Label: SELZENTRY- maraviroc tablet, film coated
- NDC Code(s): 50090-6238-0
- Packager: A-S Medication Solutions
- This is a repackaged label.
- Source NDC Code(s): 49702-223
- Category: HUMAN PRESCRIPTION DRUG LABEL
Drug Label Information
Updated November 10, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Medication Guide: HTML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use SELZENTRY safely and effectively. See full prescribing information for SELZENTRY.
SELZENTRY (maraviroc) tablets, for oral use
SELZENTRY (maraviroc) oral solution
Initial U.S. Approval: 2007WARNING: HEPATOTOXICITY
See full prescribing information for complete boxed warning.
INDICATIONS AND USAGE
SELZENTRY is a CCR5 co-receptor antagonist indicated in combination with other antiretroviral agents for the treatment of only CCR5-tropic HIV-1 infection in adults and pediatric patients weighing at least 2 kg. (1)
Limitations of Use:
- •
- Not recommended in patients with dual/mixed- or CXCR4-tropic HIV-1. (1)
DOSAGE AND ADMINISTRATION
- •
- Prior to initiation of SELZENTRY for treatment of HIV-1 infection, test all patients for CCR5 tropism using a highly sensitive tropism assay. (2.1)
- •
- SELZENTRY tablets and oral solution are taken twice daily by mouth and may be taken with or without food. SELZENTRY must be given in combination with other antiretroviral medications. (2.2)
Recommended Dosage in Adult Patients: (2.3)
Concomitant Medications
Dosage of SELZENTRY
When given with potent cytochrome P450 (CYP)3A inhibitors (with or without potent CYP3A inducers) including PIs (except tipranavir/ritonavir) (2.3, 7.1)
150 mg
twice daily
With NRTIs, tipranavir/ritonavir, nevirapine, raltegravir, and other drugs that are not potent CYP3A inhibitors or CYP3A inducers (2.3, 7.1)
300 mg
twice daily
With potent and moderate CYP3A inducers including efavirenz (without a potent CYP3A inhibitor) (2.3, 7.1)
600 mg
twice daily
A more complete list of coadministered drugs is listed in Dosage and Administration. (2)
Recommended Dosage in Pediatric Patients Weighing at Least 2 kg: Administer twice daily. Dosage should be based on body weight (kg) and concomitant medications and should not exceed the recommended adult dose. (2.4)
Recommended Dosage in Patients with Renal Impairment: Dose adjustment may be necessary in adult patients with renal impairment. (2.5)
DOSAGE FORMS AND STRENGTHS
CONTRAINDICATIONS
SELZENTRY is contraindicated in patients with severe renal impairment or end-stage renal disease (ESRD) (creatinine clearance [CrCl] less than 30 mL per minute) who are concomitantly taking potent CYP3A inhibitors or inducers. (4)
WARNINGS AND PRECAUTIONS
- •
- Hepatotoxicity accompanied by severe rash or systemic allergic reaction, including potentially life-threatening events, has been reported. Hepatic laboratory parameters including alanine aminotransferase (ALT), aspartate aminotransferase (AST), and bilirubin should be obtained prior to starting SELZENTRY and at other time points during treatment as clinically indicated. If rash or symptoms or signs of hepatitis or allergic reaction develop, hepatic laboratory parameters should be monitored and discontinuation of treatment should be considered. When administering SELZENTRY to patients with pre-existing liver dysfunction or who are co-infected with hepatitis B and/or C virus, additional monitoring may be warranted. (5.1)
- •
- Severe and potentially life-threatening skin and hypersensitivity reactions have been reported in patients taking SELZENTRY. This includes cases of Stevens-Johnson syndrome, hypersensitivity reaction, and toxic epidermal necrolysis. Immediately discontinue SELZENTRY and other suspected agents if signs or symptoms of severe skin or hypersensitivity reactions develop and monitor clinical status, including liver aminotransferases, closely. (5.2)
- •
- More cardiovascular events, including myocardial ischemia and/or infarction, were observed in treatment-experienced subjects who received SELZENTRY. Additional monitoring may be warranted. (5.3)
- •
- If patients with severe renal impairment or ESRD receiving SELZENTRY (without concomitant CYP3A inducers or inhibitors) experience postural hypotension, the dose of SELZENTRY should be reduced from 300 mg twice daily to 150 mg twice daily. (5.3)
ADVERSE REACTIONS
- •
- The most common adverse events in treatment-experienced adult subjects (greater than 8% incidence) which occurred at a higher frequency compared with placebo are upper respiratory tract infections, cough, pyrexia, rash, and dizziness. (6.1)
- •
- The most common adverse events in treatment-naive adult subjects (greater than 8% incidence) which occurred at a higher frequency than the comparator arm are upper respiratory tract infections, bronchitis, flatulence, bloating and distention, upper respiratory tract signs and symptoms, and gastrointestinal atonic and hypomotility disorders. (6.1)
- •
- The most common adverse reactions in treatment-experienced pediatric subjects (greater than or equal to 3% incidence) are vomiting, abdominal pain, diarrhea, nausea, and dizziness. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact ViiV Healthcare at 1-877-844-8872 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
- •
- Coadministration with CYP3A inhibitors, including protease inhibitors (except tipranavir/ritonavir), will increase the concentration of SELZENTRY. (7.1)
- •
- Coadministration with CYP3A inducers, including efavirenz, may decrease the concentration of SELZENTRY. (7.1)
- •
- Coadministration with St. John’s wort is not recommended. (7.1)
USE IN SPECIFIC POPULATIONS
- •
- Lactation: Women infected with HIV should be instructed not to breastfeed due to the potential for HIV transmission. (8.2)
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 11/2023
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
WARNING: HEPATOTOXICITY
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Testing prior to Initiation of SELZENTRY
2.2 General Dosing Recommendations
2.3 Recommended Dosage in Adult Patients with Normal Renal Function
2.4 Recommended Dosage in Pediatric Patients with Normal Renal Function
2.5 Recommended Dosage in Patients with Renal Impairment
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Hepatotoxicity
5.2 Severe Skin and Hypersensitivity Reactions
5.3 Cardiovascular Events
5.4 Immune Reconstitution Syndrome
5.5 Potential Risk of Infection
5.6 Potential Risk of Malignancy
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 Effect of Concomitant Drugs on the Pharmacokinetics of Maraviroc
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Impairment
8.7 Hepatic Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
12.4 Microbiology
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
14.1 Clinical Studies in Adult Subjects
14.2 Clinical Studies in Pediatric Subjects
15 REFERENCES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
-
BOXED WARNING
(What is this?)
WARNING: HEPATOTOXICITY
Hepatotoxicity has been reported with use of SELZENTRY. Severe rash or evidence of a systemic allergic reaction (e.g., fever, eosinophilia, or elevated IgE) prior to the development of hepatotoxicity may occur. Patients with signs or symptoms of hepatitis or allergic reaction following use of SELZENTRY should be evaluated immediately [see Warnings and Precautions (5.1)].
-
1 INDICATIONS AND USAGE
SELZENTRY is indicated in combination with other antiretroviral agents for the treatment of only CCR5‑tropic human immunodeficiency virus type 1 (HIV‑1) infection in adult and pediatric patients weighing at least 2 kg.
Limitations of Use
- •
- SELZENTRY is not recommended in patients with dual/mixed- or CXCR4-tropic HIV-1 [see Microbiology (12.4)].
-
2 DOSAGE AND ADMINISTRATION
2.1 Testing prior to Initiation of SELZENTRY
Prior to initiation of SELZENTRY for treatment of HIV-1 infection, test all patients for CCR5 tropism using a highly sensitive tropism assay. SELZENTRY is recommended for patients with only CCR5-tropic HIV-1 infection. Outgrowth of pre-existing low-level CXCR4- or dual/mixed-tropic HIV-1 not detected by tropism testing at screening has been associated with virologic failure on SELZENTRY [see Microbiology (12.4), Clinical Studies (14.1)].
Monitor patients for alanine aminotransferase (ALT), aspartate aminotransferase (AST), and bilirubin prior to initiation of SELZENTRY and at other time points during treatment as clinically indicated [see Warnings and Precautions (5.1)].
2.2 General Dosing Recommendations
- •
- SELZENTRY tablets and oral solution are taken twice daily by mouth and may be taken with or without food.
- •
- SELZENTRY must be given in combination with other antiretroviral medications.
- •
- The recommended dosage of SELZENTRY differs based on concomitant medications due to drug interactions.
2.3 Recommended Dosage in Adult Patients with Normal Renal Function
Table 1 displays oral dosage of SELZENTRY based on different concomitant medications [see Drug Interactions (7.1)].
Table 1. Recommended Dosage in Adults Concomitant Medications Dosage of SELZENTRY a Potent CYP3A inhibitors (with or without a potent CYP3A inducer) including: clarithromycin, cobicistat, elvitegravir/ritonavir, itraconazole, ketoconazole, nefazodone, protease inhibitors (except tipranavir/ritonavir), telithromycin.
b Noninteracting concomitant medications include all medications that are not potent CYP3A inhibitors or inducers such as: dolutegravir, enfuvirtide, nevirapine, all nucleoside reverse transcriptase inhibitors (NRTIs), raltegravir, and tipranavir/ritonavir.
c Potent and moderate CYP3A inducers (without a potent CYP3A inhibitor) including: carbamazepine, efavirenz, etravirine, phenobarbital, phenytoin, and rifampin.Potent cytochrome P450 (CYP)3A inhibitors (with or without a potent CYP3A inducer)a
150 mg twice daily
Noninteracting concomitant medicationsb
300 mg twice daily
Potent and moderate CYP3A inducers (without a potent CYP3A inhibitor)c
600 mg twice daily
2.4 Recommended Dosage in Pediatric Patients with Normal Renal Function
The recommended dosage of SELZENTRY should be based on body weight (kg) and should not exceed the recommended adult dose. The recommended dosage also differs based on concomitant medications due to drug interactions (Table 2 and Table 3) [see Drug Interactions (7.1), Use in Specific Populations (8.4)].
Before prescribing SELZENTRY tablets, assess children for the ability to swallow tablets. If a child is unable to reliably swallow SELZENTRY tablets, the oral solution formulation should be prescribed.
The recommended oral dosage of SELZENTRY tablets in pediatric patients aged 2 years and older weighing at least 10 kg is presented in Table 2.
Table 2. Recommended Dosage in Pediatric Patients Aged 2 Years and Older Weighing at Least 10 kg (Tablets) a Potent CYP3A inhibitors (with or without a CYP3A inducer) including: clarithromycin, cobicistat, elvitegravir/ritonavir, itraconazole, ketoconazole, nefazodone, protease inhibitors (except tipranavir/ritonavir), telithromycin.
b Noninteracting concomitant medications including all medications that are not potent CYP3A inhibitors or inducers such as: dolutegravir, enfuvirtide, nevirapine, all NRTIs, raltegravir, and tipranavir/ritonavir.
c Potent and moderate CYP3A inducers (without a potent CYP3A inhibitor) including: carbamazepine, efavirenz, etravirine, phenobarbital, phenytoin, and rifampin.
d Insufficient data are available to recommend use.Concomitant Medications
Dosage of SELZENTRY Based on Weight
10 kg to <14 kg
14 kg to <20 kg
20 kg to <30 kg
30 kg to <40 kg
≥40 kg
Potent CYP3A inhibitors (with or without a CYP3A inducer)a
50 mg
twice daily50 mg
twice daily75 mg
twice daily100 mg
twice daily150 mg
twice dailyNoninteracting concomitant medicationsb
150 mg twice daily
200 mg
twice daily200 mg
twice daily300 mg
twice daily300 mg
twice dailyPotent and moderate CYP3A inducers (without a potent CYP3A inhibitor)c
Not recommendedd
The recommended oral dosage of SELZENTRY oral solution in pediatric patients weighing at least 2 kg is presented in Table 3.
Table 3. Recommended Dosage in Pediatric Patients Weighing at Least 2 kg (Oral Solution) a Potent CYP3A inhibitors (with or without a CYP3A inducer) including: clarithromycin, cobicistat, elvitegravir/ritonavir, itraconazole, ketoconazole, nefazodone, protease inhibitors (except tipranavir/ritonavir), telithromycin.
b Insufficient data are available to recommend use.
c Noninteracting concomitant medications including all medications that are not potent CYP3A inhibitors or inducers such as: dolutegravir, enfuvirtide, nevirapine, all NRTIs, raltegravir, and tipranavir/ritonavir.
