Label: SULFAMETHOXAZOLE AND TRIMETHOPRIM suspension
-
Contains inactivated NDC Code(s)
NDC Code(s): 21695-701-00, 21695-701-16 - Packager: Rebel Distributors Corp
- This is a repackaged label.
- Source NDC Code(s): 50383-823
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated December 1, 2010
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
SPL UNCLASSIFIED SECTION
To reduce the development of drug-resistant bacteria and maintain the effectiveness of sulfamethoxazole and trimethoprim oral suspension and other antibacterial drugs, sulfamethoxazole and trimethoprim oral suspension should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria.
-
DESCRIPTION
Sulfamethoxazole and Trimethoprim Oral Suspension USP is a synthetic antibacterial combination product. Sulfamethoxazole is N1-(5-methyl-3-isoxazolyl) sulfanilamide. It is an almost white, odorless, tasteless compound. It has the following structural formula is:
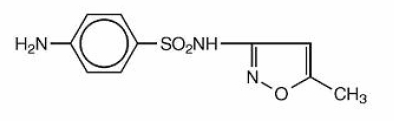
C10H11N3O3S M.W. 253.28
Trimethoprim is 2,4-diamino-5-(3,4,5- trimethoxybenzyl) pyrimidine. It is a white to light yellow, odorless, bitter compound. It has the following structural formula is:
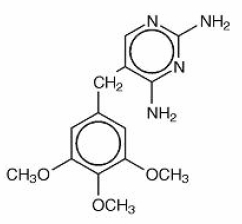
C14H18N4O3 M.W. 290.32
Each teaspoonful (5 mL) for oral administration, contains 200 mg sulfamethoxazole and 40 mg trimethoprim in a vehicle containing alcohol 0.26%, methylparaben 0.1% and sodium benzoate 0.1% (added as preservatives), carboxymethylcellulose sodium, citric acid (anhydrous), glycerin, microcrystalline cellulose, polysorbate 80, purified water, saccharin sodium, and sorbitol. The light purple, grape flavored suspension contains the following additional inactive ingredients: FD&C Red No. 40, FD&C Blue No. 1 and natural and artificial grape flavor. The pink, cherry flavored suspension contains the following additional inactive ingredients: FD&C Red No. 40, FD&C Yellow No. 6 and artificial cherry flavor.
-
CLINICAL PHARMACOLOGY
Sulfamethoxazole and trimethoprim oral suspension is rapidly absorbed following oral administration. Both sulfamethoxazole and trimethoprim exist in the blood as unbound, protein-bound, and metabolized forms; sulfamethoxazole also exists as the conjugated form. The metabolism of sulfamethoxazole occurs predominantly by N4-acetylation although the glucuronide conjugate has been identified. The principal metabolites of trimethoprim are the 1- and 3-oxides and the 3’- and 4’-hydroxy derivatives. The free forms of sulfamethoxazole and trimethoprim are considered to be the therapeutically active forms. Approximately 44% of trimethoprim and 70% of sulfamethoxazole are bound to plasma proteins. The presence of 10 mg percent sulfamethoxazole in plasma decreases the protein binding of trimethoprim by an insignificant degree; trimethoprim does not influence the protein binding of sulfamethoxazole. Peak blood levels for the individual components occur 1 to 4 hours after oral administration. The mean serum half-lives of sulfamethoxazole and trimethoprim are 10 and 8 to 10 hours, respectively. However, patients with severely impaired renal function exhibit an increase in the half-lives of both components, requiring dosage regimen adjustment (see DOSAGE AND ADMINISTRATION). Detectable amounts of trimethoprim and sulfamethoxazole are present in the blood 24 hours after drug administration. During administration of 160 mg trimethoprim and 800 mg sulfamethoxazole b.i.d., the mean steady-state plasma concentration of trimethoprim was 1.72 mcg/mL. The steady-state minimal plasma levels of free and total sulfamethoxazole were 57.4 mcg/mL and 68.0 mcg/mL, respectively. These steady-state levels were achieved after 3 days of drug administration.1
Excretion of sulfamethoxazole and trimethoprim is primarily by the kidneys through both glomerular filtration and tubular secretion. Urine concentrations of both sulfamethoxazole and trimethoprim are considerably higher than are the concentrations in the blood. The average percentage of the dose recovered in urine from 0 to 72 hours after a single oral dose is 84.5% for total sulfonamide and 66.8% for free trimethoprim. Thirty percent of the total sulfonamide is excreted as free sulfamethoxazole, with the remaining as N4-acetylated metabolite.2 When administered together, neither sulfamethoxazole nor trimethoprim affects the urinary excretion pattern of the other.
