Label: RID LICE ELIMINATION ESSENTIALS- piperonyl butoxide, pyrethrum extract kit
-
Contains inactivated NDC Code(s)
NDC Code(s): 0280-9070-01 - Packager: Bayer HealthCare LLC.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated November 4, 2019
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- PURPOSE
- Uses
- WARNINGS
- DO NOT USE
- ASK DOCTOR
- WHEN USING
- STOP USE
- KEEP OUT OF REACH OF CHILDREN
-
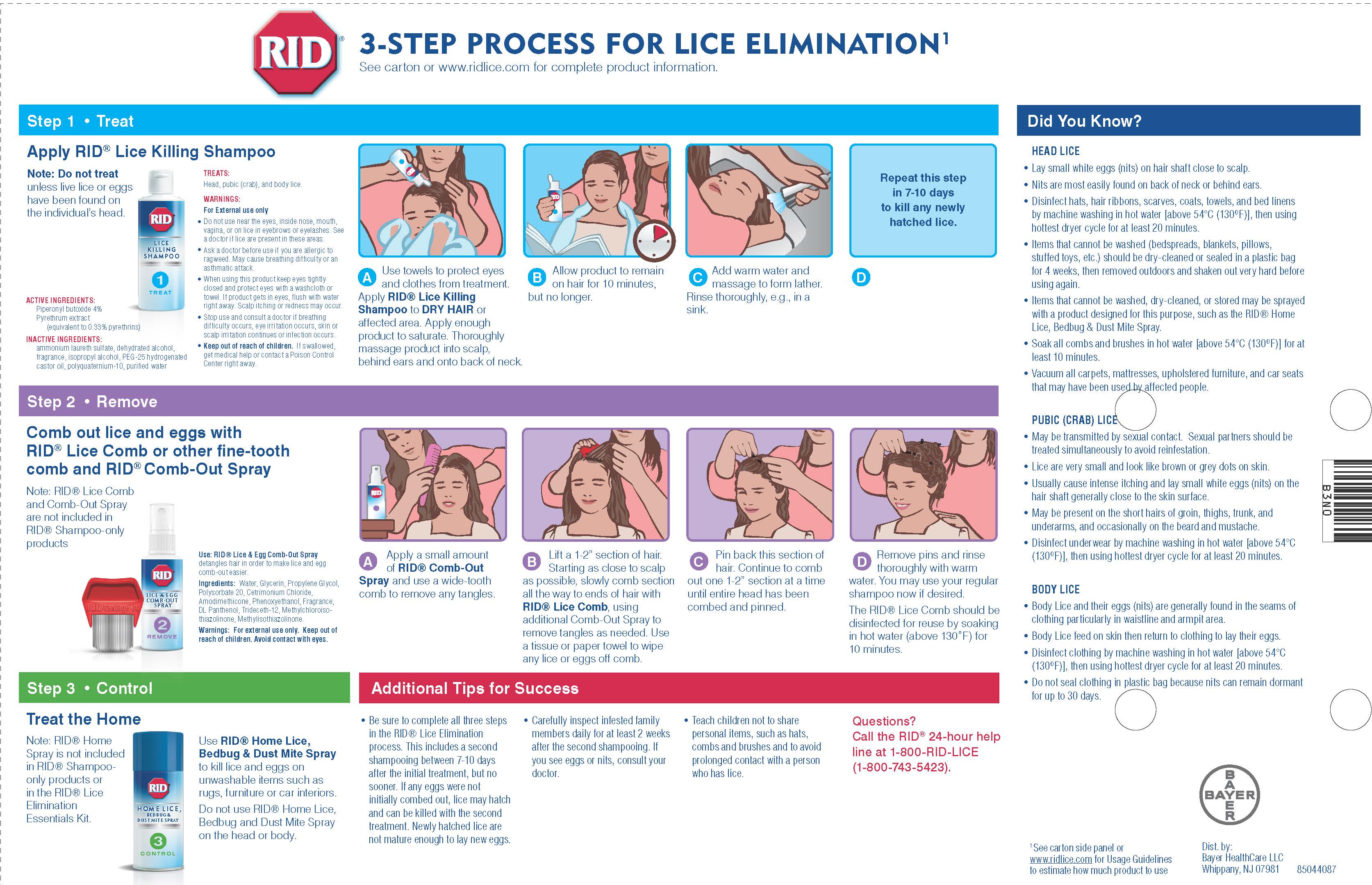
Directions
Directions
● Important: Read warnings before use
Adults and children 2 years and over:
Inspect
● check each household member with a magnifying glass in bright
light for lice/nits (eggs)
● look for tiny nits near scalp, beginning at back of neck and
behind ears
● examine small sections of hair at a time
● unlike dandruff which moves when touched, nits stick to the hair
● if either lice or nits are found, treat with this product
Treat
● apply thoroughly to DRY HAIR or other affected area. For head
lice, first apply behind ears and to back of neck.