d Potent and moderate CYP3A inducers (without a potent CYP3A inhibitor) including: carbamazepine, efavirenz, etravirine, phenobarbital, phenytoin, and rifampin.Concomitant Medications
Dosage (Volume of Solution) of SELZENTRY
Based on Weight2 kg to
<4 kg4 kg to
<6 kg6 kg to
<10 kg
10 kg to
<14 kg
14 kg to <20 kg
20 kg to <30 kg
30 kg to <40 kg
≥40 kg
Potent CYP3A inhibitors (with or without a CYP3A inducer)a
Not recommendedb
50 mg
(2.5 mL)
twice daily50 mg
(2.5 mL)
twice daily80 mg
(4 mL)
twice daily100 mg
(5 mL)
twice daily150 mg
(7.5 mL)
twice dailyNoninteracting concomitant medicationsc
30 mg
(1.5 mL)
twice daily40 mg
(2 mL)
twice daily100 mg
(5 mL)
twice daily150 mg
(7.5 mL)
twice daily200 mg
(10 mL)
twice daily200 mg
(10 mL)
twice daily300 mg
(15 mL)
twice daily300 mg
(15 mL)
twice dailyPotent and moderate CYP3A inducers (without a potent CYP3A inhibitor)d
Not recommendedb
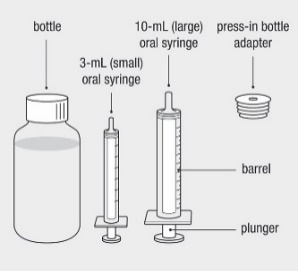
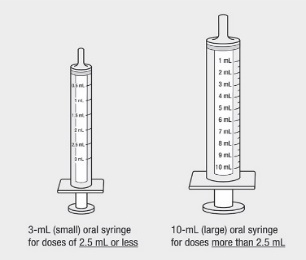
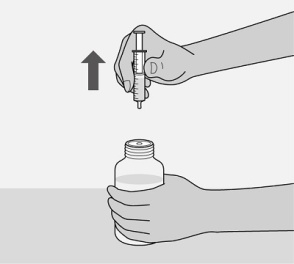
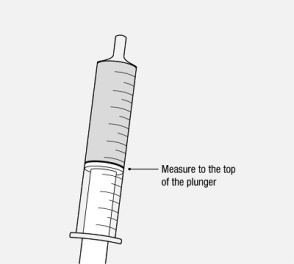
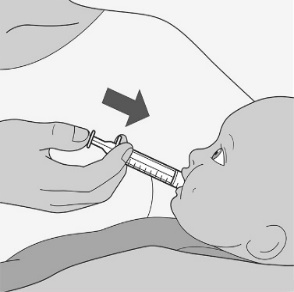
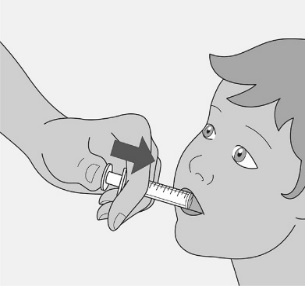
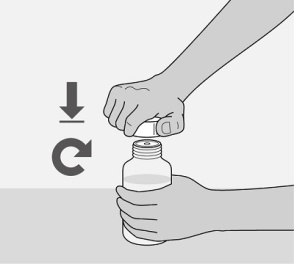
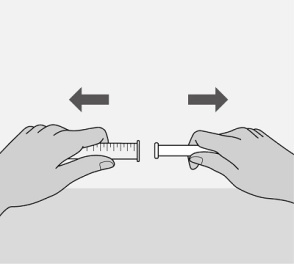
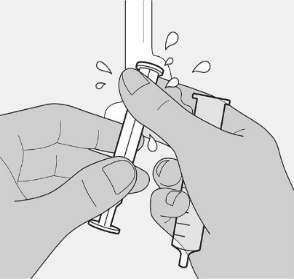
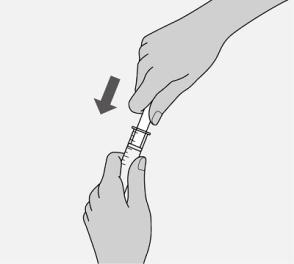
Administer the oral solution using the included press-in bottle adapter and the appropriate oral dosing syringe: for doses of 2.5 mL or less, use the 3-mL syringe; for doses greater than 2.5 mL, use the 10-mL syringe.
Care should be taken when measuring neonate doses due to the small volumes of oral solution required.
2.5 Recommended Dosage in Patients with Renal Impairment
Adult Patients
Table 4 provides dosing recommendations for patients based on renal function and concomitant medications.
Table 4. Recommended Dosage in Adults Based on Renal Function CrCl = Creatinine clearance.
a Potent CYP3A inhibitors (with or without a CYP3A inducer) including: clarithromycin, cobicistat, elvitegravir/ritonavir, itraconazole, ketoconazole, nefazodone, protease inhibitors (except tipranavir/ritonavir), telithromycin.
b Noninteracting concomitant medications include all medications that are not potent CYP3A inhibitors or inducers such as: dolutegravir, enfuvirtide, nevirapine, all NRTIs, raltegravir, and tipranavir/ritonavir.
c Dosage of SELZENTRY should be reduced to 150 mg twice daily if there are any symptoms of postural hypotension [see Contraindications (4), Warnings and Precautions (5.3)].
d Potent and moderate CYP3A inducers (without a potent CYP3A inhibitor) including: carbamazepine, efavirenz, etravirine, phenobarbital, phenytoin, and rifampin.Concomitant Medications
Dosage of SELZENTRY Based on Renal Function
Normal
(CrCl >80 mL/min)
Mild
(CrCl >50 and ≤80 mL/min)
Moderate
(CrCl ≥30 and ≤50 mL/min)
Severe
(CrCl <30 mL/min)
End-Stage Renal Disease on Regular Hemodialysis
Potent CYP3A inhibitors (with or without a CYP3A inducer)a
150 mg twice daily
150 mg twice daily
150 mg twice daily
Contra-indicated
Contra-indicated
Noninteracting concomitant medicationsb
300 mg twice daily
300 mg twice daily
300 mg twice daily
300 mg twice daily
300 mg twice dailyc
Potent and moderate CYP3A inducers (without a potent CYP3A inhibitor)d
600 mg twice daily
600 mg twice daily
600 mg twice daily
Contra-indicated
Contra-indicated
Pediatric Patients
There are no data to recommend specific doses of SELZENTRY in pediatric patients with mild or moderate renal impairment [see Use in Specific Populations (8.6)]. Additionally, SELZENTRY is contraindicated for pediatric patients with severe renal impairment or end-stage renal disease (ESRD) on regular hemodialysis who are receiving potent CYP3A inhibitors or inducers [see Contraindications (4)].
-
3 DOSAGE FORMS AND STRENGTHS
Tablets:
- •
- 25-mg blue, oval, film-coated tablets debossed with “MVC 25” on one side and plain on the other.
- •
- 75-mg blue, oval, film-coated tablets debossed with “MVC 75” on one side and plain on the other.
- •
- 150-mg blue, oval, film-coated tablets debossed with “MVC 150” on one side and plain on the other.
- •
- 300-mg blue, oval, film-coated tablets debossed with “MVC 300” on one side and plain on the other.
Oral Solution:
- •
- 20 mg per mL clear, colorless, strawberry-flavored oral solution.
-
4 CONTRAINDICATIONS
SELZENTRY is contraindicated in patients with severe renal impairment or ESRD (creatinine clearance [CrCl] less than 30 mL per minute) who are concomitantly taking potent CYP3A inhibitors or inducers [see Warnings and Precautions (5.3)].
-
5 WARNINGS AND PRECAUTIONS
5.1 Hepatotoxicity
Hepatotoxicity with allergic features including life-threatening events has been reported in clinical trials and postmarketing. Severe rash or evidence of systemic allergic reaction including drug-related rash with fever, eosinophilia, elevated IgE, or other systemic symptoms have been reported in conjunction with hepatotoxicity [see Warnings and Precautions (5.2)]. These events occurred approximately 1 month after starting treatment. Among reported cases of hepatitis, some were observed in the absence of allergic features or with no pre-existing hepatic disease.
Appropriate laboratory testing including ALT, AST, and bilirubin should be conducted prior to initiating therapy with SELZENTRY and at other time points during treatment as clinically indicated. Hepatic laboratory parameters should be obtained in any patient who develops rash, or signs or symptoms of hepatitis, or allergic reaction. Discontinuation of SELZENTRY should be considered in any patient with signs or symptoms of hepatitis, or with increased liver transaminases combined with rash or other systemic symptoms.
When administering SELZENTRY to patients with pre-existing liver dysfunction or who are co-infected with hepatitis B and/or C virus, additional monitoring may be warranted. The safety and efficacy of SELZENTRY have not been specifically studied in patients with significant underlying liver disorders.
5.2 Severe Skin and Hypersensitivity Reactions
Severe, potentially life-threatening skin and hypersensitivity reactions have been reported in patients taking SELZENTRY, in most cases concomitantly with other drugs associated with these reactions. These include cases of Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), and drug rash with eosinophilia and systemic symptoms (DRESS) [see Adverse Reactions (6.2)]. The cases were characterized by features including rash, constitutional findings, and sometimes organ dysfunction, including hepatic failure. Discontinue SELZENTRY and other suspected agents immediately if signs or symptoms of severe skin or hypersensitivity reactions develop (including, but not limited to, severe rash or rash accompanied by fever, malaise, muscle or joint aches, blisters, oral lesions, conjunctivitis, facial edema, lip swelling, eosinophilia). Delay in stopping treatment with SELZENTRY or other suspect drugs after the onset of rash may result in a life-threatening reaction. Clinical status, including liver aminotransferases, should be monitored and appropriate therapy initiated.
5.3 Cardiovascular Events
Eleven subjects (1.3%) who received SELZENTRY had cardiovascular events, including myocardial ischemia and/or infarction, during the Phase 3 trials in treatment‑experienced subjects (total exposure 609 patient‑years [300 on SELZENTRY once daily + 309 on SELZENTRY twice daily]), while no subjects who received placebo had such events (total exposure 111 patient‑years). These subjects generally had cardiac disease or cardiac risk factors prior to use of SELZENTRY, and the relative contribution of SELZENTRY to these events is not known.
In the Phase 2b/3 trial in treatment‑naive adult subjects, 3 subjects (0.8%) who received SELZENTRY had events related to ischemic heart disease and 5 subjects (1.4%) who received efavirenz had such events (total exposure 506 and 508 patient‑years for SELZENTRY and efavirenz, respectively).
When SELZENTRY was administered to healthy volunteers at doses higher than the recommended dose, symptomatic postural hypotension was seen at a greater frequency than in placebo. However, when SELZENTRY was given at the recommended dose in HIV-1–infected adult subjects in Phase 3 trials, postural hypotension was seen at a rate similar to placebo (approximately 0.5%).
Patients with cardiovascular comorbidities, risk factors for postural hypotension, or receiving concomitant medication known to lower blood pressure, could be at increased risk of cardiovascular adverse events triggered by postural hypotension. Additional monitoring may be warranted.
Postural Hypotension in Patients with Renal Impairment
An increased risk of postural hypotension may occur in patients with severe renal insufficiency or in those with ESRD due to increased maraviroc exposure in some patients. SELZENTRY should be used in patients with severe renal impairment or ESRD only if they are not receiving a concomitant potent CYP3A inhibitor or inducer. However, the use of SELZENTRY in these patients should only be considered when no alternative treatment options are available. If adult patients with severe renal impairment or ESRD experience any symptoms of postural hypotension while taking 300 mg twice daily, the dose should be reduced to 150 mg twice daily [see Dosage and Administration (2.5)].
5.4 Immune Reconstitution Syndrome
Immune reconstitution syndrome has been reported in patients treated with combination antiretroviral therapy, including SELZENTRY. During the initial phase of combination antiretroviral treatment, patients whose immune systems respond may develop an inflammatory response to indolent or residual opportunistic infections (such as infection with Mycobacterium avium infection, cytomegalovirus, Pneumocystis jirovecii pneumonia [PCP], tuberculosis, or reactivation of Herpes simplex and Herpes zoster), which may necessitate further evaluation and treatment.
Autoimmune disorders (such as Graves’ disease, polymyositis, and Guillain-Barré syndrome) have also been reported to occur in the setting of immune reconstitution; however, the time to onset is more variable, and can occur many months after initiation of treatment.
5.5 Potential Risk of Infection
SELZENTRY antagonizes the CCR5 co-receptor located on some immune cells, and therefore could potentially increase the risk of developing infections. The overall incidence and severity of infection, as well as AIDS-defining category C infections, were comparable in the treatment groups during the Phase 3 adult treatment‑experienced trials of SELZENTRY. While there was a higher rate of certain upper respiratory tract infections reported in the treatment arm receiving SELZENTRY compared with placebo (23% versus 13%), there was a lower rate of pneumonia (2% versus 5%) reported in subjects receiving SELZENTRY. A higher incidence of Herpes virus infections (11 per 100 patient‑years) was also reported in the treatment arm receiving SELZENTRY when adjusted for exposure compared with placebo (8 per 100 patient‑years).
In the Phase 2b/3 trial in treatment‑naive adult subjects, the incidence of AIDS-defining Category C events when adjusted for exposure was 1.8 for SELZENTRY compared with 2.4 for efavirenz per 100 patient‑years of exposure.
Patients should be monitored closely for evidence of infections while receiving SELZENTRY.
5.6 Potential Risk of Malignancy
While no increase in malignancy has been observed with SELZENTRY, due to this drug’s mechanism of action, it could affect immune surveillance and lead to an increased risk of malignancy.
The exposure-adjusted rate for malignancies per 100 patient‑years of exposure in adult treatment‑experienced trials was 4.6 for SELZENTRY compared with 9.3 on placebo. In treatment‑naive adult subjects, the rates were 1.0 and 2.4 per 100 patient‑years of exposure for SELZENTRY and efavirenz, respectively.
Long-term follow-up is needed to more fully assess this risk.
-
6 ADVERSE REACTIONS
The following adverse reactions are discussed in other sections of the labeling:
- •
- Hepatotoxicity [see Boxed Warning, Warnings and Precautions (5.1)]
- •
- Severe Skin and Hypersensitivity Reactions [see Warnings and Precautions (5.2)]
- •
- Cardiovascular Events [see Warnings and Precautions (5.3)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared with rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Clinical Trials Experience in Adult Subjects
Treatment‑Experienced Subjects: The safety profile of SELZENTRY is primarily based on 840 HIV-1–infected subjects who received at least 1 dose of SELZENTRY during two Phase 3 trials. A total of 426 of these subjects received the indicated twice‑daily dosing regimen.
Assessment of treatment‑emergent adverse events is based on the pooled data from 2 trials in subjects with CCR5-tropic HIV‑1 (A4001027 and A4001028). The median duration of therapy with SELZENTRY for subjects in these trials was 48 weeks, with the total exposure on SELZENTRY twice daily at 309 patient‑years versus 111 patient‑years on placebo each administered with optimized background therapy (OBT). The population was 89% male and 84% white, with mean age of 46 years (range: 17 to 75 years). Subjects received dose equivalents of 300 mg maraviroc once or twice daily.