Both sulfamethoxazole and trimethoprim distribute to sputum, vaginal fluid, and middle ear fluid; trimethoprim also distributes to bronchial secretions, and both pass the placental barrier and are excreted in human milk.
Geriatric Pharmacokinetics
The pharmacokinetics of sulfamethoxazole 800 mg and trimethoprim 160 mg were studied in 6 geriatric subjects (mean age: 78.6 years) and 6 young healthy subjects (mean age: 29.3 years) using a non-U.S. approved formulation. Pharmacokinetic values for sulfamethoxazole in geriatric subjects were similar to those observed in young adult subjects. The mean renal clearance of trimethoprim was significantly lowered in geriatric subjects compared with young adult subjects (19 mL/h/kg vs. 55 mL/h/kg). However, after normalizing by body weight, the apparent total body clearance of trimethoprim was an average 19% lower in geriatric subjects compared with young adult subjects.3
Microbiology
Sulfamethoxazole inhibits bacterial synthesis of dihydrofolic acid by competing with para-aminobenzoic acid (PABA). Trimethoprim blocks the production of tetrahydrofolic acid from dihydrofolic acid by binding to and reversibly inhibiting the required enzyme, dihydrofolate reductase. Thus, this combination blocks two consecutive steps in the biosynthesis of nucleic acids and proteins essential to many bacteria.
In vitro studies have shown that bacterial resistance develops more slowly with this combination than with either sulfamethoxazole or trimethoprim alone.
In vitro serial dilution tests have shown that the spectrum of antibacterial activity of sulfamethoxazole and trimethoprim includes the common urinary tract pathogens with the exception of Pseudomonas aeruginosa. The following organisms are usually susceptible: Escherichia coli, Klebsiella species, Enterobacter species, Morganella morganii, Proteus mirabilis and indole-positive Proteus species including Proteus vulgaris.
The usual spectrum of antimicrobial activity of sulfamethoxazole and trimethoprim includes bacterial pathogens isolated from middle ear exudate and from bronchial secretions (Haemophilus influenzae, including ampicillin-resistant strains, and Streptococcus pneumoniae), enterotoxigenic strains of Escherichia coli (ETEC) causing bacterial gastroenteritis. Shigella flexneri and Shigella sonnei are also usually susceptible.
REPRESENTATIVE MINIMUM INHIBITORY CONCENTRATION VALUES FOR ORGANISMS SUSCEPTIBLE TO SULFAMETHOXAZOLE AND TRIMETHOPRIM (MIC - mcg/mL) - *
- *Rudoy RC, Nelson JD, Haltalin KC. Antimicrobial Agents and Chemotherapy. 1974;5:439-443.
Bacteria TMP/SMX (1:19) TMP Alone SMX Alone TMP SMX Escherichia coli 0.05 to 1.5 1.0 to 245 0.05 to 0.5 0.95 to 9.5 Escherichia coli (enterotoxigenic strains) 0.015 to 0.15 0.285 to > 950 0.005 to 0.15 0.095 to 2.85 Proteus species (indole positive) 0.5 to 5.0 7.35 to 300 0.05 to 1.5 0.95 to 28.5 Morganella morganii 0.5 to 5.0 7.35 to 300 0.05 to 1.5 0.95 to 28.5 Proteus mirabilis 0.5 to 1.5 7.35 to 30 0.05 to 0.15 0.95 to 2.85 Klebsiella species 0.15 to 5.0 2.45 to 245 0.05 to 1.5 0.95 to 28.5 Enterobacter species 0.15 to 5.0 2.45 to 245 0.05 to 1.5 0.95 to 28.5 Haemophilus influenzae 0.15 to 1.5 2.85 to 95 0.015 to 0.15 0.285 to 2.85 Streptococcus pneumoniae 0.15 to 1.5 7.35 to 24.5 0.05 to 0.15 0.95 to 2.85 Shigella flexneri** < 0.01 to 0.04 < 0.16 to > 320 < 0.002 to 0.03 0.04 to 0.625 Shigella sonnei** 0.02 to 0.08 0.625 to > 320 0.004 to 0.06 0.08 to 1.25 TMP = trimethoprim
SMX = sulfamethoxazole
Susceptibility Testing: The recommended quantitative disc susceptibility method may be used for estimating the susceptibility of bacteria to sulfamethoxazole and trimethoprim. 3, 4 With this procedure, a report from the laboratory of “Susceptible to trimethoprim and sulfamethoxazole” indicates that the infection is likely to respond to therapy with this product. If the infection is confined to the urine, a report of “Intermediate susceptibility to trimethoprim and sulfamethoxazole” also indicates that the infection is likely to respond. A report of “Resistant to trimethoprim and sulfamethoxazole” indicates that the infection is unlikely to respond to therapy with this product.
