Label: ANTI AGING STAR- octinoxate cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 42248-102-02 - Packager: Zenith Medicosm SL
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated January 21, 2012
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
-
Description
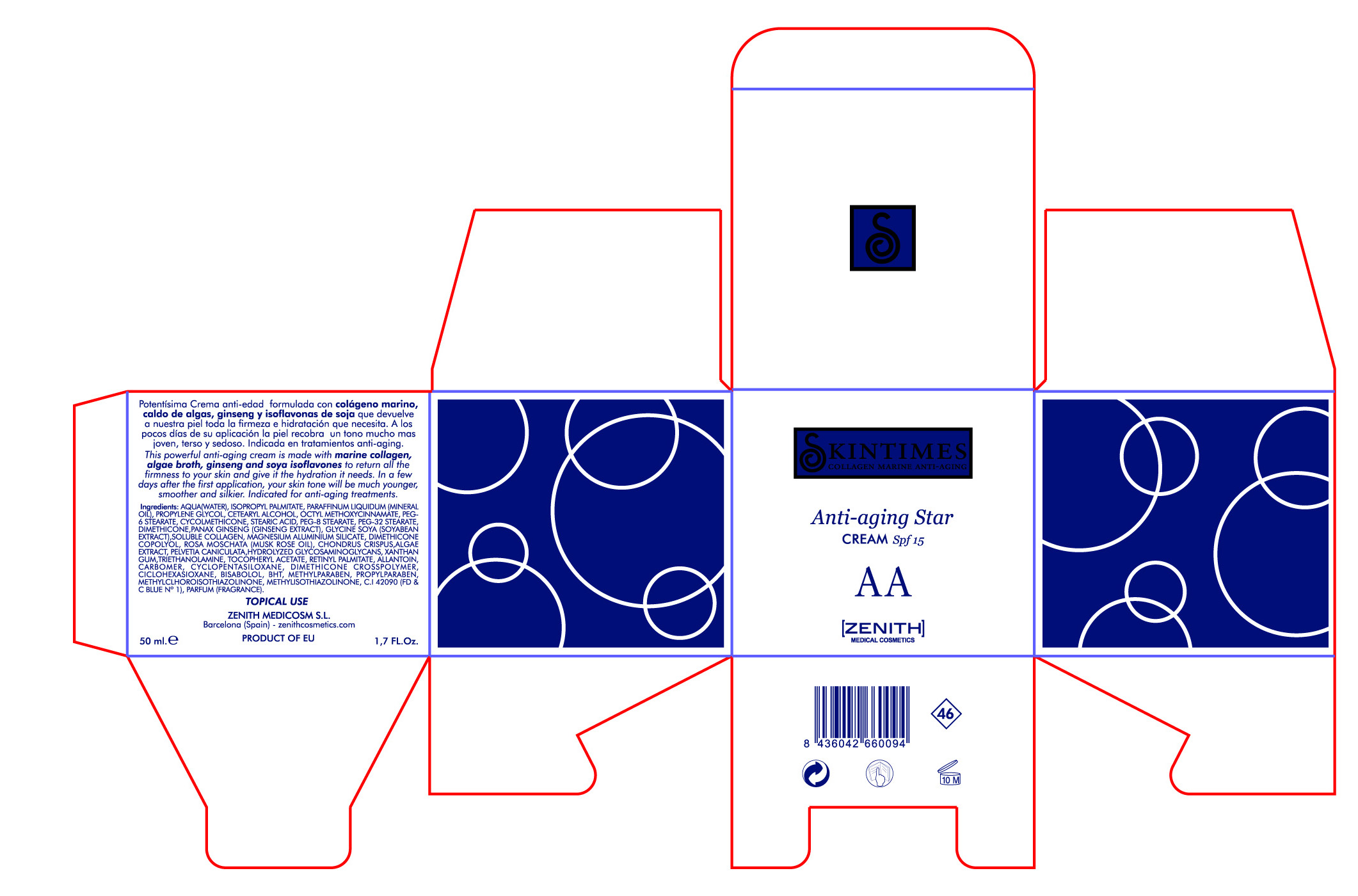
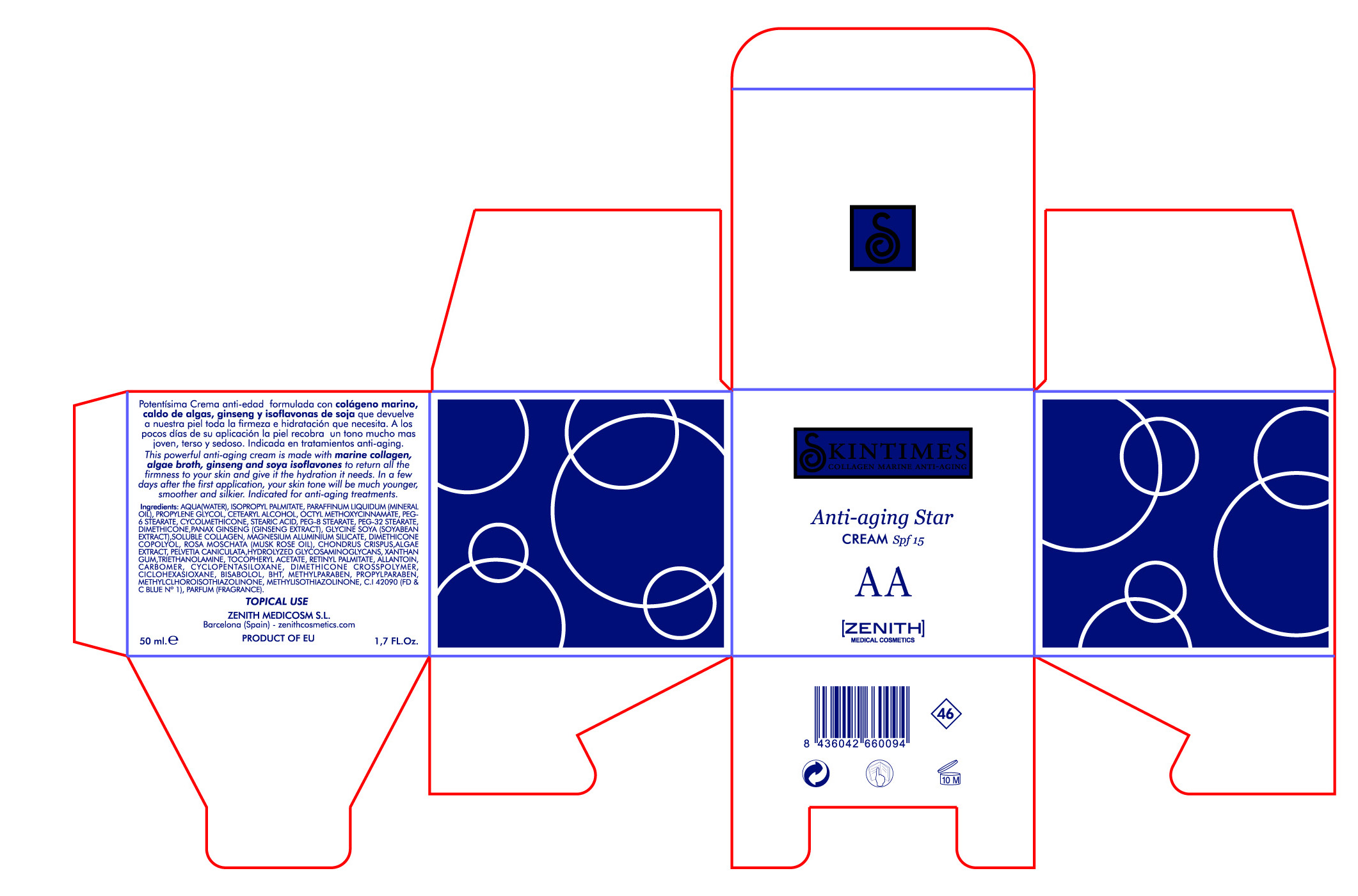
This powerful anti-aging cream is made with marine collagen, algae broth, ginseng and soya isoflavones to return all the firmness to your skin and give it the hydration it needs. In a few days after first application, your skin tone will be much younger, smoother and silkier.Indicated for anti-aging treatments.
50ml. 1.7 fl. Oz
- Warning
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ANTI AGING STAR
octinoxate creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:42248-102 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 3.75 mL in 50 mL Inactive Ingredients Ingredient Name Strength MAGNESIUM ALUMINUM SILICATE (UNII: 6M3P64V0NC) PROPYLPARABEN (UNII: Z8IX2SC1OH) ALPHA-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) DIMETHICONE (UNII: 92RU3N3Y1O) XANTHAN GUM (UNII: TTV12P4NEE) ROSA MOSCHATA OIL (UNII: J99W255AWF) WATER (UNII: 059QF0KO0R) MINERAL OIL (UNII: T5L8T28FGP) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) PEG-8 STEARATE (UNII: 2P9L47VI5E) TROLAMINE (UNII: 9O3K93S3TK) SOYBEAN (UNII: L7HT8F1ZOD) CHONDRUS CRISPUS (UNII: OQS23HUA1X) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) DIMETHICONE 20 (UNII: H8YMB5QY0D) CARBOMER 934 (UNII: Z135WT9208) HYDROLYZED GLYCOSAMINOGLYCANS (BOVINE; 50000 MW) (UNII: 997385V0VV) METHYLCHLOROISOTHIAZOLINONE (UNII: DEL7T5QRPN) METHYLPARABEN (UNII: A2I8C7HI9T) COLLAGEN, SOLUBLE, FISH SKIN (UNII: 8JC99XGU4W) ISOPROPYL PALMITATE (UNII: 8CRQ2TH63M) PEG-6 STEARATE (UNII: 8LQC57C6B0) PEG-32 STEARATE (UNII: 33GX5WQC0M) STEARIC ACID (UNII: 4ELV7Z65AP) PANAX GINSENG FRUIT (UNII: E35T4MOI3E) FUCUS VESICULOSUS (UNII: 535G2ABX9M) PELVETIA CANALICULATA (UNII: 8U1M44KESN) CYCLOMETHICONE (UNII: NMQ347994Z) VITAMIN A PALMITATE (UNII: 1D1K0N0VVC) ALLANTOIN (UNII: 344S277G0Z) CYCLOMETHICONE 5 (UNII: 0THT5PCI0R) LEVOMENOL (UNII: 24WE03BX2T) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) METHYLISOTHIAZOLINONE (UNII: 229D0E1QFA) CYCLOMETHICONE 6 (UNII: XHK3U310BA) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:42248-102-02 1 in 1 BOX 1 50 mL in 1 JAR Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part352 08/15/2011 Labeler - Zenith Medicosm SL (464239694) Registrant - Zenith Medicosm SL (464239694) Establishment Name Address ID/FEI Business Operations Zenith Medicosm SL 464239694 manufacture