The most common adverse events reported with twice‑daily therapy with SELZENTRY with frequency rates higher than placebo, regardless of causality, were upper respiratory tract infections, cough, pyrexia, rash, and dizziness. In these 2 trials, the rate of discontinuation due to adverse events was 5% for subjects who received SELZENTRY twice daily + OBT as well as those who received placebo + OBT. Most of the adverse events reported were judged to be mild to moderate in severity. The data described below occurred with twice‑daily dosing of SELZENTRY.
The total numbers of subjects reporting infections were 233 (55%) and 84 (40%) in the group receiving SELZENTRY twice daily and the placebo group, respectively. Correcting for the longer duration of exposure on SELZENTRY compared with placebo, the exposure‑adjusted frequency (rate per 100 subject‑years) of these events was 133 for both SELZENTRY twice daily and placebo.
Dizziness or postural dizziness occurred in 8% of subjects on either SELZENTRY or placebo, with 2 subjects (0.5%) on SELZENTRY permanently discontinuing therapy (1 due to syncope, 1 due to orthostatic hypotension) versus 1 subject on placebo (0.5%) permanently discontinuing therapy due to dizziness.
Treatment-emergent adverse events, regardless of causality, from Trials A4001027 and A4001028 are summarized in Table 5. Selected events occurring at greater than or equal to 2% of subjects and at a numerically higher rate in subjects treated with SELZENTRY are included; events that occurred at the same or higher rate on placebo are not displayed.
Table 5. Selected Treatment-Emergent Adverse Events (All Causality) ≥2% on SELZENTRY (and at a Higher Rate Compared with Placebo) in Trials A4001027 and A4001028 (Pooled Analysis, 48 Weeks) a 300-mg dose equivalent.
b PYE = Patient-years of exposure.SELZENTRY
Twice Dailya
Placebo
Body System/
Adverse Event
(n = 426)
%
Exposure-Adjusted Rate
(per 100 pt-yrs)
PYE = 309b
(n = 209)
%
Exposure-Adjusted Rate
(per 100 pt-yrs)
PYE = 111b
Eye Disorders
Conjunctivitis
2
3
1
3
Ocular infections, inflammations, and associated manifestations
2
3
1
2
Gastrointestinal Disorders
Constipation
6
9
3
6
General Disorders and Administration Site Conditions
Pyrexia
13
20
9
17
Pain and discomfort
4
5
3
5
Infections and Infestations
Upper respiratory tract infection
23
37
13
27
Herpes infection
8
11
4
8
Sinusitis
7
10
3
6
Bronchitis
7
9
5
9
Folliculitis
4
5
2
4
Anogenital warts
2
3
1
3
Influenza
2
3
0.5
1
Otitis media
2
3
0.5
1
Metabolism and Nutrition Disorders
Appetite disorders
8
11
7
13
Musculoskeletal and Connective Tissue Disorders
Joint-related signs and symptoms
7
10
3
5
Muscle pains
3
4
0.5
1
Neoplasms Benign, Malignant, and Unspecified
Skin neoplasms benign
3
4
1
3
Nervous System Disorders
Dizziness/postural dizziness
9
13
8
17
Paresthesias and dysesthesias
5
7
3
6
Sensory abnormalities
4
6
1
3
Disturbances in consciousness
4
5
3
6
Peripheral neuropathies
4
5
3
6
Psychiatric Disorders
Disturbances in initiating and maintaining sleep
8
11
5
10
Depressive disorders
4
6
3
5
Anxiety symptoms
4
5
3
7
Renal and Urinary Disorders
Bladder and urethral symptoms
5
7
1
3
Urinary tract signs and symptoms
3
4
1
3
Respiratory, Thoracic, and Mediastinal Disorders
Coughing and associated symptoms
14
21
5
10
Upper respiratory tract signs and symptoms
6
9
3
6
Nasal congestion and inflammations
4
6
3
5
Breathing abnormalities
4
5
2
5
Paranasal sinus disorders
3
4
0.5
1
Skin and Subcutaneous Tissue Disorders
Rash
11
16
5
11
Apocrine and eccrine gland disorders
5
7
4
7.5
Pruritus
4
5
2
4
Lipodystrophies
3
5
0.5
1
Erythema
2
3
1
2
Vascular Disorders
Vascular hypertensive disorders
3
4
2
4
Laboratory Abnormalities: Table 6 shows the treatment-emergent Grade 3-4 laboratory abnormalities that occurred in greater than 2% of subjects receiving SELZENTRY.
Table 6. Maximum Shift in Laboratory Test Values (without Regard to Baseline) ≥2% of Grade 3-4 Abnormalities (ACTG Criteria) in Trials A4001027 and A4001028 (Pooled Analysis, 48 Weeks) ULN = Upper limit of normal; OBT = Optimized background therapy.
a Percentages based on total subjects evaluated for each laboratory parameter.Laboratory Parameter Preferred Term
Limit
SELZENTRY
Twice Daily + OBT
(n = 421)a
%
Placebo + OBT
(n = 207)a
%
Aspartate aminotransferase
>5.0 x ULN
4.8
2.9
Alanine aminotransferase
>5.0 x ULN
2.6
3.4
Total bilirubin
>2.5 x ULN
5.5
5.3
Amylase
>2.0 x ULN
5.7
5.8
Lipase
>2.0 x ULN
4.9
6.3
Absolute neutrophil count
<750/mm3
4.3
2.4
Treatment‑Naive Subjects: Treatment-Emergent Adverse Events: Treatment-emergent adverse events, regardless of causality, from Trial A4001026, a double-blind, comparative, controlled trial in which 721 treatment-naive subjects received SELZENTRY 300 mg twice daily (n = 360) or efavirenz 600 mg once daily (n = 361) in combination with lamivudine/zidovudine (COMBIVIR) for 96 weeks, are summarized in Table 7. Selected events occurring in greater than or equal to 2% of subjects and at a numerically higher rate in subjects treated with SELZENTRY are included; events that occurred at the same or higher rate on efavirenz are not displayed.
Table 7. Selected Treatment-Emergent Adverse Events (All Causality) ≥2% on SELZENTRY (and at a Higher Rate Compared with Efavirenz) in Trial A4001026 (96 Weeks) Body System/
Adverse Event
SELZENTRY
300 mg Twice Daily +Lamivudine/Zidovudine
(n = 360)
%
Efavirenz
600 mg Once Daily +Lamivudine/Zidovudine
(n = 361)
%
Blood and Lymphatic System Disorders
Anemias NEC
8
5
Neutropenias
4
3
Ear and Labyrinth Disorders
Ear disorders NEC
3
2
Gastrointestinal Disorders
Flatulence, bloating, and distention
10
7
Gastrointestinal atonic and hypomotility disorders NEC
9
5
Gastrointestinal signs and symptoms NEC
3
2
General Disorders and Administration Site Conditions
Body temperature perception
3
1
Infections and Infestations
Upper respiratory tract infection
32
30
Bronchitis
13
9
Herpes infection
7
6
Bacterial infections NEC
6
3
Herpes zoster/varicella
5
4
Tinea infections
4
3
Lower respiratory tract and lung infections
3
2
Neisseria infections
3
0
Viral infections NEC
3
2
Musculoskeletal and Connective Tissue Disorders
Joint-related signs and symptoms
6
5
Nervous System Disorders
Paresthesias and dysesthesias
4
3
Memory loss (excluding dementia)
3
1
Renal and Urinary Disorders
Bladder and urethral symptoms
4
3
Reproductive System and Breast Disorders
Erection and ejaculation conditions and disorders
3
2
Respiratory, Thoracic, and Mediastinal Disorders
Upper respiratory tract signs and symptoms
9
5
Skin and Subcutaneous Disorders
Nail and nail bed conditions (excluding infections and infestations)
6
2
Lipodystrophies
4
3
Acnes
3
2
Alopecias
2
1
Laboratory Abnormalities:
Table 8. Maximum Shift in Laboratory Test Values (without Regard to Baseline) ≥2% of Grade 3-4 Abnormalities (ACTG Criteria) in Trial A4001026 (96 Weeks) ULN = Upper limit of normal.
a n = Total number of subjects evaluable for laboratory abnormalities.
Percentages based on total subjects evaluated for each laboratory parameter. If the same subject in a given treatment group had greater than 1 occurrence of the same abnormality, only the most severe is counted.Laboratory Parameter Preferred Term
Limit
SELZENTRY
300 mg Twice Daily +
Lamivudine/Zidovudine
(n = 353)a%
Efavirenz
600 mg Once Daily+
Lamivudine/Zidovudine
(n = 350)a%
Aspartate aminotransferase
>5.0 x ULN
4.0
4.0
Alanine aminotransferase
>5.0 x ULN
3.9
4.0
Creatine kinase
>10.0 x ULN
3.9
4.8
Amylase
>2.0 x ULN
4.3
6.0
Absolute neutrophil count
<750/mm3
5.7
4.9
Hemoglobin
<7.0 g/dL
2.9
2.3
Less Common Adverse Events in Clinical Trials: The following adverse events occurred in less than 2% of subjects treated with SELZENTRY or at a rate similar to the comparator. These events have been included because of their seriousness and either increased frequency on SELZENTRY or are potential risks due to the mechanism of action. Events attributed to the subjects’ underlying HIV-1 infection are not listed.
Blood and Lymphatic System: Marrow depression and hypoplastic anemia.
Cardiac Disorders: Unstable angina, acute cardiac failure, coronary artery disease, coronary artery occlusion, myocardial infarction, myocardial ischemia.
Hepatobiliary Disorders: Hepatic cirrhosis, hepatic failure, cholestatic jaundice, portal vein thrombosis, jaundice.
Infections and Infestations: Endocarditis, infective myositis, viral meningitis, pneumonia, treponema infections, septic shock, Clostridium difficile colitis, meningitis.
Musculoskeletal and Connective Tissue Disorders: Myositis, osteonecrosis, rhabdomyolysis, blood creatine kinase increased.
Neoplasms Benign, Malignant, and Unspecified (Including Cysts and Polyps): Abdominal neoplasm, anal cancer, basal cell carcinoma, Bowen’s disease, cholangiocarcinoma, diffuse large B-cell lymphoma, lymphoma, metastases to liver, esophageal carcinoma, nasopharyngeal carcinoma, squamous cell carcinoma, squamous cell carcinoma of skin, tongue neoplasm (malignant stage unspecified), anaplastic large cell lymphomas T- and null-cell types, bile duct neoplasms malignant, endocrine neoplasms malignant and unspecified.
Nervous System Disorders: Cerebrovascular accident, convulsions and epilepsy, tremor (excluding congenital), facial palsy, hemianopia, loss of consciousness, visual field defect.
Clinical Trials Experience in Pediatric Subjects
HIV-1–Infected Pediatric Subjects: Trial A4001031 is an open-label trial in which 103 treatment-experienced, CCR5-tropic, HIV-1–infected pediatric subjects aged 2 to less than 18 years weighing at least 10 kg received SELZENTRY twice daily in combination with OBT. The dose of SELZENTRY was based on body surface area (BSA) and on whether the subject was receiving potent CYP3A inhibitors and/or inducers. The median duration of therapy with SELZENTRY was 131 weeks with 72% of subjects receiving study treatment for greater than 48 weeks and 62% of subjects receiving study treatment for 96 weeks.
In these 103 children and adolescents, the safety profile through 96 weeks was similar to that for adults. Most of the adverse reactions reported were mild to moderate; severe (Grade 3 and 4) adverse reactions occurred in 2% of subjects. The most common adverse reactions (all grades) reported with twice-daily therapy with SELZENTRY were vomiting (12%), abdominal pain (4%), diarrhea (4%), nausea (4%), and dizziness (3%). Three subjects (3%) discontinued due to adverse events.
Maraviroc-related gastrointestinal adverse events through 48 weeks (nausea, vomiting, diarrhea, constipation, and abdominal pain/cramps) were observed more commonly in subjects who received the SELZENTRY oral solution (21%) compared with those who received SELZENTRY tablets (16%). Subjects were permitted to change formulations after Week 48.
HIV-1–Exposed Neonates: The IMPAACT P2007 trial was an open-label trial in which 47 full-term HIV-1–exposed neonates (born to HIV-1–infected mothers) received at least one dose of SELZENTRY in combination with other antiretrovirals, mostly zidovudine and/or nevirapine [see Clinical Pharmacology (12.3)]. Cohort 1 received 2 single doses of SELZENTRY: the first within 3 days of birth and the second at 7 to 14 days of age. Cohort 2 received SELZENTRY twice daily for 6 weeks beginning within 3 days of birth and continued through Week 6. Both cohorts received SELZENTRY with or without exposure to maternal efavirenz (in utero only in Cohort 1, and both in utero and after birth while breastfeeding in Cohort 2). The population was 51% male and 81% black. All infants were followed for safety through 16 weeks, with a total of 37 infants evaluable for safety.
There were no additional adverse reactions observed in neonates compared with those seen in adults. All adverse reactions reported were mild to moderate. The most common adverse reaction (all grades) reported with SELZENTRY was hemoglobin decreased (14%). One subject (3%) discontinued due to an adverse event (Grade 3 staphylococcal sepsis).
6.2 Postmarketing Experience
The following adverse events have been identified during postapproval use of SELZENTRY. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Skin and Subcutaneous Tissue Disorders
Stevens‑Johnson syndrome (SJS), drug rash with eosinophilia and systemic symptoms (DRESS), toxic epidermal necrolysis (TEN).
-
7 DRUG INTERACTIONS
7.1 Effect of Concomitant Drugs on the Pharmacokinetics of Maraviroc
Maraviroc is metabolized by CYP3A and is also a substrate for P-glycoprotein (P-gp), organic anion-transporting polypeptide (OATP)1B1, and multidrug resistance-associated protein (MRP)2. The pharmacokinetics of maraviroc are likely to be modulated by inhibitors and inducers of CYP3A and P-gp and may be modulated by inhibitors of OATP1B1 and MRP2. Therefore, a dosage adjustment may be required when maraviroc is coadministered with those drugs [see Dosage and Administration (2.3, 2.4)].