-
INDICATIONS AND USAGE
To reduce the development of drug-resistant bacteria and maintain the effectiveness of sulfamethoxazole and trimethoprim oral suspension and other antibacterial drugs, sulfamethoxazole and trimethoprim oral suspension should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
URINARY TRACT INFECTIONS
For the treatment of urinary tract infections due to susceptible strains of the following organisms: Escherichia coli, Klebsiella species, Enterobacter species, Morganella morganii, Proteus mirabilis, and Proteus vulgaris. It is recommended that initial episodes of uncomplicated urinary tract infections be treated with a single effective antibacterial agent rather than the combination.
ACUTE OTITIS MEDIA
For the treatment of acute otitis media in pediatric patients due to susceptible strains of Streptococcus pneumoniae or Haemophilus influenzae when, in the judgment of the physician, sulfamethoxazole and trimethoprim oral suspension offers some advantage over the use of other antimicrobial agents. To date, there are limited data on the safety of repeated use of sulfamethoxazole and trimethoprim in pediatric patients under two years of age. This product is not indicated for prophylactic or prolonged administration in otitis media at any age.
ACUTE EXACERBATIONS OF CHRONIC BRONCHITIS IN ADULTS
For the treatment of acute exacerbations of chronic bronchitis due to susceptible strains of Streptococcus pneumoniae or Haemophilus influenzae when, in the judgment of the physician, sulfamethoxazole and trimethoprim oral suspension offers some advantage over the use of a single antimicrobial agent.
TRAVELERS’ DIARRHEA IN ADULTS
For the treatment of travelers’ diarrhea due to susceptible strains of enterotoxigenic E. coli.
-
CONTRAINDICATIONS
Sulfamethoxazole and trimethoprim oral suspension is contraindicated in patients with a known hypersensitivity to trimethoprim or sulfonamides and in patients with documented megaloblastic anemia due to folate deficiency. Sulfamethoxazole and trimethoprim oral suspension is also contraindicated in pregnant patients at term and in nursing mothers, because sulfonamides pass the placenta and are excreted in the milk and may cause kernicterus. Sulfamethoxazole and trimethoprim oral suspension is contraindicated in pediatric patients less than two months of age.
-
WARNINGS
FATALITIES ASSOCIATED WITH THE ADMINISTRATION OF SULFONAMIDES, ALTHOUGH RARE, HAVE OCCURRED DUE TO SEVERE REACTIONS, INCLUDING STEVENS-JOHNSON SYNDROME, TOXIC EPIDERMAL NECROLYSIS, FULMINANT HEPATIC NECROSIS, AGRANULOCYTOSIS, APLASTIC ANEMIA, AND OTHER BLOOD DYSCRASIAS.
SULFONAMIDES, INCLUDING SULFONAMIDE-CONTAINING PRODUCTS SUCH AS SULFAMETHOXAZOLE/TRIMETHOPRIM SHOULD BE DISCONTINUED AT THE FIRST APPEARANCE OF SKIN RASH OR ANY SIGN OF ADVERSE REACTION. In rare instances, a skin rash may be followed by a more severe reaction, such as Stevens-Johnson syndrome, toxic epidermal necrolysis, hepatic necrosis and serious blood disorder (see PRECAUTIONS).
Clinical signs, such as rash, sore throat, fever, arthralgia, pallor, purpura, or jaundice may be early indications of serious reactions.
