Label: VITAMIN A D- lanolin, petrolatum ointment
- NDC Code(s): 21130-803-26
- Packager: Lucerne North America LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated August 28, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredients
- Purpose
- Uses
- Warnings
- When using this product
- Stop use and ask a doctor if
- Do not use on
- Keep out of reach of children
- Directions
- Other information
- Inactive ingredients
- SPL UNCLASSIFIED SECTION
-
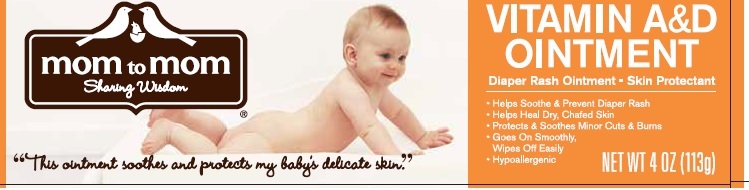
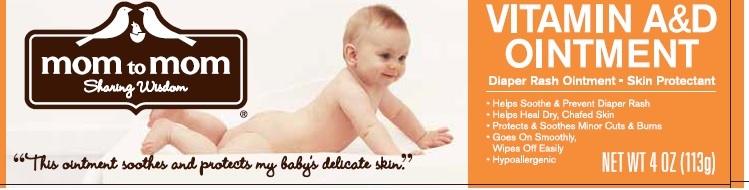
Principal Display Panel
mom to mom
Sharing Wisdom
"This ointment soothes and protects my baby's delicate skin."
VITAMIN A&D OINTMENT
Diaper Rash Ointment - Skin Protectant
- Helps Soothe & Prevent Diaper Rash
- Helps Heal Dry, Chafed Skin
- Protects & Soothes Minor Cuts & Burns
- Goes On Smoothly
- Wipes off Easily
Hypoallergenic
NET WT 4 OZ (113 g)
-
INGREDIENTS AND APPEARANCE
VITAMIN A D
lanolin, petrolatum ointmentProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:21130-803 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LANOLIN (UNII: 7EV65EAW6H) (LANOLIN - UNII:7EV65EAW6H) LANOLIN 133 mg in 1 g PETROLATUM (UNII: 4T6H12BN9U) (PETROLATUM - UNII:4T6H12BN9U) PETROLATUM 459 mg in 1 g Inactive Ingredients Ingredient Name Strength COD LIVER OIL (UNII: BBL281NWFG) LIGHT MINERAL OIL (UNII: N6K5787QVP) MICROCRYSTALLINE WAX (UNII: XOF597Q3KY) PARAFFIN (UNII: I9O0E3H2ZE) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:21130-803-26 1 in 1 CARTON 11/12/2012 1 113 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part347 11/12/2012 Labeler - Lucerne North America LLC (009137209) Registrant - Vi-Jon, LLC (790752542) Establishment Name Address ID/FEI Business Operations Vi-Jon, LLC 790752542 manufacture(21130-803) Establishment Name Address ID/FEI Business Operations Vi-Jon, LLC 088520668 manufacture(21130-803)