Concomitant use of maraviroc and St. John's wort (Hypericum perforatum) or products containing St. John's wort is not recommended. Coadministration of maraviroc with St. John's wort is expected to substantially decrease maraviroc concentrations and may result in suboptimal levels of maraviroc and lead to loss of virologic response and possible resistance to maraviroc.
Additional drug interaction information is available [see Clinical Pharmacology (12.3)].
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Exposure Registry
There is a pregnancy exposure registry that monitors pregnancy outcomes in women exposed to SELZENTRY during pregnancy. Physicians are encouraged to register patients by calling the Antiretroviral Pregnancy Registry (APR) at 1-800-258-4263.
Risk Summary
Limited data on the use of SELZENTRY during pregnancy from the APR and case reports are not sufficient to inform a drug-associated risk of birth defects and miscarriage. In animal reproduction studies, no evidence of adverse developmental outcomes was observed with maraviroc. During organogenesis in the rat and rabbit, systemic exposures (AUC) to maraviroc were approximately 20 times (rats) and 5 times (rabbits) the exposure in humans at the recommended 300-mg twice-daily dose. In the rat pre- and post-natal development study, maternal systemic exposure (AUC) to maraviroc was approximately 14 times the exposure in humans at the recommended 300-mg twice-daily dose (see Data).
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Animal Data: Maraviroc was administered orally to pregnant rats (up to 1,000 mg per kg per day) and rabbits (up to 75 mg per kg per day) on gestation Days 6 to 17 and 7 to 19, respectively. No adverse effects on embryo-fetal development were observed at these dose levels, resulting in exposures (AUC) approximately 20 times (rats) and 5 times (rabbits) higher than human exposures at the recommended daily dose. In the rat pre- and post-natal development study, maraviroc was administered orally at up to 1,000 mg per kg per day on gestation Day 6 to lactation/post-partum Day 20, with development of the offspring (including fertility and reproductive performance) unaffected by maternal administration of maraviroc at an exposure (AUC) approximately 14 times higher than human exposure at the recommended daily dose.
8.2 Lactation
Risk Summary
The Centers for Disease Control and Prevention recommend that HIV‑1–infected mothers in the United States not breastfeed their infants to avoid risking postnatal transmission of HIV‑1 infection.
There are no data on the presence of maraviroc in human milk, the effects on the breastfed infant, or the effects on milk production. When administered to lactating rats, maraviroc was present in milk (see Data). Because of the potential for (1) HIV transmission (in HIV-negative infants), (2) developing viral resistance (in HIV-positive infants), and (3) serious adverse reactions in a breastfed infant similar to those seen in adults, instruct mothers not to breastfeed if they are receiving SELZENTRY.
Data
Maraviroc (and related metabolites) was excreted into the milk of lactating rats following a single oral dose of maraviroc (100 mg per kg) on lactation Day 12, with a maximal milk concentration achieved one hour post-administration at a milk concentration approximately 2.5 times that of maternal plasma concentrations.
8.4 Pediatric Use
The safety and efficacy of SELZENTRY have been established in pediatric patients aged from birth to less than 18 years. The use of SELZENTRY in pediatric patients was supported by pharmacokinetic and safety data described below and by previous demonstration of efficacy in adult patients [see Indications and Usage (1), Dosage and Administration (2.4)].
HIV-1–Infected Pediatric Patients Aged 2 to Less Than 18 Years: The safety, pharmacokinetic profile, and antiviral activity of SELZENTRY were evaluated in treatment-experienced, CCR5-tropic, HIV-1–infected pediatric subjects aged 2 to less than 18 years weighing at least 10 kg in an open-label, multicenter clinical trial, A4001031 [see Adverse Reactions (6.1), Clinical Studies (14.2)]. Pharmacokinetics were evaluated in a total of 98 pediatric subjects: 85 subjects received SELZENTRY and concomitant medications that included potent CYP3A inhibitors with or without potent CYP3A inducers, 10 subjects received SELZENTRY and noninteracting medications (not containing potent CYP3A inhibitors or potent CYP3A inducers), and three subjects received SELZENTRY and medications that included potent CYP3A inducers without potent CYP3A inhibitors [see Clinical Pharmacology (12.3)].
HIV-1–Infected Pediatric Patients Aged Older Than 6 Weeks to Less Than 2 Years: No clinical trials have been conducted in children aged older than 6 weeks to less than 2 years. Dosing recommendations for SELZENTRY in this population when concomitantly receiving noninteracting medications are based on population pharmacokinetic modeling and simulation only [see Dosage and Administration (2.4), Clinical Pharmacology (12.3)].
HIV-1–Infected Neonates Aged from Birth to 6 Weeks: The recommendation of SELZENTRY for the treatment of HIV-1 infection in this pediatric population is based on safety and pharmacokinetic data obtained from clinical trial IMPAACT P2007. In IMPAACT P2007, the safety and pharmacokinetic profiles of SELZENTRY were evaluated in full-term HIV-1–exposed neonates (born to HIV-1–infected mothers) aged from birth through 6 weeks [see Adverse Reactions (6.1)]. Pharmacokinetics were evaluated in 38 of 47 enrolled neonates who received SELZENTRY as a single dose (n = 13) or multiple doses (n = 25) up to 6 weeks of age concomitantly with other antiretrovirals (mostly zidovudine and/or nevirapine) with or without maternal exposure to efavirenz. HIV-1 status was assessed by nucleic acid test at birth, Week 6, and Week 16; all 47 enrolled neonates were HIV-1 negative at completion of the study [see Clinical Pharmacology (12.3)].
There are insufficient data to make dosing recommendations for use of SELZENTRY in pediatric patients concomitantly receiving potent CYP3A inhibitors and weighing less than 10 kg, or in any pediatric patients concomitantly receiving potent CYP3A inducers without a potent CYP3A inhibitor [see Dosage and Administration (2.4, 2.5)].
SELZENTRY is not recommended in pre-term neonates or in pediatric patients weighing less than 2 kg.
8.5 Geriatric Use
There were insufficient numbers of subjects aged 65 and over in the clinical trials to determine whether they respond differently from younger subjects. In general, caution should be exercised when administering SELZENTRY in elderly patients, also reflecting the greater frequency of decreased hepatic and renal function, of concomitant disease and other drug therapies.
8.6 Renal Impairment
Recommended doses of SELZENTRY for adult patients with impaired renal function (CrCl less than or equal to 80 mL per minute) are based on the results of a pharmacokinetic trial conducted in healthy adult subjects with various degrees of renal impairment. Maraviroc has not been studied in pediatric patients with renal impairment. There are no data to recommend specific doses of SELZENTRY in pediatric patients with mild to moderate renal impairment [see Use in Specific Populations (8.4)]. SELZENTRY is contraindicated in pediatric patients with severe renal impairment or ESRD on regular hemodialysis who are receiving potent CYP3A inhibitors [see Contraindications (4)].
The pharmacokinetics of maraviroc in adult subjects with mild and moderate renal impairment was similar to that in subjects with normal renal function [see Clinical Pharmacology (12.3)]. A limited number of adult subjects with mild and moderate renal impairment in the Phase 3 clinical trials (n = 131 and n = 12, respectively) received the same dose of SELZENTRY as that administered to subjects with normal renal function. In these subjects, there was no apparent difference in the adverse event profile for maraviroc compared with subjects with normal renal function.
If adult patients with severe renal impairment or ESRD not receiving a concomitant potent CYP3A inhibitor or inducer experience any symptoms of postural hypotension while taking SELZENTRY 300 mg twice daily, the dose should be reduced to 150 mg twice daily. No trials have been performed in subjects with severe renal impairment or ESRD co-treated with potent CYP3A inhibitors or inducers. Hence, no dose of SELZENTRY can be recommended, and SELZENTRY is contraindicated for these patients [see Dosage and Administration (2.3), Contraindications (4), Warnings and Precautions (5.3), Clinical Pharmacology (12.3)].
8.7 Hepatic Impairment
Maraviroc is principally metabolized by the liver; therefore, when administering this drug to patients with hepatic impairment, maraviroc concentrations may be increased. Maraviroc concentrations are higher when SELZENTRY 150 mg is administered with a potent CYP3A inhibitor compared with following administration of 300 mg without a CYP3A inhibitor, so patients with moderate hepatic impairment who receive SELZENTRY 150 mg with a potent CYP3A inhibitor should be monitored closely for maraviroc-associated adverse events. Maraviroc has not been studied in subjects with severe hepatic impairment or in pediatric patients with any degree of hepatic impairment [see Warnings and Precautions (5.1), Clinical Pharmacology (12.3)].
-
10 OVERDOSAGE
The highest single dose administered in clinical trials was 1,200 mg. The dose-limiting adverse event was postural hypotension, which was observed at 600 mg. While the recommended dose for SELZENTRY in patients receiving a CYP3A inducer without a CYP3A inhibitor is 600 mg twice daily, this dose is appropriate due to enhanced metabolism.
Prolongation of the QT interval was seen in dogs and monkeys at plasma concentrations 6 and 12 times, respectively, those expected in humans at the intended exposure of 300-mg equivalents twice daily. However, no significant QT prolongation was seen in the trials in treatment-experienced subjects with HIV using the recommended doses of maraviroc, or in a specific pharmacokinetic trial to evaluate the potential of maraviroc to prolong the QT interval [see Clinical Pharmacology (12.2)].
There is no specific antidote for overdose with maraviroc. Treatment of overdose should consist of general supportive measures including keeping the patient in a supine position, careful assessment of patient vital signs, blood pressure, and electrocardiogram.
Administration of activated charcoal may also be used to aid in removal of unabsorbed drug. Hemodialysis had a minimal effect on maraviroc clearance and exposure in a trial in subjects with ESRD [see Clinical Pharmacology (12.3)].
-
11 DESCRIPTION
SELZENTRY (maraviroc) is a selective, slowly reversible, small molecule antagonist of the interaction between human CCR5 and HIV-1 gp120. Blocking this interaction prevents CCR5-tropic HIV-1 entry into cells.
SELZENTRY film-coated tablets for oral administration contain 25, 75, 150, or 300 mg of maraviroc and the following inactive ingredients: dibasic calcium phosphate (anhydrous), magnesium stearate, microcrystalline cellulose, and sodium starch glycolate. The film coat (Opadry II Blue [85G20583]) contains FD&C blue #2 aluminum lake, soya lecithin, polyethylene glycol (macrogol 3350), polyvinyl alcohol, talc, and titanium dioxide.
SELZENTRY oral solution contains 20 mg per mL of maraviroc and the following inactive ingredients: citric acid (anhydrous), purified water, sodium benzoate, sodium citrate dihydrate, strawberry flavoring (501440T), and sucralose.
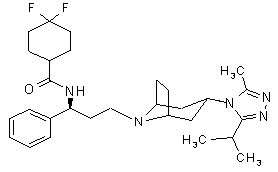
Maraviroc is chemically described as 4,4-difluoro-N-{(1S)-3-[exo-3-(3-isopropyl-5-methyl-4H-1,2,4-triazol-4-yl)-8-azabicyclo[3.2.1]oct-8-yl]-1-phenylpropyl}cyclohexanecarboxamide.
The molecular formula is C29H41F2N5O and the structural formula is:
Maraviroc is a white to pale-colored powder with a molecular weight of 513.67. It is highly soluble across the physiological pH range (pH 1.0 to 7.5).
-
12 CLINICAL PHARMACOLOGY
12.2 Pharmacodynamics
Exposure-Response Relationship in Treatment-Experienced Adult Subjects
The relationship between maraviroc, modeled plasma trough concentration (Cmin) (1 to 9 samples per subject taken on up to 7 visits), and virologic response was evaluated in 973 treatment-experienced HIV-1-infected subjects with varied optimized background antiretroviral regimens in Trials A4001027 and A4001028. The Cmin, baseline viral load, baseline CD4+ cell count, and overall sensitivity score (OSS) were found to be important predictors of virologic success (defined as viral load less than 400 copies per mL at 24 weeks). Table 9 illustrates the proportions of subjects with virologic success (%) within each Cmin quartile for 150-mg twice-daily and 300-mg twice-daily groups.
Table 9. Treatment-Experienced Subjects with Virologic Success by Cmin Quartile (Q1-Q4) 150 mg Twice Daily
(with CYP3A Inhibitors)300 mg Twice Daily
(without CYP3A Inhibitors)n
Median Cmin
% Subjects with Virologic Success
n
Median Cmin
% Subjects with Virologic Success
Placebo
160
-
30.6
35
-
28.6
Q1
78
33
52.6
22
13
50.0
Q2
77
87
63.6
22
29
68.2
Q3
78
166
78.2
22
46
63.6
Q4
78
279
74.4
22
97
68.2
Exposure-Response Relationship in Treatment-Naive Adult Subjects
The relationship between maraviroc, modeled plasma trough concentration (Cmin) (1 to 12 samples per subject taken on up to 8 visits), and virologic response was evaluated in 294 treatment-naive HIV–1-infected subjects receiving maraviroc 300 mg twice daily in combination with lamivudine/zidovudine in Trial A4001026. Table 10 illustrates the proportion (%) of subjects with virologic success less than 50 copies per mL at 48 weeks within each Cmin quartile for the 300-mg twice-daily dose.
Table 10. Treatment-Naive Subjects with Virologic Success by Cmin Quartile (Q1-Q4) 300 mg Twice Daily
n
Median Cmin
% Subjects with Virologic Success
Q1
75
23
57.3
Q2
72
39
72.2
Q3
73
56
74.0
Q4
74
81
83.8
Eighteen of 75 (24%) subjects in Q1 had no measurable maraviroc concentration on at least one occasion versus 1 of 73 and 1 of 74 in Q3 and Q4, respectively.
