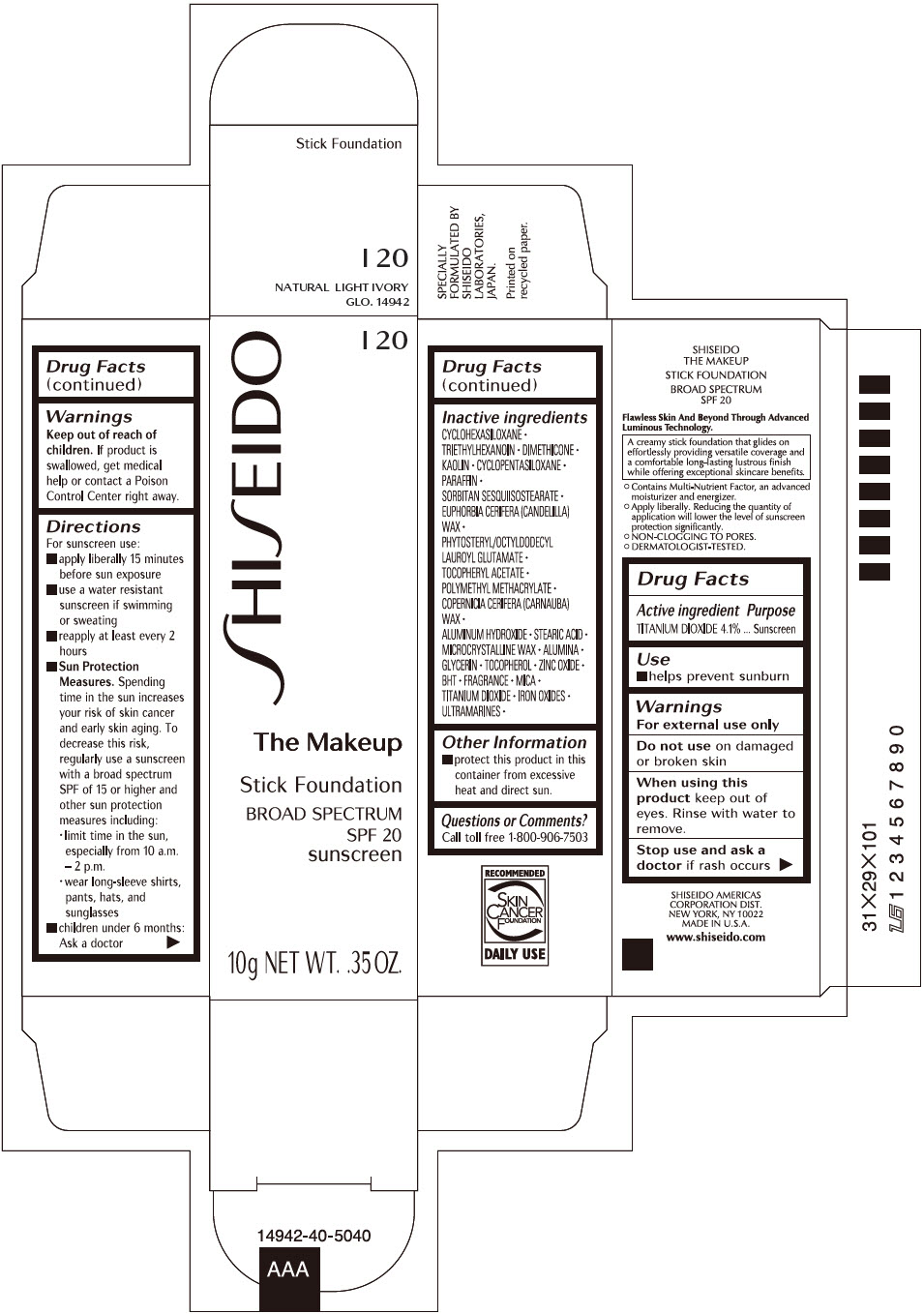
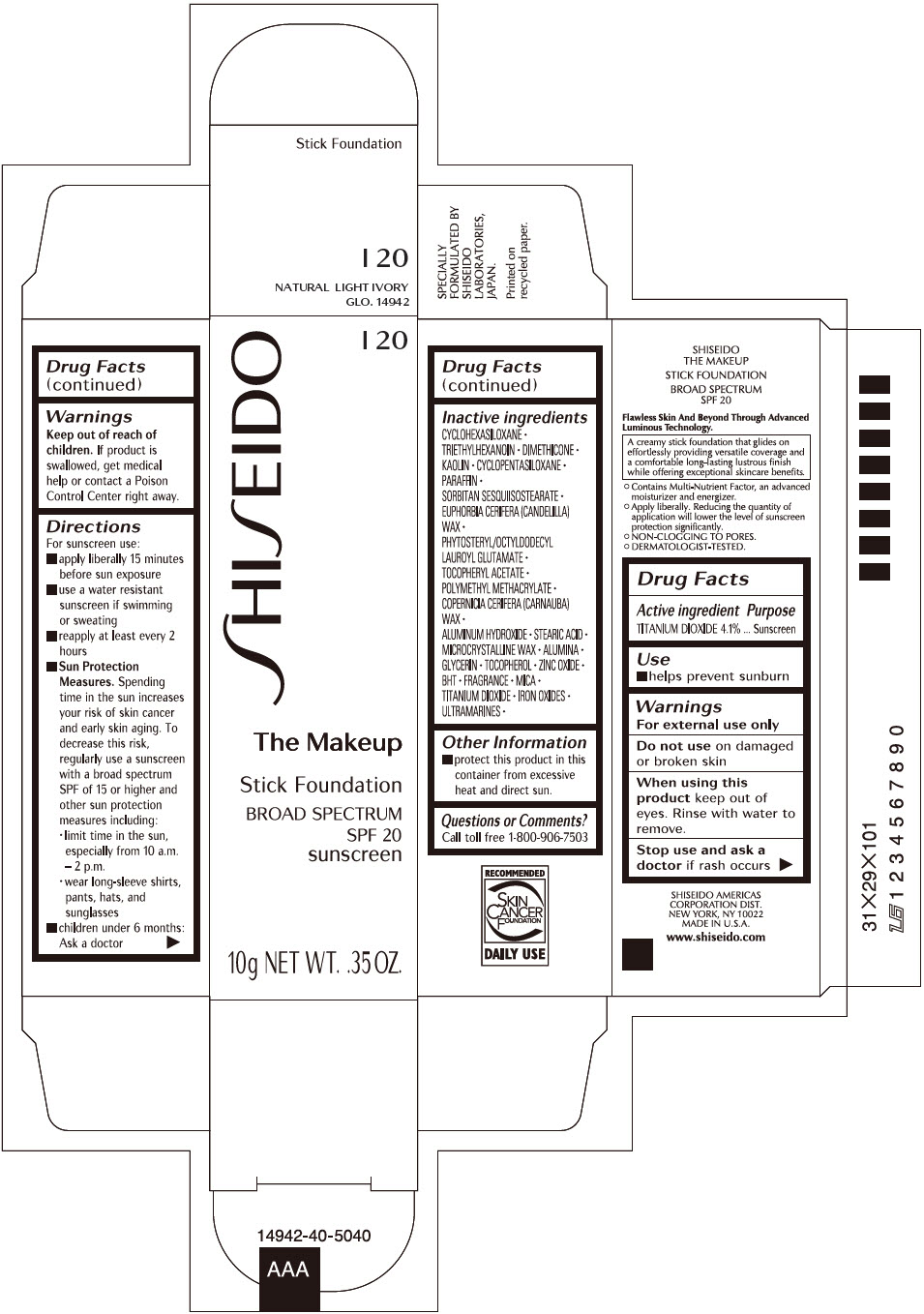
Label: SHISEIDO THE MAKEUP FOUNDATION I20- titanium dioxide stick
SHISEIDO THE MAKEUP FOUNDATION I40- titanium dioxide stick
SHISEIDO THE MAKEUP FOUNDATION I60- titanium dioxide stick
SHISEIDO THE MAKEUP FOUNDATION B20- titanium dioxide stick