Cough, shortness of breath, and pulmonary infiltrates are hypersensitivity reactions of the respiratory tract that have been reported in association with sulfonamide treatment.
The sulfonamides should not be used for the treatment of group A beta-hemolytic streptococcal infections. In an established infection, they will not eradicate the streptococcus and, therefore, will not prevent sequelae such as rheumatic fever.
Clostridium difficile associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including sulfamethoxazole and trimethoprim oral suspension, USP, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibiotic use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antiobiotic use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibiotic treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
-
PRECAUTIONS
General
Prescribing sulfamethoxazole and trimethoprim oral suspension in the absence of a proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria. Sulfamethoxazole and trimethoprim oral suspension should be given with caution to patients with impaired renal or hepatic function, to those with possible folate deficiency (e.g., the elderly, chronic alcoholics, patients receiving anticonvulsant therapy, patients with malabsorption syndrome, and patients in malnutrition states), and to those with severe allergy or bronchial asthma. In glucose-6-phosphate dehydrogenase-deficient individuals, hemolysis may occur. This reaction is frequently dose-related (see CLINICAL PHARMACOLOGYand DOSAGE AND ADMINISTRATION).
Use in the Elderly
There may be an increased risk of severe adverse reactions in elderly patients, particularly when complicating conditions exist, e.g., impaired kidney and/or liver function, or concomitant use of other drugs. Severe skin reactions, generalized bone marrow suppression (see WARNINGS and ADVERSE REACTIONS ), or a specific decrease in platelets (with or without purpura) are the most frequently reported severe adverse reactions in elderly patients. In those concurrently receiving certain diuretics, primarily thiazides, an increased incidence of thrombocytopenia with purpura has been reported. Appropriate dosage adjustments should be made for patients with impaired kidney function (see DOSAGE AND ADMINISTRATION).
Use in the Treatment of and Prophylaxis for Pneumocystis carinii Pneumonia in Patients with Acquired Immunodeficiency Syndrome (AIDS)
The incidence of side effects, particularly rash, fever, leukopenia, and elevated aminotransferase (transaminase) values in AIDS patients who are being treated with sulfamethoxazole and trimethoprim therapy for Pneumocystis carinii pneumonia has been reported to be greatly increased compared with the incidence normally associated with the use of sulfamethoxazole and trimethoprim in non-AIDS patients. The incidence of hyperkalemia and hyponatremia appears to be increased in AIDS patients receiving sulfamethoxazole and trimethoprim. Adverse effects are generally less severe in patients receiving sulfamethoxazole and trimethoprim for prophylaxis. A history of mild intolerance to sulfamethoxazole and trimethoprim in AIDS patients does not appear to predict intolerance of subsequent secondary prophylaxis. However, if a patient develops skin rash or any sign of adverse reaction, therapy with sulfamethoxazole and trimethoprim should be re-evaluated (see WARNINGS).
The concomitant use of leucovorin with sulfamethoxazole/trimethoprim for the acute treatment of Pneumocystis carinii pneumonia in patients with HIV infection was associated with increased rates of treatment failure and morbidity in a placebo-controlled study.
Information for patients
Patients should be counseled that antibacterial drugs including sulfamethoxazole and trimethoprim oral suspension should only be used to treat bacterial infections. They do not treat viral infections (e.g., the common cold). When sulfamethoxazole and trimethoprim oral suspension is prescribed to treat a bacterial infection, patients should be told that although it is common to feel better early in the course of therapy, the medication should be taken exactly as directed. Skipping doses or not completing the full course of therapy may (1) decrease the effectiveness of the immediate treatment and (2) increase the likelihood that bacteria will develop resistance and will not be treatable by sulfamethoxazole and trimethoprim oral suspension or other antibaceterial drugs in the future.
Patients should be instructed to maintain an adequate fluid intake in order to prevent crystalluria and stone formation.
Diarrhea is a common problem caused by antibiotics which usually ends when the antibiotic is discontinued. Sometimes after starting treatment with antibiotics, patients can develop watery and bloody stools (with and without stomach cramps and fever) even as late as two or more months after having taken the last dose of the antibiotic. If this occurs, patients should contact their physician as soon as possible.