● allow product to remain for 10 minutes, but no longer
● use warm water to form a lather, shampoo, then thoroughly
rinse
● for head lice, towel dry hair and comb out tangles
Remove lice and their eggs (nits)
● use a fine-tooth or special lice/nit comb. Remove any remaining
nits by hand (using a throw-away glove).
● hair should remain slightly damp while removing nits
● if hair dries during combing, dampen slightly with water
● for head lice, part hair into sections. Do one section at a time
starting on top of head. Longer hair may take 1 to 2 hours.
● lift a 1- to 2-inch wide strand of hair. Place comb as close to
scalp as possible and comb with a firm, even motion away from
scalp.
● pin back each strand of hair after combing
● clean comb often. Wipe nits away with tissue and discard in a
plastic bag. Seal bag and discard to prevent lice from coming
back.
● after combing, thoroughly recheck for lice/nits. Repeat combing
if necessary.
● check daily for any lice/nits that you missed
● a second treatment must be done in 7 to 10 days to kill any
newly hatched lice
● if infestation continues, see a doctor for other treatments
Children under 2 years: ask a doctor
- OTHER SAFETY INFORMATION
- INACTIVE INGREDIENT
- SPL UNCLASSIFIED SECTION
- Use
- WARNINGS
-
Directions
Directions For Use
● Before using, read Consumer Information Insert for complete directions.
● Use AFTER shampooing with RID® Lice Killing Shampoo.
● Towel dry hair. Apply a small amount of RID® Lice & Egg Comb-Out Spray and use a wide-toothed
comb to remove any tangles.
● Comb out the dead lice, eggs, and nits, one small section at a time, with the enclosed RIDvantage® Jr.
Lice Comb. (See insert for additional combing instructions). This step is very important.
● Use additional RID® Lice & Egg Comb-Out Spray to remove tangles, if needed, during the comb-out
process.
● After you have combed entire head, rinse hair thoroughly with warm water. Disinfect combs with hot
water (130ºF).
-
Other Information
Other Information
● It is important to wash in hot water (130ºF) all clothing, bedding, towels, and hair products (combs,
brushes) used by infested persons in the two days prior to treatment.
● Dry clean non-washable fabrics.
● To eliminate infestation of furniture and bedding that cannot be washed or dry cleaned, a multi-use lice
spray such as RID® Home Lice, Bedbug & Dust Mite Spray may be used.
● RID® Lice & Egg Comb-Out Spray does not kill lice or their eggs. Use this product only after treating
with RID® Lice Killing Shampoo.
● Store in a cool, dry place. Keep from freezing.
- Ingredients
- QUESTIONS
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
RID LICE ELIMINATION ESSENTIALS
piperonyl butoxide, pyrethrum extract kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:0280-9070 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0280-9070-01 1 in 1 CARTON; Type 0: Not a Combination Product 09/28/2016 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 BOTTLE 118 mL Part 2 1 BOTTLE, SPRAY 59 mL Part 1 of 2 RID LICE KILLING
iperonyl butoxide, pyrethrum extract shampooProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PYRETHRUM EXTRACT (UNII: ZUM06L90GV) (PYRETHRUM EXTRACT - UNII:ZUM06L90GV) PYRETHRUM EXTRACT 3.3 mg in 1 mL PIPERONYL BUTOXIDE (UNII: LWK91TU9AH) (PIPERONYL BUTOXIDE - UNII:LWK91TU9AH) PIPERONYL BUTOXIDE 40 mg in 1 mL Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) ISOPROPYL ALCOHOL (UNII: ND2M416302) POLYQUATERNIUM-10 (1000 MPA.S AT 2%) (UNII: GMR4PEN8PK) AMMONIUM LAURETH-2 SULFATE (UNII: 698O4Z48G6) HYDROGENATED CASTOR OIL (UNII: ZF94AP8MEY) WATER (UNII: 059QF0KO0R) Product Characteristics Color white (Clear) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 118 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part358G 09/28/2016 Part 2 of 2 RID LICE AND EGG COMB OUT
tonics, dressings, and other hair grooming aids sprayProduct Information Route of Administration TOPICAL Other Ingredients Ingredient Kind Ingredient Name Quantity INGR PROPYLENE GLYCOL (UNII: 6DC9Q167V3) INGR DISODIUM SEC-TRIDECETH-12 SULFOSUCCINATE (UNII: 3C19U43ACC) INGR WATER (UNII: 059QF0KO0R) INGR GLYCERIN (UNII: PDC6A3C0OX) INGR POLYSORBATE 20 (UNII: 7T1F30V5YH) INGR CETRIMONIUM CHLORIDE (UNII: UC9PE95IBP) INGR AMODIMETHICONE (1300 CST) (UNII: 3V7U636DWN) INGR METHYLISOTHIAZOLINONE (UNII: 229D0E1QFA) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 59 mL in 1 BOTTLE, SPRAY; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date Cosmetic 09/23/2016 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part358G 09/28/2016 Labeler - Bayer HealthCare LLC. (112117283)