinemia. The clearance of salicylates may be increased with concurrent use of corticosteroids.
• Potassium depleting agents (e.g., diuretics, Amphotericin B): When corticosteroids are administered concomitantly with potassium-depleting agents, patients should be observed closely for development of hypokalemia.
• Skin tests: Corticosteroids may suppress reactions to skin tests.
• Toxoids and live or inactivated vaccines: Due to inhibition of antibody response, patients on prolonged corticosteroid therapy may exhibit a diminished response to toxoids and live or inactivated vaccines. Corticosteroids may also potentiate the replication of some organisms contained in live attenuated vaccines.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Teratogenic Effects: Pregnancy Category D
[See Warnings and Precautions (5.10)]
Multiple cohort and case controlled studies in humans suggest that maternal corticosteroid use during the first trimester increases the rate of cleft lip with or without cleft palate from about 1/1000 infants to 3- 5/1000 infants. Two prospective case control studies showed decreased birth weight in infants exposed to maternal corticosteroids in utero.
Triamcinolone acetonide was teratogenic in rats, rabbits, and monkeys. In rats and rabbits, triamcinolone acetonide was teratogenic at inhalation doses of 0.02 mg/kg and above and in monkeys, triamcinolone acetonide was teratogenic at an inhalation dose of 0.5 mg/kg (1/4 and 7 times the recommended human dose). Dose-related teratogenic effects in rats and rabbits included cleft palate and/or internal hydrocephaly and axial skeletal defects, whereas the effects observed in monkeys were cranial malformations. These effects are similar to those noted with other corticosteroids.
Corticosteroids should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. Infants born to mothers who received corticosteroids during pregnancy should be carefully observed for signs of hypoadrenalism.
8.3 Nursing Mothers
Corticosteroids are secreted in human milk. Reports suggest that steroid concentrations in human milk are 5 to 25% of maternal serum levels, and that total infant daily doses are small, less than 0.2% of the maternal daily dose. The risk of infant exposure to steroids through breast milk should be weighed against the known benefits of breastfeeding for both the mother and baby.
8.4 Pediatric Use
The efficacy and safety of corticosteroids in the pediatric population are based on the well established course of effect of corticosteroids which is similar in pediatric and adult populations.
The adverse effects of corticosteroids in pediatric patients are similar to those in adults. [See Adverse Reactions (6)].
Like adults, pediatric patients should be carefully observed with frequent measurements of blood pressure, weight, height, intraocular pressure, and clinical evaluation for the presence of infection, psychosocial disturbances, thromboembolism, peptic ulcers, cataracts, and osteoporosis. Children, who are treated with corticosteroids by any route, including systemically administered corticosteroids, may experience a decrease in their growth velocity. This negative impact of corticosteroids on growth has been observed at low systemic doses and in the absence of laboratory evidence of HPA axis suppression (i.e., cosyntropin stimulation and basal cortisol plasma levels). Growth velocity may therefore be a more sensitive indicator of systemic corticosteroid exposure in children than some commonly used tests of HPA axis function. The linear growth of children treated with corticosteroids by any route should be monitored, and the potential growth effects of prolonged treatment should be weighed against clinical benefits obtained and the availability of other treatment alternatives. In order to minimize the potential growth effects of corticosteroids, children should be titrated to the lowest effective dose.
8.5 Geriatric Use
No overall differences in safety or effectiveness were observed between elderly subjects and younger subjects, and other reported clinical experience with triamcinolone has not identified differences in responses between the elderly and younger patients. However, the incidence of corticosteroid-induced side effects may be increased in geriatric patients and are dose-related. Osteoporosis is the most frequently encountered complication, which occurs at a higher incidence rate in corticosteroid-treated geriatric patients as compared to younger populations and in age-matched controls. Losses of bone mineral density appear to be greatest early on in the course of treatment and may recover over time after steroid withdrawal or use of lower doses.