Laboratory tests
Complete blood counts should be done frequently in patients receiving sulfamethoxazole and trimethoprim; if a significant reduction in the count of any formed blood element is noted, sulfamethoxazole and trimethoprim oral suspension should be discontinued. Urinalyses with careful microscopic examination and renal function tests should be performed during therapy, particularly for those patients with impaired renal function.
Drug Interactions
In elderly patients concurrently receiving certain diuretics, primarily thiazides, an increased incidence of thrombocytopenia with purpura has been reported. In the literature, two cases of hyperkalemia in elderly patients have been reported after concomitant intake of sulfamethoxazole/trimethoprim and an angiotensin converting enzyme inhibitor.
It has been reported that sulfamethoxazole and trimethoprim may prolong the prothrombin time in patients who are receiving the anticoagulant warfarin. This interaction should be kept in mind when sulfamethoxazole and trimethoprim is given to patients already on anticoagulant therapy, and the coagulation time should be reassessed.
Sulfamethoxazole and trimethoprim may inhibit the hepatic metabolism of phenytoin. Sulfamethoxazole and trimethoprim, given at a common clinical dosage, increased the phenytoin half-life by 39% and decreased the phenytoin metabolic clearance rate by 27%. When administering these drugs concurrently, one should be alert for possible excessive phenytoin effect.
Sulfonamides can also displace methotrexate from plasma protein binding sites,thus increasing free methotrexate concentrations.
Drug/Laboratory Test Interactions
This combination product, specifically the trimethoprim component, can interfere with a serum methotrexate assay as determined by the competitive binding protein technique (CBPA) when a bacterial dihydrofolate reductase is used as the binding protein. No interference occurs, however, if methotrexate is measured by a radioimmunoassay (RIA).
The presence of trimethoprim and sulfamethoxazole may also interfere with the Jaffé alkaline picrate reaction assay for creatinine, resulting in overestimations of about 10% in the range of normal values.
Carcinogenesis, mutagenesis, impairment of fertility
Carcinogenesis
Long-term studies in animals to evaluate carcinogenic potential have not been conducted with sulfamethoxazole and trimethoprim.
Mutagenesis
Bacterial mutagenic studies have not been performed with sulfamethoxazole and trimethoprim in combination. Trimethoprim was demonstrated to be non-mutagenic in the Ames assay. In studies at two laboratories, no chromosomal damage was detected in cultured Chinese hamster ovary cells at concentration approximately 500 times human plasma levels; at concentrations approximately 1,000 times human plasma levels in these same cells, a low level of chromosomal damage was induced at one of these laboratories. No chromosomal abnormalities were observed in cultured human leukocytes at concentrations of trimethoprim up to 20 times human steady-state plasma levels. No chromosomal effects were detected in peripheral lymphocytes of human subjects receiving 320 mg of trimethoprim in combination with up to 1,600 mg of sulfamethoxazole per day for as long as 112 weeks.
Pregnancy
Teratogenic effects
Pregnancy Category C. In rats, oral doses of 533 mg/kg sulfamethoxazole or 200 mg/kg trimethoprim produced teratologic effects manifested mainly as cleft palates. The highest dose which did not cause cleft palates in rats was 512 mg/kg sulfamethoxazole or 192 mg/kg trimethoprim when administered separately. In two studies in rats, no teratogenicity was observed when 512 mg/kg of sulfamethoxazole was used in combination with 128 mg/kg of trimethoprim. In one study, however, cleft palates were observed in one litter out of 9 when 355 mg/kg of sulfamethoxazole was used in combination with 88 mg/kg of trimethoprim.
In some rabbit studies, an overall increase in fetal loss (dead and resorbed and malformed conceptuses) was associated with doses of trimethoprim six times the human therapeutic dose.
While there are no large, well-controlled studies in the use of trimethoprim and sulfamethoxazole in pregnant women, Brumfitt and Pursell,5 in a retrospective study, reported the outcome of 186 pregnancies during which the mother received either placebo or trimethoprim and sulfamethoxazole. The incidence of congenital abnormalities was 4.5% (3 of 66) in those who received placebo and 3.3% (4 of 120) in those receiving trimethoprim and sulfamethoxazole. There were no abnormalities in the 10 children whose mothers received the drug during the first trimester. In a separate survey, Brumfitt and Pursell also found no congenital abnormalities in 35 children whose mothers had received oral trimethoprim and sulfamethoxazole at the time of conception or shortly thereafter.