Effects on Electrocardiogram
A placebo-controlled, randomized, crossover trial to evaluate the effect on the QT interval of healthy male and female volunteers was conducted with 3 single oral doses of maraviroc and moxifloxacin. The placebo-adjusted mean maximum (upper 1-sided 95% CI) increases in QTc from baseline after 100, 300, and 900 mg of maraviroc were –2 (0), -1 (1), and 1 (3) msec, respectively, and 13 (15) msec for moxifloxacin 400 mg. No subject in any group had an increase in QTc of greater than or equal to 60 msec from baseline. No subject experienced an interval exceeding the potentially clinically relevant threshold of 500 msec.
12.3 Pharmacokinetics
Table 11. Mean Maraviroc Pharmacokinetic Parameters in Adults a The estimated exposure is lower compared with other trials possibly due to sparse sampling, food effect, compliance, and concomitant medications. Patient Population
Maraviroc Dose
n
AUC12 (ng.h/mL)
Cmax (ng/mL)
Cmin (ng/mL)
Healthy volunteers (Phase 1)
300 mg twice daily
64
2,908
888
43.1
Asymptomatic HIV subjects (Phase 2a)
300 mg twice daily
8
2,550
618
33.6
Treatment-experienced HIV subjects (Phase 3)a
300 mg twice daily
94
1,513
266
37.2
150 mg twice daily
(+ CYP3A inhibitor)
375
2,463
332
101
Treatment-naive HIV subjects (Phase 2b/3)a
300 mg twice daily
344
1,865
287
60
Absorption
Peak maraviroc plasma concentrations are attained 0.5 to 4 hours following single oral doses of 1 to 1,200 mg administered to uninfected volunteers. The pharmacokinetics of oral maraviroc are not dose proportional over the dose range.
The absolute bioavailability of a 100‑mg dose is 23% and is predicted to be 33% at 300 mg. Maraviroc is a substrate for the efflux transporter P-gp.
Effect of Food on Oral Absorption: Coadministration of a 300‑mg tablet with a high‑fat breakfast reduced maraviroc Cmax and AUC by 33% and coadministration of 75 mg of oral solution with a high-fat breakfast reduced maraviroc AUC by 73% in healthy adult volunteers. Studies with the tablet formulation demonstrated a reduced food effect at higher doses.
There were no food restrictions in the adult trials (using the tablet formulation) or in the pediatric trial (using both tablet and oral solution formulations) that demonstrated the efficacy/antiviral activity and safety of maraviroc [see Clinical Studies (14.1, 14.2)].
Distribution
Maraviroc is bound (approximately 76%) to human plasma proteins, and shows moderate affinity for albumin and alpha‑1 acid glycoprotein. The volume of distribution of maraviroc is approximately 194 L.
Elimination
Metabolism: Trials in humans and in vitro studies using human liver microsomes and expressed enzymes have demonstrated that maraviroc is principally metabolized by the cytochrome P450 system to metabolites that are essentially inactive against HIV‑1. In vitro studies indicate that CYP3A is the major enzyme responsible for maraviroc metabolism. In vitro studies also indicate that polymorphic enzymes CYP2C9, CYP2D6, and CYP2C19 do not contribute significantly to the metabolism of maraviroc.
Maraviroc is the major circulating component (~42% drug‑related radioactivity) following a single oral dose of 300 mg [14C]-maraviroc. The most significant circulating metabolite in humans is a secondary amine (~22% radioactivity) formed by N‑dealkylation. This polar metabolite has no significant pharmacological activity. Other metabolites are products of mono‑oxidation and are only minor components of plasma drug‑related radioactivity.
Excretion: The terminal half‑life of maraviroc following oral dosing to steady state in healthy subjects was 14 to 18 hours. A mass balance/excretion trial was conducted using a single 300‑mg dose of 14C-labeled maraviroc. Approximately 20% of the radiolabel was recovered in the urine and 76% was recovered in the feces over 168 hours. Maraviroc was the major component present in urine (mean of 8% dose) and feces (mean of 25% dose). The remainder was excreted as metabolites.
Specific Populations
Patients with Hepatic Impairment: Maraviroc is primarily metabolized and eliminated by the liver. A trial compared the pharmacokinetics of a single 300‑mg dose of SELZENTRY in subjects with mild (Child‑Pugh Class A, n = 8) and moderate (Child‑Pugh Class B, n = 8) hepatic impairment with pharmacokinetics in healthy subjects (n = 8). The mean Cmax and AUC were 11% and 25% higher, respectively, for subjects with mild hepatic impairment, and 32% and 46% higher, respectively, for subjects with moderate hepatic impairment compared with subjects with normal hepatic function. These changes do not warrant a dose adjustment. Maraviroc concentrations are higher when SELZENTRY 150 mg is administered with a potent CYP3A inhibitor compared with following administration of 300 mg without a CYP3A inhibitor, so patients with moderate hepatic impairment who receive SELZENTRY 150 mg with a potent CYP3A inhibitor should be monitored closely for maraviroc‑associated adverse events. The pharmacokinetics of maraviroc have not been studied in subjects with severe hepatic impairment [see Warnings and Precautions (5.1)].
Patients with Renal Impairment: A trial compared the pharmacokinetics of a single 300‑mg dose of SELZENTRY in adult subjects with severe renal impairment (CrCl less than 30 mL per minute, n = 6) and ESRD (n = 6) with healthy volunteers (n = 6). Geometric mean ratios for maraviroc Cmax and AUCinf were 2.4‑fold and 3.2‑fold higher, respectively, for subjects with severe renal impairment, and 1.7‑fold and 2.0‑fold higher, respectively, for subjects with ESRD as compared with subjects with normal renal function in this trial. Hemodialysis had a minimal effect on maraviroc clearance and exposure in subjects with ESRD. Exposures observed in subjects with severe renal impairment and ESRD were within the range observed in previous 300‑mg single‑dose trials of SELZENTRY in healthy volunteers with normal renal function. However, maraviroc exposures in the subjects with normal renal function in this trial were 50% lower than those observed in previous trials. Based on the results of this trial, no dose adjustment is recommended for patients with renal impairment receiving SELZENTRY without a potent CYP3A inhibitor or inducer. However, if patients with severe renal impairment or ESRD experience any symptoms of postural hypotension while taking SELZENTRY 300 mg twice daily, their dose should be reduced to 150 mg twice daily [see Dosage and Administration (2.3), Warnings and Precautions (5.3)].
In addition, the trial compared the pharmacokinetics of multiple‑dose SELZENTRY in combination with saquinavir/ritonavir 1,000/100 mg twice daily (a potent CYP3A inhibitor combination) for 7 days in subjects with mild renal impairment (CrCl greater than 50 and less than or equal to 80 mL per minute, n = 6) and moderate renal impairment (CrCl greater than or equal to 30 and less than or equal to 50 mL per minute, n = 6) with healthy volunteers with normal renal function (n = 6). Subjects received 150 mg of SELZENTRY at different dose frequencies (healthy volunteers – every 12 hours; mild renal impairment – every 24 hours; moderate renal impairment – every 48 hours). Compared with healthy volunteers (dosed every 12 hours), geometric mean ratios for maraviroc AUCtau, Cmax, and Cmin were 50% higher, 20% higher, and 43% lower, respectively, for subjects with mild renal impairment (dosed every 24 hours). Geometric mean ratios for maraviroc AUCtau, Cmax, and Cmin were 16% higher, 29% lower, and 85% lower, respectively, for subjects with moderate renal impairment (dosed every 48 hours) compared with healthy volunteers (dosed every 12 hours). Based on the data from this trial, no adjustment in dose is recommended for patients with mild or moderate renal impairment [see Dosage and Administration (2.3)].
Pediatric Patients: Aged 2 to Less Than 18 Years: The pharmacokinetics of maraviroc were evaluated in CCR5-tropic, HIV-1–infected, treatment-experienced pediatric subjects aged 2 to less than 18 years. In the dose-finding stage of Trial A4001031, doses were administered with food on intensive pharmacokinetic evaluation days and optimized to achieve an average concentration over the dosing interval (Cavg) of greater than 100 ng per mL. Throughout the trial, on non-intensive pharmacokinetic evaluation days maraviroc was taken with or without food. The initial dose of maraviroc was based on BSA and concomitant medication category (i.e., presence of CYP3A inhibitors and/or inducers). The conversion of dosing to a weight (kg)-band basis in children provides comparable exposures with those observed in the trial at the corresponding BSA.
Maraviroc pharmacokinetic parameters in pediatric subjects aged 2 to less than 18 years receiving potent CYP3A inhibitors with or without a potent CYP3A inducer were similar to those observed in adults (Table 12).
Table 12. Maraviroc Pharmacokinetic Parameters in Treatment-Experienced Pediatric Patients Receiving SELZENTRY with Potent CYP3A Inhibitors (with or without a Potent CYP3A Inducer) a Model-predicted steady-state pharmacokinetic parameters are presented. Weight
Dose of SELZENTRY
Maraviroc Pharmacokinetic Parametera
Geometric Mean
AUC12 (ng.h/mL)
Cavg (ng/mL)
Cmax (ng/mL)
Cmin (ng/mL)
10 kg to <20 kg
50 mg
twice daily2,349
196
324
78
20 kg to <30 kg
75 mg
twice daily3,020
252
394
118
30 kg to <40 kg
100 mg
twice daily3,229
269
430
126
≥40 kg
150 mg
twice daily4,044
337
563
152
Aged from Birth to Less Than 6 Weeks: The pharmacokinetics of maraviroc were evaluated in 38 of 47 enrolled HIV-1–exposed neonates (born to HIV-1–infected mothers) aged from birth up to 6 weeks [see Adverse Reactions (6.1)]. In the IMPAACT P2007 trial, 13 neonates received weight-based maraviroc dosing as single doses at birth and approximately 7 days, and 25 neonates received maraviroc twice daily up to 6 weeks of age without exposure to potent CYP3A inhibitors and/or inducers. Maraviroc pharmacokinetic parameters in neonates weighing at least 2 kg at birth (Table 13) were similar to those observed in adults. Exposure to maternal efavirenz both in utero (for a minimum of 2 weeks immediately prior to delivery) and after birth while breastfeeding did not have a meaningful impact on maraviroc pharmacokinetic parameters.
Table 13. Maraviroc Pharmacokinetic Parameters in Full-Term Neonates (Birth Up to 6 Weeks of Age) Receiving SELZENTRY with Noninteracting Concomitant Medicationsa a Noninteracting concomitant medications include all medications that are not potent CYP3A inhibitors or inducers.
b AUCinf calculated for single-dose pharmacokinetics.Pharmacokinetic Sampling Time
n
Median Dose (range)
Maraviroc Pharmacokinetic Parameter
Geometric Mean
AUC12 (ng.h/mL)
Cavg (ng/mL)
Cmax (ng/mL)
Cmin (ng/mL)
Day 1
13
30 mg
(20 to 40 mg)
single dose3,510b
292
380
-
Week 1
25
25 mg
(20 to 30 mg)twice daily
1,216
101
262
23
Week 4
25
30 mg
(20 to 40 mg)twice daily
1,385
115
295
43
Clinical pharmacokinetic data with maraviroc in pediatric patients aged older than 6 weeks to less than 2 years are not available and clinical pharmacokinetic data in pediatric patients aged 2 to less than 18 years receiving noninteracting concomitant medications are limited. Based on population pharmacokinetic modeling and simulation, the recommended dosing regimen of SELZENTRY for this population is predicted to result in similar maraviroc exposures when compared with exposures achieved in adults receiving SELZENTRY 300 mg twice daily (with noninteracting concomitant medications) [see Dosage and Administration (2.4)].
Geriatric Patients: Pharmacokinetics of maraviroc have not been fully evaluated in the elderly (aged 65 years and older). Based on population pharmacokinetic analyses, age did not have a clinically relevant effect on maraviroc exposure in subjects up to age 65 years [see Use in Specific Populations (8.5)].
Race and Gender: Based on population pharmacokinetics and 2 clinical CYP3A5 genotype analyses for race, no dosage adjustment is recommended based on race or gender.
Drug Interaction Studies
Effect of Concomitant Drugs on the Pharmacokinetics of Maraviroc: Maraviroc is a substrate of CYP3A and P-gp and hence its pharmacokinetics are likely to be modulated by inhibitors and inducers of these enzymes/transporters. The CYP3A/P-gp inhibitors ketoconazole, lopinavir/ritonavir, ritonavir, darunavir/ritonavir, saquinavir/ritonavir, and atazanavir ± ritonavir all increased the Cmax and AUC of maraviroc (Table 14). The CYP3A and/or P-gp inducers rifampin, etravirine, and efavirenz decreased the Cmax and AUC of maraviroc (Table 14). While not studied, potent CYP3A and/or P-gp inducers carbamazepine, phenobarbital, and phenytoin are expected to decrease maraviroc concentrations. Based on in vitro study results, maraviroc is also a substrate of OATP1B1 and MRP2; its pharmacokinetics may be modulated by inhibitors of these transporters.
Tipranavir/ritonavir (net CYP3A inhibitor/P-gp inducer) did not affect the steady‑state pharmacokinetics of maraviroc (Table 14). Cotrimoxazole and tenofovir did not affect the pharmacokinetics of maraviroc.
Table 14. Effect of Coadministered Agents on the Pharmacokinetics of Maraviroc a Compared with historical data. Coadministered Drug
and Dose
n
Dose of SELZENTRY
Ratio (90% CI) of Maraviroc Pharmacokinetic Parameters with/without Coadministered Drug
(No Effect = 1.00)
Cmin
AUCtau
Cmax
CYP3A and/or P-gp Inhibitors
Ketoconazole
400 mg q.d.
12
100 mg b.i.d.
3.75
(3.01, 4.69)
5.00
(3.98, 6.29)
3.38
(2.38, 4.78)
Ritonavir
100 mg b.i.d.
8
100 mg b.i.d.
4.55
(3.37, 6.13)
2.61
(1.92, 3.56)
1.28
(0.79, 2.09)
Saquinavir (soft gel capsules) /ritonavir
1,000 mg/100 mg b.i.d.
11
100 mg b.i.d.