-
11 DESCRIPTION
TRIESENCE® (triamcinolone acetonide injectable suspension) 40 mg/mL is a synthetic corticosteroid with anti-inflammatory action. Each mL of the sterile, aqueous suspension provides 40 mg of triamcinolone acetonide, with sodium chloride for isotonicity, 0.5% (w/v) carboxymethylcellulose sodium and 0.015% polysorbate 80. It also contains potassium chloride, calcium chloride (dihydrate), magnesium chloride (hexahydrate), sodium acetate (trihydrate), sodium citrate (dihydrate) and water for injection. Sodium hydroxide and hydrochloric acid may be present to adjust pH to a target value 6 - 7.5.
The chemical name for triamcinolone acetonide is 9-Fluro- 11β, 16α, 17,21-tetrahydroxypregna-1,4-diene-3,20-dione cyclic 16,17- acetal with acetone. Its structural formula of C24H31FO6 is:
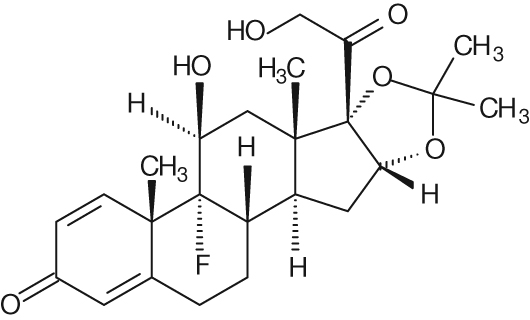
434.50 MW
Triamcinolone acetonide occurs as a white to cream-colored, crystalline powder having not more than a slight odor and is practically insoluble in water and very soluble in alcohol.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Naturally occurring glucocorticoids (hydrocortisone and cortisone), which also have salt-retaining properties, are used as replacement therapy in adrenocortical deficiency states. Their synthetic analogs such as prednisolone and triamcinolone are primarily used for their anti-inflammatory effects in disorders of many organ systems.
Triamcinolone acetonide possesses glucocorticoid activity typical of this class of drug, but with little or no mineralocorticoid activity. For the purposes of comparison, the following is the equivalent milligram dosage of the various glucocorticoids:
Cortisone, 25 Prednisone, 5 Paramethasone, 2 Hydrocortisone, 20 Methylprednisolone, 4 Betamethasone, 0.75 Prednisolone, 5 Triamcinolone, 4 Dexamethasone, 0.75 Corticosteroids have been demonstrated to depress the production of eosinophils and lymphocytes, but erythropoiesis and production of polymorphonuclear leukocytes are stimulated. Inflammatory processes (edema, fibrin deposition, capillary dilatation, migration of leukocytes and phagocytosis) and the later stages of wound healing (capillary proliferation, deposition of collagen, cicatrization) are inhibited.
12.3 Pharmacokinetics
Aqueous humor pharmacokinetics of triamcinolone have been assessed in 5 patients following a single intravitreal administration (4 mg) of triamcinolone acetonide. Aqueous humor samples were obtained from 5 patients (5 eyes) via an anterior chamber paracentesis on Days 1, 3, 10, 17 and 31 post injection. Peak aqueous humor concentrations of triamcinolone ranged from 2151 to 7202 ng/mL, half-life 76 to 635 hours, and the area under the concentration-time curve (AUC0-t) from 231 to 1911 ng.h/mL following the single intravitreal administration. The mean elimination half-life was 18.7 ± 5.7 days in 4 nonvitrectomized eyes (4 patients). In a patient who had undergone vitrectomy (1 eye), the elimination half-life of triamcinolone from the vitreous was much faster (3.2 days) relative to patients that had not undergone vitrectomy.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No evidence of mutagenicity was detected from in-vitro tests conducted with triamcinolone acetonide including a reverse mutation test in Salmonella bacteria and a forward mutation test in Chinese hamster ovary cells. With regard to carcinogenicity, in a two-year study in rats, triamcinolone acetonide caused no treatment-related carcinogenicity at oral doses up to 0.001mg/kg and in a two-year study in mice, triamcinolone acetonide caused no treatment-related carcinogenicity at oral doses up to 0.003 mg/kg (less than 1/25th of the recommended human dose). In male and female rats, triamcinolone acetonide caused no change in pregnancy rate at oral doses up to 0.015 mg/kg, but caused increased fetal resorptions and stillbirths and decreases in pup weight and survival at doses of 0.005 mg/kg (less than 1/10th of the recommended human dose).