Because trimethoprim and sulfamethoxazole may interfere with folic acid metabolism, sulfamethoxazole and trimethoprim oral suspension should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Pediatric use
Sulfamethoxazole and trimethoprim oral suspension is not recommended for pediatric patients younger than 2 months of age (see INDICATIONS AND USAGE and CONTRAINDICATIONS).
Clinical studies of sulfamethoxazole and trimethoprim oral suspension did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects.
There may be an increased risk of severe adverse reactions in elderly patients, particularly when complicating conditions exist, e.g., impaired kidney and/or liver function, possible folate deficiency, or concomitant use of other drugs. Severe skin reactions, generalized bone marrow suppression (see WARNINGSand ADVERSE REACTIONS ), a specific decrease in platelets (with or without purpura), and hyperkalemia are the most frequently reported severe adverse reaction in elderly patients. In those concurrently receiving certain diuretics, primarily thiazides, an increased incidence of thrombocytopenia with purpura has been reported. Increased digoxin blood levels can occur with concomitant sulfamethoxazole and trimethoprim therapy, especially in elderly patients. Serum digoxin levels should be monitored. Hematological changes indicative of folic acid deficiency may occur in elderly patients. These effects are reversible by folinic acid therapy. Appropriate dosage adjustments should be made for patients with impaired kidney function and duration of use should be as short as possible to minimize risks of undesired reactions (see DOSAGE AND ADMINISTRATION). The trimethoprim component of this product may cause hyperkalemia when administered to patients with underlying disorders of potassium metabolism, with renal insufficiency, or when given concomitantly with drugs known to induce hyperkalemia, such as angiotensin converting enzyme inhibitors. Close monitoring of serum potassium is warranted in these patients. Discontinuation of sulfamethoxazole and trimethoprim treatment is recommended to help lower potassium serum levels.
Pharmacokinetics parameters for sulfamethoxazole were similar for geriatric subjects and younger adult subjects. The mean maximum serum trimethoprim concentration was higher and mean renal clearance of trimethoprim was lower in geriatric subjects compared with younger subjects3 (see CLINICAL PHARMACOLOGY, Geriatric Pharmacokinetics).
-
ADVERSE REACTIONS
The most common adverse effects are gastrointestinal disturbances (nausea, vomiting, anorexia) and allergic skin reactions (such as rash and urticaria). FATALITIES ASSOCIATED WITH THE ADMINISTRATION OF SULFONAMIDES, ALTHOUGH RARE, HAVE OCCURRED DUE TO SEVERE REACTIONS, INCLUDING STEVENS-JOHNSON SYNDROME, TOXIC EPIDERMAL NECROLYSIS, FULMINANT HEPATIC NECROSIS, AGRANULOCYTOSIS, APLASTIC ANEMIA, OTHER BLOOD DYSCRASIAS, AND HYPERSENSITIVITY OF THE RESPIRATORY TRACT (SEE WARNINGS ).
Hematologic
Agranulocytosis, aplastic anemia, thrombocytopenia, leukopenia, neutropenia, hemolytic anemia, megaloblastic anemia, hypoprothrombinemia, methemoglobinemia, eosinophilia.
Allergic
Stevens-Johnson syndrome, toxic epidermal necrolysis, anaphylaxis, allergic myocarditis, erythema multiforme, exfoliative dermatitis, angioedema, drug fever, chills, Henoch-Schönlein purpura, serum sickness-like syndrome, generalized allergic reactions, generalized skin eruptions, photosensitivity, conjunctival and scleral injection, pruritus, urticaria, and rash. In addition, periarteritis nodosa and systemic lupus erythematosus have been reported.
Gastrointestinal
Hepatitis, including cholestatic jaundice and hepatic necrosis, elevation of serum transaminase and bilirubin, pseudomembranous enterocolitis, pancreatitis, stomatitis, glossitis, nausea, emesis, abdominal pain, diarrhea, anorexia.
Genitourinary
Renal failure, interstitial nephritis, BUN and serum creatinine elevation, toxic nephrosis with oliguria and anuria, and crystalluria.
Neurologic
Aseptic meningitis, convulsions, peripheral neuritis, ataxia, vertigo, tinnitus, headache.