11.3
(8.96, 14.1)
9.77
(7.87, 12.14)
4.78
(3.41, 6.71)
Lopinavir/ritonavir
400 mg/100 mg b.i.d.
11
300 mg b.i.d.
9.24
(7.98, 10.7)
3.95
(3.43, 4.56)
1.97
(1.66, 2.34)
Atazanavir
400 mg q.d.
12
300 mg b.i.d.
4.19
(3.65, 4.80)
3.57
(3.30, 3.87)
2.09
(1.72, 2.55)
Atazanavir/ritonavir
300 mg/100 mg q.d.
12
300 mg b.i.d.
6.67
(5.78, 7.70)
4.88
(4.40, 5.41)
2.67
(2.32, 3.08)
Darunavir/ritonavir
600 mg/100 mg b.i.d.
12
150 mg b.i.d.
8.00
(6.35, 10.1)
4.05
(2.94, 5.59)
2.29
(1.46, 3.59)
Elvitegravir/ritonavir
150 mg/100 mg q.d.
11
150 mg b.i.d.
4.23
(3.47, 5.16)
2.86
(2.33, 3.51)
2.15
(1.71, 2.69)
CYP3A and/or P-gp Inducers
Efavirenz
600 mg q.d.
12
100 mg b.i.d.
0.55
(0.43, 0.72)
0.55
(0.49, 0.62)
0.49
(0.38, 0.63)
Efavirenz
600 mg q.d.
12
200 mg b.i.d.
(+ efavirenz):
100 mg b.i.d. (alone)
1.09
(0.89, 1.35)
1.15
(0.98, 1.35)
1.16
(0.87, 1.55)
Rifampicin
600 mg q.d.
12
100 mg b.i.d.
0.22
(0.17, 0.28)
0.37
(0.33, 0.41)
0.34
(0.26, 0.43)
Rifampicin
600 mg q.d.
12
200 mg b.i.d.
(+ rifampicin):
100 mg b.i.d. (alone)
0.66
(0.54, 0.82)
1.04
(0.89, 1.22)
0.97
(0.72, 1.29)
Etravirine
200 mg b.i.d.
14
300 mg b.i.d.
0.61
(0.53, 0.71)
0.47
(0.38, 0.58)
0.40
(0.28, 0.57)
Nevirapinea
200 mg b.i.d.
(+ lamivudine 150 mg b.i.d., tenofovir 300 mg q.d.)8
300 mg
single dose
–
1.01
(0.65, 1.55)
1.54
(0.94, 2.51)
CYP3A and/or P-gp Inhibitors and Inducers
Lopinavir/ritonavir + efavirenz
400 mg/100 mg b.i.d. + 600 mg q.d.
11
300 mg b.i.d.
6.29
(4.72, 8.39)
2.53
(2.24, 2.87)
1.25
(1.01, 1.55)
Saquinavir (soft gel capsules) /ritonavir + efavirenz 1,000 mg/100 mg b.i.d. + 600 mg q.d.
11
100 mg b.i.d.
8.42
(6.46, 10.97)
5.00
(4.26, 5.87)
2.26
(1.64, 3.11)
Darunavir/ritonavir + etravirine
600 mg/100 mg b.i.d. + 200 mg b.i.d.
10
150 mg b.i.d.
5.27
(4.51, 6.15)
3.10
(2.57, 3.74)
1.77
(1.20, 2.60)
Fosamprenavir/ritonavir
700 mg/100 mg b.i.d.
14
300 mg b.i.d.
4.74
(4.03, 5.57)
2.49
(2.19, 2.82)
1.52
(1.27, 1.82)
Fosamprenavir/ritonavir
1,400 mg/100 mg q.d.
14
300 mg q.d.
1.80
(1.53, 2.13)
2.26
(1.99, 2.58)
1.45
(1.20, 1.74)
Tipranavir/ritonavir
500 mg/200 mg b.i.d.
12
150 mg b.i.d.
1.80
(1.55, 2.09)
1.02
(0.85, 1.23)
0.86
(0.61, 1.21)
Other
Raltegravir
400 mg b.i.d.
17
300 mg b.i.d.
0.90
(0.85, 0.96)
0.86
(0.80, 0.92)
0.79
(0.67, 0.94)
Effect of Maraviroc on the Pharmacokinetics of Concomitant Drugs: Maraviroc is unlikely to inhibit the metabolism of coadministered drugs metabolized by the following cytochrome P enzymes (CYP1A2, CYP2B6, CYP2C8, CYP2C9, CYP2C19, and CYP3A) or to inhibit the uptake of OATP1B1 or the export of MRP2 because maraviroc did not inhibit activity of those enzymes or transporters at clinically relevant concentrations in vitro. Maraviroc does not induce CYP1A2 in vitro. Additionally, in vitro studies have shown that maraviroc is not a substrate for, and does not inhibit, any of the major renal uptake inhibitors (organic anion transporter [OAT]1, OAT3, organic cation transporter [OCT]2, novel organic cation transporter [OCTN]1, and OCTN2) at clinically relevant concentrations.
In vitro results suggest that maraviroc could inhibit P-gp in the gut. However, maraviroc did not significantly affect the pharmacokinetics of digoxin in vivo, indicating maraviroc may not significantly inhibit or induce P-gp clinically.
Drug interaction trials were performed with maraviroc and other drugs likely to be coadministered or commonly used as probes for pharmacokinetic interactions (Table 14).
Coadministration of fosamprenavir 700 mg/ritonavir 100 mg twice daily and maraviroc 300 mg twice daily decreased the Cmin and AUC of amprenavir by 36% and 35%, respectively. Coadministration of fosamprenavir 1,400 mg/ritonavir 100 mg once daily and maraviroc 300 mg once daily decreased the Cmin and AUC of amprenavir by 15% and 30%, respectively. No dosage adjustment is necessary when SELZENTRY is dosed 150 mg twice daily in combination with fosamprenavir/ritonavir dosed once or twice daily. Fosamprenavir should be given with ritonavir when coadministered with SELZENTRY.
Maraviroc had no significant effect on the pharmacokinetics of elvitegravir, zidovudine, or lamivudine. Maraviroc decreased the Cmin and AUC of raltegravir by 27% and 37%, respectively, which is not clinically significant. Maraviroc had no clinically relevant effect on the pharmacokinetics of midazolam, the oral contraceptives ethinylestradiol and levonorgestrel, no effect on the urinary 6β-hydroxycortisol/cortisol ratio, suggesting no induction of CYP3A in vivo. Maraviroc had no effect on the debrisoquine metabolic ratio (MR) at 300 mg twice daily or less in vivo and did not cause inhibition of CYP2D6 in vitro until concentrations greater than 100 microM. However, there was 234% increase in debrisoquine MR on treatment compared with baseline at 600 mg once daily, suggesting potential inhibition of CYP2D6 at higher doses.
12.4 Microbiology
Mechanism of Action
Maraviroc is a member of a therapeutic class called CCR5 co-receptor antagonists. Maraviroc selectively binds to the human chemokine receptor CCR5 present on the cell membrane, preventing the interaction of HIV-1 gp120 and CCR5 necessary for CCR5-tropic HIV-1 to enter cells. CXCR4-tropic and dual-tropic HIV-1 entry is not inhibited by maraviroc.
Antiviral Activity in Cell Culture
Maraviroc inhibits the replication of CCR5-tropic laboratory strains and primary isolates of HIV-1 in models of acute peripheral blood leukocyte infection. The mean EC50 value (50% effective concentration) for maraviroc against HIV-1 group M isolates (subtypes A to J and circulating recombinant form AE) and group O isolates ranged from 0.1 to 4.5 nM (0.05 to 2.3 ng per mL) in cell culture.
When used with other antiretroviral agents in cell culture, the combination of maraviroc was not antagonistic with non-nucleoside reverse transcriptase inhibitors (NNRTIs: efavirenz and nevirapine), NRTIs (abacavir, didanosine, emtricitabine, lamivudine, stavudine, tenofovir, zalcitabine, and zidovudine), or protease inhibitors (PIs: amprenavir, atazanavir, darunavir, indinavir, lopinavir, nelfinavir, ritonavir, saquinavir, and tipranavir). Maraviroc was not antagonistic with the HIV-1 gp41 fusion inhibitor enfuvirtide. Maraviroc was not active against CXCR4-tropic and dual-tropic viruses (EC50 value greater than 10 microM). The antiviral activity of maraviroc against HIV-2 has not been evaluated.
Resistance in Cell Culture: HIV-1 variants with reduced susceptibility to maraviroc have been selected in cell culture following serial passage of 2 CCR5-tropic viruses (CCl/85 and RU570). The maraviroc-resistant viruses remained CCR5-tropic with no evidence of a change from a CCR5-tropic virus to a CXCR4-using virus. Two amino acid residue substitutions in the V3-loop region of the HIV-1 envelope glycoprotein (gp160), A316T, and I323V (HXB2 numbering), were shown to be necessary for the maraviroc-resistant phenotype in the HIV-1 isolate CCl/85. In the RU570 isolate a 3-amino acid residue deletion in the V3 loop, ΔQAI (HXB2 positions 315 to 317), was associated with maraviroc resistance. The relevance of the specific gp120 substitutions observed in maraviroc-resistant isolates selected in cell culture to clinical maraviroc resistance is not known. Maraviroc-resistant viruses were characterized phenotypically by concentration-response curves that did not reach 100% inhibition in phenotypic drug assays, rather than increases in EC50 values.
Cross-Resistance in Cell Culture: Maraviroc had antiviral activity against HIV‑1 clinical isolates resistant to NNRTIs, NRTIs, PIs, and the gp41 fusion inhibitor enfuvirtide in cell culture (EC50 values ranged from 0.7 to 8.9 nM [0.36 to 4.57 ng per mL]). Maraviroc‑resistant viruses that emerged in cell culture remained susceptible to enfuvirtide and the protease inhibitor saquinavir.
Clinical Resistance: Virologic failure on maraviroc can result from genotypic and phenotypic resistance to maraviroc, through outgrowth of undetected CXCR4-using virus present before maraviroc treatment (see Tropism), through resistance to background therapy drugs (Table 15), or due to low exposure to maraviroc [see Clinical Pharmacology (12.2)].
Antiretroviral Treatment-Experienced Adult Subjects (Trials A4001027 and A4001028): Week 48 data from treatment-experienced subjects failing maraviroc-containing regimens with CCR5-tropic virus (n = 58) have identified 22 viruses that had decreased susceptibility to maraviroc characterized in phenotypic drug assays by concentration-response curves that did not reach 100% inhibition. Additionally, CCR5-tropic virus from 2 of these treatment-failure subjects had greater than or equal to 3-fold shifts in EC50 values for maraviroc at the time of failure.
Fifteen of these viruses were sequenced in the gp120 encoding region and multiple amino acid substitutions with unique patterns in the heterogeneous V3 loop region were detected. Changes at either amino acid position 308 or 323 (HXB2 numbering) were seen in the V3 loop in 7 of the subjects with decreased maraviroc susceptibility. Substitutions outside the V3 loop of gp120 may also contribute to reduced susceptibility to maraviroc.
Antiretroviral Treatment-Naive Adult Subjects (Trial A4001026): Treatment-naive subjects receiving SELZENTRY had more virologic failures and more treatment-emergent resistance to the background regimen drugs compared with those receiving efavirenz (Table 15).
Table 15. Development of Resistance to Maraviroc or Efavirenz and Background Drugs in Antiretroviral Treatment-Naive Trial A4001026 for Patients with Only CCR5-Tropic Virus at Screening Using Enhanced Sensitivity TROFILE Assay a Includes subjects failing with CXCR4- or dual/mixed-tropism because these viruses are not intrinsically susceptible to maraviroc. Maraviroc
Efavirenz
Total N in dataset (as-treated)
273
241
Total virologic failures (as-treated)
85 (31%)
56 (23%)
Evaluable virologic failures with post baseline genotypic and phenotypic data
73
43
Lamivudine resistance
39 (53%)
13 (30%)
Zidovudine resistance
2 (3%)
0
Efavirenz resistance
–
23 (53%)
Phenotypic resistance to maraviroca
19 (26%)
–
In an as‑treated analysis of treatment‑naive subjects at 96 weeks, 32 subjects failed a maraviroc‑containing regimen with CCR5‑tropic virus and had a tropism result at failure; 7 of these subjects had evidence of maraviroc phenotypic resistance defined as concentration‑response curves that did not reach 95% inhibition. One additional subject had a greater than or equal to 3‑fold shift in the EC50 value for maraviroc at the time of failure. A clonal analysis of the V3 loop amino acid envelope sequences was performed from 6 of the 7 subjects. Changes in V3 loop amino acid sequence differed between each of these different subjects, even for those infected with the same virus clade, suggesting that there are multiple diverse pathways to maraviroc resistance. The subjects who failed with CCR5‑tropic virus and without a detectable maraviroc shift in susceptibility were not evaluated for genotypic resistance.
Of the 32 maraviroc virologic failures failing with CCR5‑tropic virus, 20 (63%) also had genotypic and/or phenotypic resistance to background drugs in the regimen (lamivudine, zidovudine).
Tropism: In both treatment‑experienced and treatment‑naive subjects, detection of CXCR4‑using virus prior to initiation of therapy has been associated with a reduced virologic response to maraviroc.
Antiretroviral Treatment‑Experienced Subjects (Trials A4001027 and A4001028): In the majority of cases, treatment failure on maraviroc was associated with detection of CXCR4‑using virus (i.e., CXCR4- or dual/mixed‑tropic) which was not detected by the tropism assay prior to treatment. CXCR4‑using virus was detected at failure in approximately 55% of subjects who failed treatment on maraviroc by Week 48, as compared with 9% of subjects who experienced treatment failure in the placebo arm. To investigate the likely origin of the on‑treatment CXCR4‑using virus, a detailed clonal analysis was conducted on virus from 20 representative subjects (16 subjects from the maraviroc arms and 4 subjects from the placebo arm) in whom CXCR4‑using virus was detected at treatment failure. From analysis of amino acid sequence differences and phylogenetic data, it was determined that CXCR4‑using virus in these subjects emerged from a low level of pre-existing CXCR4‑using virus not detected by the tropism assay (which is population-based) prior to treatment rather than from a co-receptor switch from CCR5‑tropic virus to CXCR4‑using virus resulting from mutation in the virus.