13.2 Animal Toxicology and/or Pharmacology
Studies were conducted with triamcinolone acetonide, including those employing the proposed dosage form, i.e., 4.0% triamcinolone acetonide injectable suspension formulation containing 0.5% carboxymethylcellulose and 0.015% polysorbate-80 in a balanced salt solution.
Triamcinolone acetonide was demonstrated to be non-inflammatory when injected intravitreally in NZW rabbits, non-cytotoxic to mouse L-929 cells in an in-vitro assay and non-sensitizing in a guinea-pig maximization assay. Furthermore, the results of single-dose intravitreal injection studies with triamcinolone acetonide in both rabbits and monkeys demonstrate that the drug is well tolerated for up to one month with only minor findings of slight decrease in body weight gain and slight corneal thinning.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
TRIESENCE® (triamcinolone acetonide injectable suspension) 40 mg/mL is supplied as 1 mL of a 40 mg/mL sterile triamcinolone acetonide suspension in a flint Type 1 single use glass vial with a gray rubber stopper and an open target aluminum seal. Each labeled vial is sealed in a polycarbonate blister with a backing material which provides tamper evidence and is stored in a carton.
• 1 mL single use vial (NDC 0065-0543-01)
Storage
Store at 4° - 25° C (39° - 77° F); Do Not Freeze. Protect from light by storing in carton.
-
17 PATIENT COUNSELING INFORMATION
Patients should discuss with their physician if they have had recent or ongoing infections or if they have recently received a vaccine.
There are a number of medicines that can interact with corticosteroids such as triamcinolone. Patients should inform their health-care provider of all the medicines they are taking, including over-thecounter and prescription medicines (such as phenytoin, diuretics, digitalis or digoxin, rifampin, amphotericin B, cyclosporine, insulin or diabetes medicines, ketoconazole, estrogens including birth control pills and hormone replacement therapy, blood thinners such as warfarin, aspirin or other NSAIDS, barbiturates), dietary supplements, and herbal products. If patients are taking any of these drugs, alternate therapy, dosage adjustment, and/or special test may be needed during the treatment.
Patients should be advised of common adverse reactions that could occur with corticosteroid use to include elevated intraocular pressure, cataracts, fluid retention, alteration in glucose tolerance, elevation in blood pressure, behavioral and mood changes, increased appetite and weight gain.
U.S. Patent No. 6,395,294
© 2007, 2008 Alcon, Inc.
ALCON LABORATORIES, INC.
Fort Worth, Texas 76134 USA
9012681-0216
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
TRIESENCE
triamcinolone acetonide injection, suspensionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0065-0543 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TRIAMCINOLONE ACETONIDE (UNII: F446C597KA) (TRIAMCINOLONE ACETONIDE - UNII:F446C597KA) TRIAMCINOLONE ACETONIDE 40 mg in 1 mL Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) CARBOXYMETHYLCELLULOSE SODIUM (UNII: K679OBS311) POLYSORBATE 80 (UNII: 6OZP39ZG8H) POTASSIUM CHLORIDE (UNII: 660YQ98I10) CALCIUM CHLORIDE (UNII: M4I0D6VV5M) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) SODIUM ACETATE (UNII: 4550K0SC9B) SODIUM CITRATE (UNII: 1Q73Q2JULR) WATER (UNII: 059QF0KO0R) SODIUM HYDROXIDE (UNII: 55X04QC32I) HYDROCHLORIC ACID (UNII: QTT17582CB) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0065-0543-01 1 in 1 CARTON 11/15/2007 1 1 in 1 BLISTER PACK 1 1 mL in 1 VIAL, GLASS; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA022048 11/15/2007 Labeler - Alcon Laboratories, Inc. (008018525)