-
OVERDOSAGE
Acute
The amount of a single dose of sulfamethoxazole and trimethoprim that is either associated with symptoms of overdosage or is likely to be life-threatening has not been reported. Signs and symptoms of overdosage reported with sulfonamides include anorexia, colic, nausea, vomiting, dizziness, headache, drowsiness and unconsciousness. Pyrexia, hematuria, and crystalluria may be noted. Blood dyscrasias and jaundice are potential late manifestations of overdosage. Signs of acute overdosage with trimethoprim include nausea, vomiting, dizziness, headache, mental depression, confusion and bone marrow depression. General principles of treatment include the institution of gastric lavage or emesis; forcing oral fluids; and the administration of intravenous fluids if urine output is low and renal function is normal. Acidification of the urine will increase renal elimination of trimethoprim. The patient should be monitored with blood counts and appropriate blood chemistries, including electrolytes. If a significant blood dyscrasia or jaundice occurs, specific therapy should be instituted for these complications. Peritoneal dialysis is not effective and hemodialysis is only moderately effective in eliminating trimethoprim and sulfamethoxazole.
Chronic
Use of sulfamethoxazole and trimethoprim at high doses and/or for extended periods of time may cause bone marrow depression manifested as thrombocytopenia, leukopenia, and/or megaloblastic anemia. If signs of bone marrow depression occur, the patient should be given leucovorin; 5 to 15 mg leucovorin daily has been recommended by some investigators.
-
DOSAGE AND ADMINISTRATION
Contraindicated in pediatric patients less than 2 months of age.
Urinary Tract Infections and Shigellosis in Adults and Pediatric Patients and Acute Otitis Media in Pediatric Patients
Adults
The usual adult dosage in the treatment of urinary tract infections is four teaspoonfuls (20mL) Sulfamethoxazole and trimethoprim oral suspension every 12 hours for 10 to 14 days. An identical daily dosage is used for 5 days in treatment of shigellosis.
Pediatric Patients
The recommended dose for pediatric patients with urinary tract infections or acute otitis media is 40 mg/kg sulfamethoxazole and 8 mg/kg trimethoprim per 24 hours, given in two divided doses every 12 hours for 10 days. An identical daily dosage is used for 5 days in the treatment of shigellosis. The following table is a guideline for the attainment of this dosage:
Pediatric Patients Two months of age or older:
Weight Dose - Every 12 hours lb kg Teaspoonfuls 22 10 1 (5 mL) 44 20 2 (10 mL) 66 30 3 (15 mL) 88 40 4 (20 mL) For Patients with Impaired Renal Function: When renal function is impaired, a reduced dosage should be employed using the following table:
Creatinine Clearance (mL/min) Recommended Dosage Regimen Above 30 Use Standard Regimen 15 - 30 1/2 the Usual Regimen Below 15 Use Not Recommended Acute Exacerbations of Chronic Bronchitis in Adults
The usual adult dosage in the treatment of acute exacerbations of chronic bronchitis is four teaspoonfuls (20 mL) sulfamethoxazole and trimethoprim oral suspension every 12 hours for 14 days.
Travelers’ Diarrhea in Adults
For the treatment of travelers' diarrhea, the usual adult dosage is four teaspoonfuls (20mL) of sulfamethoxazole and trimethoprim oral suspension every 12 hours for 5 days.
Pneumocystis Carinii Pneumonia
Treatment
Adults and Pediatric Patients: The recommended dosage for treatment of patients with documented Pneumocystis carinii pneumonia is 75 to 100 mg/kg sulfamethoxazole and 15 to 20 mg/kg trimethoprim per 24 hours given in equally divided doses every 6 hours for 14 to 21 days. The following table is a guideline for the upper limit of this dosage:
Weight Dose - Every 6 Hours lb kg Teaspoonfuls 18 8 1 (5 mL) 35 16 2 (10 mL) 53 24 3 (15 mL) 70 32 4 (20 mL) 88 40 5 (25 mL) 106 48 6 (30 mL) 141 64 8 (40 mL) 176 80 10 (50 mL) For the lower limit dose (75 mg/kg sulfamethoxazole and 15 mg/kg trimethoprim per 24 hours) administer 75% of the dose in the above table.
Prophylaxis
Adults
The recommended dosage for prophylaxis in adults is four teaspoonfuls (20 mL) of the suspension daily.