Detection of CXCR4‑using virus prior to initiation of therapy has been associated with a reduced virological response to maraviroc. Furthermore, subjects failing twice-daily maraviroc at Week 48 with CXCR4‑using virus had a lower median increase in CD4+ cell counts from baseline (+41 cells per mm3) than those subjects failing with CCR5‑tropic virus (+162 cells per mm3). The median increase in CD4+ cell count in subjects failing in the placebo arm was +7 cells per mm3.
Antiretroviral Treatment‑Naive Subjects (Trial A4001026): In a 96‑week trial of antiretroviral treatment‑naive subjects, 14% (12 of 85) who had only CCR5‑tropic virus at screening with an enhanced sensitivity tropism assay (TROFILE) and failed therapy on maraviroc had CXCR4‑using virus at the time of treatment failure. A detailed clonal analysis was conducted in 2 previously antiretroviral treatment‑naive subjects enrolled in a Phase 2a monotherapy trial who had CXCR4‑using virus detected after 10 days’ treatment with maraviroc. Consistent with the detailed clonal analysis conducted in treatment‑experienced subjects, the CXCR4‑using variants appear to emerge from outgrowth of a pre‑existing undetected CXCR4‑using virus. Screening with an enhanced sensitivity tropism assay reduced the number of maraviroc virologic failures with CXCR4- or dual/mixed‑tropic virus at failure to 12 compared with 24 when screening with the original tropism assay. All but one (11 of 12; 92%) of the maraviroc failures failing with CXCR4- or dual/mixed‑tropic virus also had genotypic and phenotypic resistance to the background drug lamivudine at failure and 33% (4 of 12) developed zidovudine-associated resistance substitutions.
Subjects who had only CCR5‑tropic virus at baseline and failed maraviroc therapy with CXCR4‑using virus had a median increase in CD4+ cell counts from baseline of +113 cells per mm3 while those subjects failing with CCR5‑tropic virus had an increase of +135 cells per mm3. The median increase in CD4+ cell count in subjects failing in the efavirenz arm was +95 cells per mm3.
Antiretroviral Treatment‑Experienced Pediatric Subjects (Trial A4001031): In the Week 48 analysis of Trial A4001031 (n = 103), the mechanisms of resistance to maraviroc observed in the treatment-experienced pediatric population were similar to those observed in adult populations: reasons for virologic failure included failing with CXCR4- or dual/mixed-tropic virus, evidence of reduced maraviroc susceptibility as measured by a decrease in maximal percentage inhibition (MPI), and emergence of resistance to background drug in the regimen.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Long-term oral carcinogenicity studies of maraviroc were carried out in rasH2 transgenic mice (6 months) and in rats for up to 96 weeks (females) and 104 weeks (males). No drug-related increases in tumor incidence were found in mice at 1,500 mg per kg per day and in male and female rats at 900 mg per kg per day. The highest exposures in rats were approximately 11 times those observed in humans at the therapeutic dose of 300 mg twice daily for the treatment of HIV-1 infection.
Mutagenesis
Maraviroc was not genotoxic in the reverse mutation bacterial test (Ames test in Salmonella and E. coli), a chromosome aberration test in human lymphocytes, and mouse bone marrow micronucleus test.
Impairment of Fertility
Maraviroc did not impair mating or fertility of male or female rats and did not affect sperm of treated male rats at approximately 20-fold higher exposures (AUC) than in humans given the recommended 300-mg twice-daily dose.
-
14 CLINICAL STUDIES
14.1 Clinical Studies in Adult Subjects
The clinical efficacy and safety of SELZENTRY are derived from analyses of data from 3 trials in adult subjects infected with CCR5-tropic HIV-1: Trials A4001027 and A4001028 in antiretroviral treatment-experienced adult subjects and Trial A4001026 in treatment-naive subjects. These trials were supported by a 48-week trial in antiretroviral treatment-experienced adult subjects infected with dual/mixed-tropic HIV-1, Trial A4001029.
Trials in CCR5-Tropic, Treatment-Experienced Subjects
Trials A4001027 and A4001028 were double-blind, randomized, placebo-controlled, multicenter trials in subjects infected with CCR5-tropic HIV-1. Subjects were required to have an HIV-1 RNA greater than 5,000 copies per mL despite at least 6 months of prior therapy with at least 1 agent from 3 of the 4 antiretroviral drug classes (greater than or equal to 1 NRTI, greater than or equal to 1 NNRTI, greater than or equal to 2 PIs, and/or enfuvirtide) or documented resistance to at least 1 member of each class. All subjects received an optimized background regimen consisting of 3 to 6 antiretroviral agents (excluding low-dose ritonavir) selected on the basis of the subject’s prior treatment history and baseline genotypic and phenotypic viral resistance measurements. In addition to the optimized background regimen, subjects were then randomized in a 2:2:1 ratio to SELZENTRY 300 mg once daily, SELZENTRY 300 mg twice daily, or placebo. Doses were adjusted based on background therapy as described in Dosage and Administration (2), Table 1.
In the pooled analysis for Trials A4001027 and A4001028, the demographics and baseline characteristics of the treatment groups were comparable (Table 16). Of the 1,043 subjects with a CCR5-tropism result at screening, 7.6% had a dual/mixed-tropism result at the baseline visit 4 to 6 weeks later. This illustrates the background change from CCR5- to dual/mixed-tropism result over time in this treatment-experienced population, prior to a change in antiretroviral regimen or administration of a CCR5 co-receptor antagonist.
Table 16. Demographic and Baseline Characteristics of Subjects in Trials A4001027 and A4001028 NNRTI = Non-nucleoside reverse transcriptase inhibitors; NRTI = nucleoside reverse transcriptase inhibitors; OBT = optimized background therapy; PI = protease inhibitor.
a OSS - Sum of active drugs in OBT based on combined information from genotypic and phenotypic testing.
b Resistance substitutions based on IAS guidelines.1SELZENTRY
Twice Daily
(n = 426)
Placebo
(n = 209)
Age (years)
Mean (range)
46.3 (21-73)
45.7 (29-72)
Sex:
Male
382 (89.7%)
185 (88.5%)
Female
44 (10.3%)
24 (11.5%)
Race:
White
363 (85.2%)
178 (85.2%)
Black
51 (12.0%)
26 (12.4%)
Other
12 (2.8%)
5 (2.4%)
Region:
U.S.
276 (64.8%)
135 (64.6%)
Non-U.S.
150 (35.2%)
74 (35.4%)
Subjects with previous enfuvirtide use
142 (33.3%)
62 (29.7%)
Subjects with enfuvirtide as part of OBT
182 (42.7%)
91 (43.5%)
Baseline plasma HIV-1 RNA (log10 copies/mL)
Mean (range)
4.85 (2.96-6.88)
4.86 (3.46-7.07)
Subjects with screening viral load >100,000 copies/mL
179 (42.0%)
84 (40.2%)
Baseline CD4+ cell count (cells/mm3)
Median (range)
167 (2-820)
171 (1-675)
Subjects with baseline CD4+ cell count ≤200 cells/mm3)
250 (58.7%)
118 (56.5%)
Subjects with Overall Susceptibility Score (OSS):a
0
57 (13.4%)
35 (16.7%)
1
136 (31.9%)
44 (21.1%)
2
104 (24.4%)
59 (28.2%)
≥3
125 (29.3%)
66 (31.6%)
Subjects with enfuvirtide resistance substitutions
90 (21.2%)
45 (21.5%)
Median number of resistance-associated:b
PI substitutions
10
10
NNRTI substitutions
1
1
NRTI substitutions
6
6
The Week 48 results for the pooled Trials A4001027 and A4001028 are shown in Table 17.
Table 17. Outcomes of Randomized Treatment at Week 48 in Trials A4001027 and A4001028 a One additional subject died while receiving open-label therapy with SELZENTRY subsequent to discontinuing double-blind placebo due to insufficient response. Outcome
SELZENTRY
Twice Daily
(n = 426)
Placebo
(n = 209)
Mean Difference
Mean change from Baseline to Week 48 in HIV-1 RNA (log10 copies/mL)
-1.84
-0.78
-1.05
<400 copies/mL at Week 48
239 (56%)
47 (22%)
34%
<50 copies/mL at Week 48
194 (46%)
35 (17%)
29%
Discontinuations:
Insufficient clinical response
97 (23%)
113 (54%)
–
Adverse events
19 (4%)
11 (5%)
–
Other
27 (6%)
18 (9%)
–
Subjects with treatment-emergent CDC Category C events
22 (5%)
16 (8%)
–
Deaths (during trial or within 28 days of last dose)
9 (2%)a
1 (0.5%)
–
After 48 weeks of therapy, the proportions of subjects with HIV-1 RNA less than 400 copies per mL receiving SELZENTRY compared with placebo were 56% and 22%, respectively. The mean changes in plasma HIV-1 RNA from baseline to Week 48 were –1.84 log10 copies per mL for subjects receiving SELZENTRY + OBT compared with –0.78 log10 copies per mL for subjects receiving OBT only. The mean increase in CD4+ cell count was higher on SELZENTRY twice daily + OBT (124 cells per mm3) than on placebo + OBT (60 cells per mm3).
Trial in Dual/Mixed-Tropic, Treatment-Experienced Subjects
Trial A4001029 was an exploratory, randomized, double-blind, multicenter trial to determine the safety and efficacy of SELZENTRY in subjects infected with dual/mixed co-receptor tropic HIV-1. The inclusion/exclusion criteria were similar to those for Trials A4001027 and A4001028 above and the subjects were randomized in a 1:1:1 ratio to SELZENTRY once daily, SELZENTRY twice daily, or placebo. No increased risk of infection or HIV-1 disease progression was observed in the subjects who received SELZENTRY. Use of SELZENTRY was not associated with a significant decrease in HIV-1 RNA compared with placebo in these subjects and no adverse effect on CD4+ cell count was noted.
Trial in Treatment-Naive Subjects
Trial A4001026 was a randomized, double-blind, multicenter trial in subjects infected with CCR5-tropic HIV-1 classified by the original TROFILE tropism assay. Subjects were required to have plasma HIV-1 RNA greater than or equal to 2,000 copies per mL and could not have: 1) previously received any antiretroviral therapy for greater than 14 days, 2) an active or recent opportunistic infection or a suspected primary HIV-1 infection, or 3) phenotypic or genotypic resistance to zidovudine, lamivudine, or efavirenz. Subjects were randomized in a 1:1:1 ratio to SELZENTRY 300 mg once daily, SELZENTRY 300 mg twice daily, or efavirenz 600 mg once daily, each in combination with lamivudine/zidovudine. The efficacy and safety of SELZENTRY are based on the comparison of SELZENTRY twice daily versus efavirenz. In a pre-planned interim analysis at 16 weeks, SELZENTRY 300 mg once daily failed to meet the pre-specified criteria for demonstrating non-inferiority and was discontinued.
The demographic and baseline characteristics of the maraviroc and efavirenz treatment groups were comparable (Table 18). Subjects were stratified by screening HIV-1 RNA levels and by geographic region. The median CD4+ cell counts and mean HIV-1 RNA at baseline were similar for both treatment groups.
Table 18. Demographic and Baseline Characteristics of Subjects in Trial A4001026 SELZENTRY
300 mg Twice Daily + Lamivudine/Zidovudine(n = 360)
Efavirenz
600 mg Once Daily + Lamivudine/Zidovudine(n = 361)
Age (years):
Mean
36.7
37.4
Range
20-69
18-77
Female, n%
104 (29)
102 (28)
Race, n%:
White
204 (57)
198 (55)
Black
123 (34)
133 (37)
Asian
6 (2)
5 (1)
Other
27 (8)
25 (7)
Median (range) CD4+ cell count (cells/microL)
241 (5-1,422)
254 (8-1,053)
Median (range) HIV-1 RNA (log10 copies/mL)
4.9 (3-7)
4.9 (3-7)
The treatment outcomes at 96 weeks for Trial A4001026 are shown in Table 19. Treatment outcomes are based on reanalysis of the screening samples using a more sensitive tropism assay, enhanced sensitivity TROFILE HIV tropism assay, which became available after the Week 48 analysis; approximately 15% of the subjects identified as CCR5-tropic in the original analysis had dual/mixed- or CXCR4-tropic virus. Screening with enhanced sensitivity version of the TROFILE tropism assay reduced the number of maraviroc virologic failures with CXCR4- or dual/mixed-tropic virus at failure to 12 compared with 24 when screening with the original TROFILE HIV tropism assay.
Table 19. Trial Outcome (Snapshot) at Week 96 Using Enhanced Sensitivity Assaya a The total number of subjects (311, 303) in Table 19 represents the subjects who had a CCR5-tropic virus in the reanalysis of screening samples using the more sensitive tropism assay. This reanalysis reclassified approximately 15% of subjects shown in Table 18 as having dual/mixed- or CXCR4-tropic virus. These numbers are different than those presented in Table 18 because the numbers in Table 18 reflect the subjects with CCR5-tropic virus according to the original tropism assay.
b Week 48 results: Virologic responders (less than 400): 228 of 311 (73%) in SELZENTRY, 219 of 303 (72%) in efavirenz;
Virologic responders (less than 50): 213 of 311 (69%) in SELZENTRY, 207 of 303 (68%) in efavirenz.
c Other reasons for discontinuation include lost to follow-up, withdrawn, protocol violation, and other.Outcome at Week 96b
SELZENTRY
300 mg Twice Daily + Lamivudine/Zidovudine(n = 311)
n (%)
Efavirenz
600 mg Once Daily +Lamivudine/Zidovudine
(n = 303)
n (%)
Virologic Responders:
(HIV-1 RNA <400 copies/mL)
199 (64)
195 (64)
Virologic Failure:
Non-sustained HIV-1 RNA suppression
39 (13)
22 (7)
HIV-1 RNA never suppressed
9 (3)
1 (<1)
Virologic Responders:
(HIV-1 RNA <50 copies/mL)
183 (59)
190 (63)
Virologic Failure:
Non-sustained HIV-1 RNA suppression
43 (14)
25 (8)
HIV-1 RNA never suppressed
21 (7)
3 (1)
Discontinuations due to:
Adverse events
19 (6)
47 (16)
Death
2 (1)
2 (1)
Otherc
43 (14)
36 (12)
The median increase from baseline in CD4+ cell counts at Week 96 was 184 cells per mm3 for the arm receiving SELZENTRY compared with 155 cells per mm3 for the efavirenz arm.