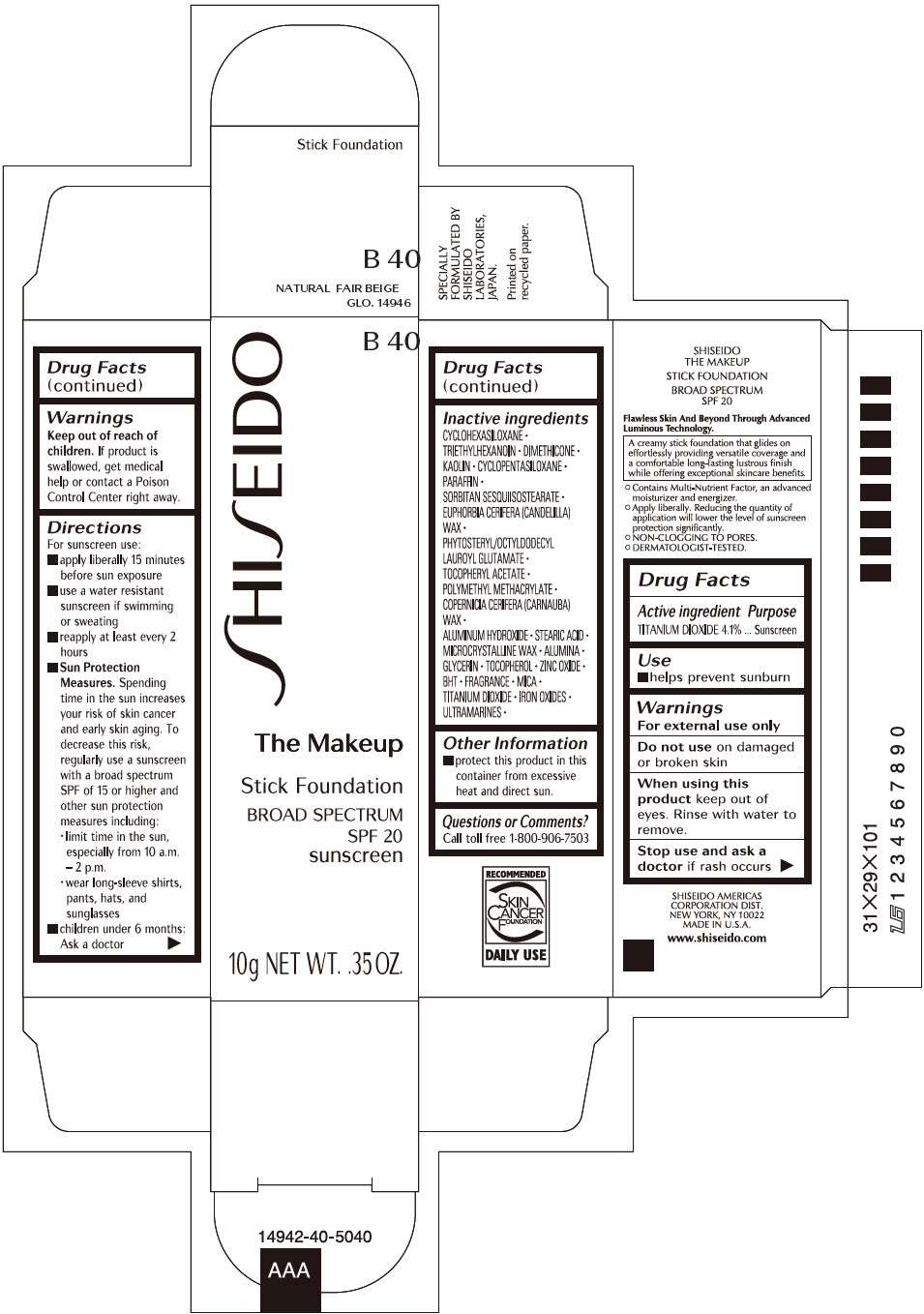
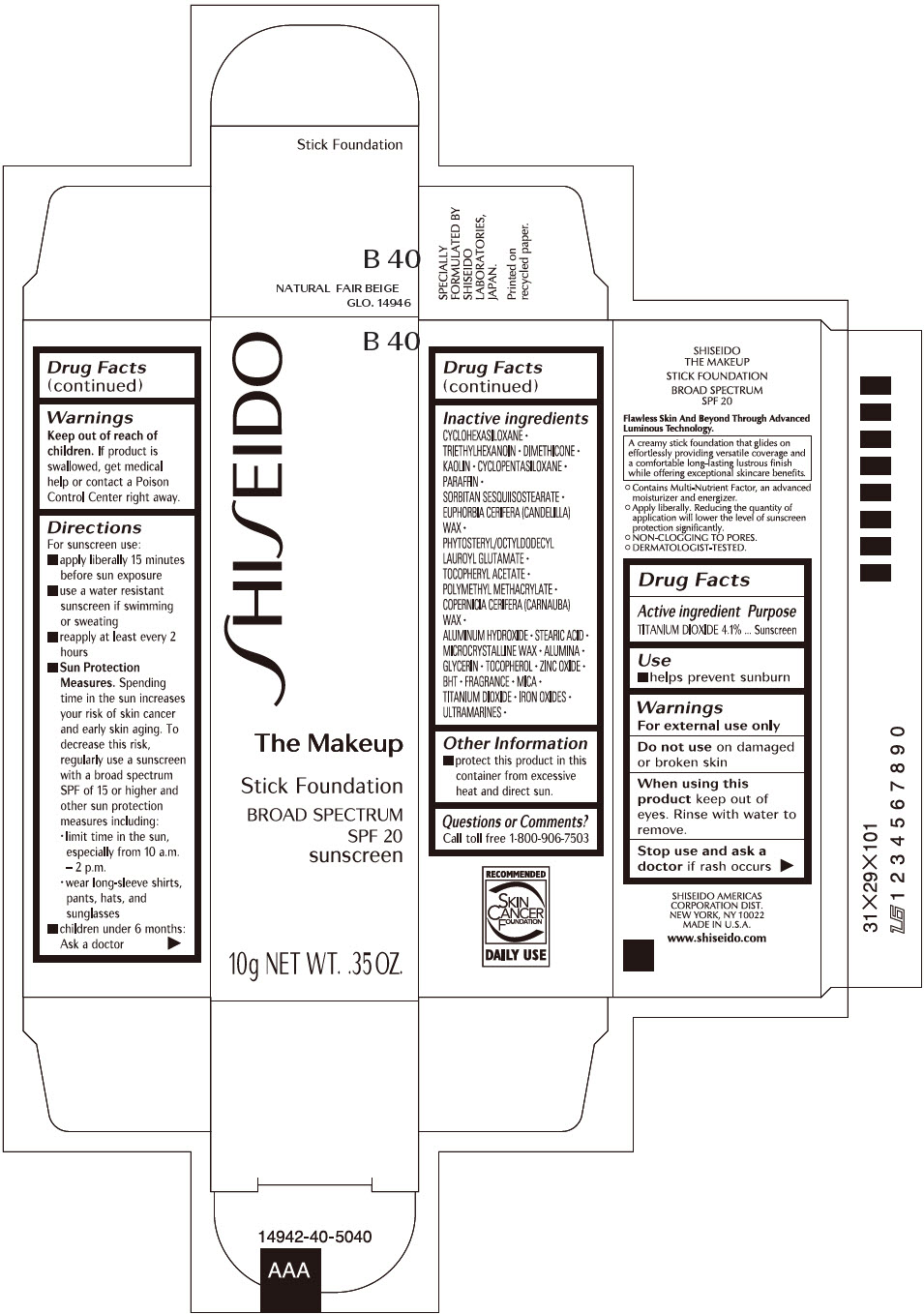
SHISEIDO THE MAKEUP FOUNDATION B40- titanium dioxide stick
SHISEIDO THE MAKEUP FOUNDATION B60- titanium dioxide stick
-
NDC Code(s):
58411-458-20,
58411-459-20,
58411-460-20,
58411-461-20, view more58411-462-20, 58411-534-20
- Packager: SHISEIDO AMERICAS CORPORATION
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated December 29, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredients
- Purpose
- Uses
- Warnings
-
Directions
For sunscreen use:
- apply liberally 15 minutes before sun exposure
- use a water resistant sunscreen if swimming or sweating
- reapply at least every 2 hours
- children under 6 months: ask a doctor
-
Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a broad spectrum SPF of 15 or higher and other sun protection measures including:
- limit time in the sun, especially from 10 a.m. – 2 p.m.