Pediatric Patients
For pediatric patients, the recommended dose is 750 mg/m2/day sulfamethoxazole with 150 mg/m2/day trimethoprim given orally in equally divided doses twice a day, on 3 consecutive days per week. The total daily dose should not exceed 1,600 mg sulfamethoxazole and 320 mg trimethoprim. The following table is a guideline for the attainment of this dosage in pediatric patients:
Body Surface Area Dose - Every 12 Hours (m2) Teaspoonfuls 0.26 1/2 (2.5 mL) 0.53 1 (5 mL) 1.06 2 (10 mL) -
HOW SUPPLIED
Sulfamethoxazole and Trimethoprim Oral Suspension, USP is supplied in a purple grape-flavored suspension and in a pink cherry-flavored suspension containing 40 mg trimethoprim and 200 mg sulfamethoxazole per 5 mL (teaspoonful) both packaged in 1 pint (473 mL) bottles.
Store at 20°-25°C (68°-77°F) [see USP Controlled Room Temperature]. Protect from light.
Dispense in a tight, light-resistant container as defined in the USP, with a child-resistant closure (as required).
To report SUSPECTED ADVERSE REACTIONS, contact Hi-Tech Pharmacal Co., Inc. at 1-800-262-9010 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Rx Only
-
REFERENCES
- Kremers P, Duvivier J, Heusghem C. Pharmacokinetic Studies of Co-Trimoxazole in Man after Single and Repeated Doses. J Clin Pharmacol. 1974; 14:112-117.
- Kaplan SA, Weinfeld RE, Abruzzo CW, McFaden K, Jack ML, Weissman L. Pharmacokinetic Profile of Trimethoprim-Sulfamethoxazole in Man. J Infect Dis. 1973; 128 (Suppl): S547-S555.
- Varoquaux, O, et al. Antibiotic Susceptibility Discs; Certification Procedure. Federal Register. 1972; 37:20527-20529.
- Bauer AW, Kirby WMM, Sherris JC, Turck M. Antibiotic Susceptibility Testing by Standardized Single Disk Method. Am J Clin Pathol. 1966; 45:493-496.
- Brumfitt W, Pursell R. Trimethoprim-Sulfamethoxazole in the Treatment of Bacteriuria in Women. J Infect Dis. 1973; 128 (Suppl):S657-S663.
- Marinella MA. Trimethoprim-Induced Hyperkalemia: An Analysis of Reported Cases. Gerontology 45:209-212, 1999.
Manufactured by:
Hi-Tech Pharmacal Co., Inc.
Amityville, NY 11701
Rev. 823/824:05 10/09
CR #1115
Repackaged by:
Rebel Distributors Corp
Thousand Oaks, CA 91320
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
SULFAMETHOXAZOLE AND TRIMETHOPRIM
sulfamethoxazole and trimethoprim suspensionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:21695-701(NDC:50383-823) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SULFAMETHOXAZOLE (UNII: JE42381TNV) (SULFAMETHOXAZOLE - UNII:JE42381TNV) SULFAMETHOXAZOLE 200 mg in 5 mL TRIMETHOPRIM (UNII: AN164J8Y0X) (TRIMETHOPRIM - UNII:AN164J8Y0X) TRIMETHOPRIM 40 mg in 5 mL Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) METHYLPARABEN (UNII: A2I8C7HI9T) SODIUM BENZOATE (UNII: OJ245FE5EU) CARBOXYMETHYLCELLULOSE SODIUM (UNII: K679OBS311) ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) GLYCERIN (UNII: PDC6A3C0OX) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) POLYSORBATE 80 (UNII: 6OZP39ZG8H) WATER (UNII: 059QF0KO0R) SACCHARIN SODIUM (UNII: SB8ZUX40TY) SORBITOL (UNII: 506T60A25R) FD&C RED NO. 40 (UNII: WZB9127XOA) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) CHERRY (UNII: BUC5I9595W) Product Characteristics Color Score Shape Size Flavor CHERRY Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:21695-701-00 100 mL in 1 BOTTLE 2 NDC:21695-701-16 473 mL in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA074650 12/29/1997 Labeler - Rebel Distributors Corp (118802834) Establishment Name Address ID/FEI Business Operations Rebel Distributors Corp 118802834 RELABEL, REPACK