14.2 Clinical Studies in Pediatric Subjects
Trial in CCR5-Tropic, Treatment-Experienced Subjects
Trial A4001031 is an open-label, multicenter trial in pediatric subjects aged 2 to less than 18 years infected with only CCR5‑tropic HIV‑1. Subjects were required to have HIV‑1 RNA greater than 1,000 copies per mL at screening. All subjects (n = 103) received SELZENTRY twice daily and OBT. Dosing of SELZENTRY was based on BSA and doses were adjusted based on whether the subject was receiving potent CYP3A inhibitors and/or inducers.
The population was 52% female and 69% black, with mean age of 10 years (range: 2 to 17 years). At baseline, mean plasma HIV-1 RNA was 4.4 log10 copies per mL (range: 2.4 to 6.2 log10 copies per mL), mean CD4+ cell count was 551 cells per mm3 (range: 1 to 1,654 cells per mm3), and mean CD4+ percent was 21% (range: 0% to 42%).
At 48 weeks, 48% of subjects treated with SELZENTRY and OBT achieved plasma HIV-1 RNA less than 48 copies per mL and 65% of subjects achieved plasma HIV-1 RNA less than 400 copies per mL. The mean CD4+ cell count (percent) increase from baseline to Week 48 was 247 cells per mm3 (5%).
- 15 REFERENCES
- 16 HOW SUPPLIED/STORAGE AND HANDLING
-
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Medication Guide and Instructions for Use).
Hepatotoxicity
Inform patients that hepatotoxicity, including life-threatening cases, has been reported with SELZENTRY; therefore, it is important to inform the healthcare professional if patients have underlying hepatitis B or C or elevations in liver-associated tests prior to treatment. Inform patients to stop SELZENTRY and seek medical evaluation immediately if they develop signs or symptoms of hepatitis or allergic reaction following use of SELZENTRY. Advise patients that laboratory tests for liver enzymes and bilirubin will be ordered prior to starting SELZENTRY, at other times during treatment, and if they develop severe rash or signs and symptoms of hepatitis or an allergic reaction on treatment [see Dosage and Administration (2.1), Warnings and Precautions (5.1, 5.2)].
Cardiovascular Events
When administering SELZENTRY in patients with cardiovascular comorbidities, a history of postural hypotension or receiving concomitant medication known to lower blood pressure, advise patients that they may be at increased risk for cardiovascular events. Advise patients to avoid driving or operating machinery if they experience dizziness while taking SELZENTRY [see Warnings and Precautions (5.3)].
Drug Interactions
Advise patients to inform their healthcare provider of concomitant HIV medications as dosage of SELZENTRY may be modified depending on other HIV medications taken with SELZENTRY. Advise patients that coadministration of SELZENTRY with St. John’s wort is not recommended as it can lead to loss of virologic response and possible resistance to SELZENTRY [see Dosage and Administration (2.2), Drug Interactions (7.1)].
Missed Dosage
Inform patients that it is important to take SELZENTRY in combination with other antiretroviral medications on a regular dosing schedule with or without food. Advise patients to avoid missing doses as it can result in development of resistance. Instruct patients that if they miss a dose, to take it as soon as they remember. Advise patients not to double their next dose or take more than the prescribed dose [see Dosage and Administration (2.2)].
Pregnancy
Inform patients that there is insufficient data on the safety of SELZENTRY in pregnancy. Inform patients that there is an antiretroviral pregnancy registry that monitors pregnancy outcomes in women exposed to SELZENTRY during pregnancy [see Use in Specific Populations (8.1)].
Lactation
Instruct women with HIV-1 infection not to breastfeed because HIV-1 can be passed to the baby in breast milk [see Use in Specific Populations (8.2)].
SELZENTRY and COMBIVIR are trademarks owned by or licensed to the ViiV Healthcare group of companies.
TROFILE is a trademark owned by or licensed to Monogram BioSciences, Inc., and is not owned by or licensed to the ViiV Healthcare group of companies. The maker of this brand is not affiliated with and does not endorse the ViiV Healthcare group of companies or its products.
Manufactured for:
ViiV Healthcare
Durham, NC 27701
©2022 ViiV Healthcare group of companies or its licensor.
SEL:16PI
PHARMACIST‑DETACH HERE AND GIVE MEDICATION GUIDE TO PATIENT
- _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _
-
MEDICATION GUIDE
MEDICATION GUIDE
SELZENTRY (sell-ZEN-tree)
(maraviroc)
tablets
SELZENTRY (sell-ZEN-tree)
(maraviroc)
oral solution
What is the most important information I should know about SELZENTRY?
SELZENTRY can cause serious side effects including serious liver problems (liver toxicity). Some people who take SELZENTRY can develop a severe rash or an allergic reaction before liver problems happen and may be life-threatening. Stop taking SELZENTRY and call your healthcare provider right away if you get any of the following signs or symptoms of liver problems:
- •
- an itchy rash on your body (allergic reaction)
- •
- your skin or the white part of your eyes turns yellow (jaundice)
- •
- dark or “tea-colored” urine
- •
- vomiting
- •
- pain, aching, or tenderness on the right side of your stomach area
Your healthcare provider will do blood tests to check your liver before you begin treatment with SELZENTRY and as needed during treatment with SELZENTRY.
What is SELZENTRY?
SELZENTRY is a prescription Human Immunodeficiency Virus-1 (HIV-1) medicine given with other HIV-1 medicines to treat CCR5-tropic HIV-1 infection in adults and children weighing at least 4.4 lb (2 kg).
HIV-1 is the virus that causes Acquired Immune Deficiency Syndrome (AIDS).
Use of SELZENTRY is not recommended in people with dual/mixed- or CXCR4‑tropic HIV‑1.
SELZENTRY should not be used in premature newborns or children weighing less than 4.4 pounds (2 kg).
Do not take SELZENTRY if you have severe kidney problems or are on hemodialysis and are also taking certain other medications.
Before you take SELZENTRY, tell your healthcare provider about all of your medical conditions, including if you:
- •
- have or have had liver problems including hepatitis B or C virus infection.
- •
- have heart problems.
- •
- have kidney problems.
- •
- have low blood pressure or take medicines to lower blood pressure.
- •
- are pregnant or plan to become pregnant. It is not known if SELZENTRY may harm your unborn baby.
Pregnancy Registry. There is a pregnancy registry for women who take SELZENTRY during pregnancy. The purpose of this registry is to collect information about the health of you and your baby. Talk to your healthcare provider about how you can take part in this registry. - •
- are breastfeeding or plan to breastfeed. Do not breastfeed if you take SELZENTRY. You should not breastfeed if you have HIV-1 because of the risk of passing HIV-1 to your baby. Talk to your healthcare provider about the best way to feed your baby.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
- •
- Some medicines may interact with SELZENTRY. Keep a list of your medicines to show your healthcare provider and pharmacist.
- •
- You can ask your healthcare provider or pharmacist for a list of medicines that interact with SELZENTRY.
Do not start taking a new medicine without telling your healthcare provider. Your healthcare provider can tell you if it is safe to take SELZENTRY with other medicines. Your healthcare provider may need to change your dose of SELZENTRY when you take it with certain medicines. You should not take SELZENTRY if you also take St. John’s wort (Hypericum perforatum).
How should I take SELZENTRY?
- •
- Take SELZENTRY exactly as your healthcare provider tells you.
- •
- Do not change your dose or stop taking SELZENTRY without first talking with your healthcare provider.
- •
- If you miss a dose of SELZENTRY, take it as soon as you remember. Do not take 2 doses at the same time. If you are not sure about your dosing, call your healthcare provider.
- •
- Stay under the care of a healthcare provider during treatment with SELZENTRY.
- •
- Swallow SELZENTRY tablets whole. Do not chew the tablets.
- •
- SELZENTRY may be taken with or without food.
- •
- Your healthcare provider will prescribe a dose of SELZENTRY based on your child’s body weight and other medicines they are taking.
- •
- Tell your healthcare provider if your child has trouble swallowing tablets. SELZENTRY comes as tablets or as a liquid (oral solution).
- •
- SELZENTRY oral solution should be given with the supplied press-in bottle adapter and oral dosing syringe. See the Instructions for Use that comes with SELZENTRY oral solution for information about the right way to take a dose.
- •
- Do not run out of SELZENTRY. The virus in your blood may increase and the virus in your blood may become harder to treat. When your supply starts to run low, get more from your healthcare provider or pharmacy.
- •
- If you take too much SELZENTRY, call your healthcare provider or go to the nearest hospital emergency room right away.
What are the possible side effects of SELZENTRY?
SELZENTRY can cause serious side effects including:
- •
- See “What is the most important information I should know about SELZENTRY?”
- •
- Severe skin rash and allergic reactions. Severe and potentially life-threatening skin reactions and allergic reactions have been reported in some people taking SELZENTRY. If you develop a rash with any of the following symptoms, stop using SELZENTRY and contact your healthcare provider right away:
- •
- fever
- •
- generally ill feeling
- •
- muscle aches
- •
- blisters or sores in your mouth
- •
- blisters or peeling of the skin
- •
- redness or swelling of the eyes
- •
- swelling of the mouth or face or lips
- •
- problems breathing
- •
- yellowing of the skin or whites of your eyes
- •
- dark or tea-colored urine
- •
- pain, aching, or tenderness on the right side below the ribs
- •
- loss of appetite
- •
- nausea/vomiting
- •
- Heart problems including heart attack.
- •
- Low blood pressure when standing up (postural hypotension) that can cause dizziness or fainting. You should avoid driving or operating heavy machinery if you have dizziness during treatment with SELZENTRY.
- •
- Changes in your immune system (Immune Reconstitution Syndrome) can happen when you start taking HIV-1 medicines. Your immune system may get stronger and begin to fight infections that have been hidden in your body for a long time. Tell your healthcare provider right away if you develop new symptoms during treatment with SELZENTRY.
- •
- Possible chance of infection or cancer. SELZENTRY affects other immune system cells and therefore may possibly increase your chance for getting other infections or cancer.
The most common side effects of SELZENTRY in adults include colds and cold-like symptoms, cough, fever, rash, bloating and gas, indigestion, constipation, and dizziness.
The most common side effects of SELZENTRY in children include vomiting, abdominal pain, diarrhea, nausea, and dizziness.
The most common side effect of SELZENTRY in newborns is decreased hemoglobin (protein inside red blood cells).
These are not all the possible side effects of SELZENTRY. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1‑800‑FDA‑1088.
How should I store SELZENTRY?
- •
- Store SELZENTRY tablets and oral solution at room temperature between 68°F to 77°F (20°C to 25°C).
- •
- Throw away any unused oral solution 60 days after first opening the bottle.
Keep SELZENTRY and all medicines out of the reach of children.
General information about the safe and effective use of SELZENTRY
Medicines are sometimes prescribed for purposes other than those mentioned in a Medication Guide. Do not use SELZENTRY for a condition for which it was not prescribed. Do not give SELZENTRY to other people, even if they have the same symptoms that you have. It may harm them.
You can ask your pharmacist or healthcare provider for the information about SELZENTRY that is written for health professionals.
What are the ingredients in SELZENTRY?
Active ingredient: maraviroc
Inactive ingredients:
Tablets: Dibasic calcium phosphate (anhydrous), magnesium stearate, microcrystalline cellulose, and sodium starch glycolate. Tablet film-coating contains: FD&C blue #2 aluminum lake, soya lecithin, polyethylene glycol (macrogol 3350), polyvinyl alcohol, talc, and titanium dioxide.
Oral Solution: Citric acid, purified water, sodium benzoate, sodium citrate dihydrate, strawberry flavoring (501440T), and sucralose.
Manufactured for:
ViiV Healthcare
Durham, NC 27701
SELZENTRY is a trademark owned by or licensed to the ViiV Healthcare group of companies.
©2022 ViiV Healthcare group of companies or its licensor.
SEL:10MG
For more information go to www.selzentry.com.
This Medication Guide has been approved by the U.S. Food and Drug Administration.
Revised: 9/2022
-
INSTRUCTIONS FOR USE
- maraviroc tablet, film coated
-
INGREDIENTS AND APPEARANCE
SELZENTRY
maraviroc tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:50090-6238(NDC:49702-223) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MARAVIROC (UNII: MD6P741W8A) (MARAVIROC - UNII:MD6P741W8A) MARAVIROC 150 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS DIBASIC CALCIUM PHOSPHATE (UNII: L11K75P92J) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) FD&C BLUE NO. 2 (UNII: L06K8R7DQK) ALUMINUM OXIDE (UNII: LMI26O6933) HYDROGENATED SOYBEAN LECITHIN (UNII: H1109Z9J4N) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POLYVINYL ALCOHOL, UNSPECIFIED (UNII: 532B59J990) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color BLUE Score no score Shape OVAL Size 12mm Flavor Imprint Code MVC150 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:50090-6238-0 60 in 1 BOTTLE; Type 0: Not a Combination Product 11/29/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA022128 05/16/2011 Labeler - A-S Medication Solutions (830016429) Establishment Name Address ID/FEI Business Operations A-S Medication Solutions 830016429 RELABEL(50090-6238)