- wear long-sleeve shirts, pants, hats, and sunglasses
- Children under 6 months: Ask a doctor
-
Inactive Ingredients
CYCLOHEXASILOXANE ∙ TRIETHYLHEXANOIN ∙ DIMETHICONE ∙ KAOLIN ∙ CYCLOPENTASILOXANE ∙ PARAFFIN ∙ SORBITAN SESQUIISOSTEARATE ∙ EUPHORBIA CERIFERA (CANDELILLA) WAX ∙ PHYTOSTERYL/OCTYLDODECYL LAUROYL GLUTAMATE ∙ TOCOPHERYL ACETATE ∙ POLYMETHYL METHACRYLATE ∙ COPERNICIA CERIFERA (CARNAUBA) WAX ∙ ALUMINUM HYDROXIDE ∙ STEARIC ACID ∙ MICROCRYSTALLINE WAX ∙ ALUMINA ∙ GLYCERIN ∙ TOCOPHEROL ∙ ZINC OXIDE ∙ BHT ∙ FRAGRANCE ∙ MICA ∙ TITANIUM DIOXIDE ∙ IRON OXIDES ∙ ULTRAMARINES ∙
- Other information
- Questions or comments?
- PRINCIPAL DISPLAY PANEL - 10 g Cartridge Carton - I 20
- PRINCIPAL DISPLAY PANEL - 10 g Cartridge Carton - I 40
- PRINCIPAL DISPLAY PANEL - 10 g Cartridge Carton - I 60
- PRINCIPAL DISPLAY PANEL - 10 g Cartridge Carton - B 20
- PRINCIPAL DISPLAY PANEL - 10 g Cartridge Carton - B 40
- PRINCIPAL DISPLAY PANEL - 10 g Cartridge Carton - B 60
-
INGREDIENTS AND APPEARANCE
SHISEIDO THE MAKEUP FOUNDATION I20
titanium dioxide stickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:58411-458 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 400 mg in 10 g Inactive Ingredients Ingredient Name Strength CYCLOMETHICONE 6 (UNII: XHK3U310BA) TRIETHYLHEXANOIN (UNII: 7K3W1BIU6K) DIMETHICONE (UNII: 92RU3N3Y1O) KAOLIN (UNII: 24H4NWX5CO) CYCLOMETHICONE 5 (UNII: 0THT5PCI0R) PARAFFIN (UNII: I9O0E3H2ZE) SORBITAN SESQUIISOSTEARATE (UNII: VU97D01BF9) CANDELILLA WAX (UNII: WL0328HX19) PHYTOSTERYL/OCTYLDODECYL LAUROYL GLUTAMATE (UNII: 65954KGO9Q) .ALPHA.-TOCOPHEROL ACETATE, DL- (UNII: WR1WPI7EW8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) ALUMINUM HYDROXIDE (UNII: 5QB0T2IUN0) STEARIC ACID (UNII: 4ELV7Z65AP) MICROCRYSTALLINE WAX (UNII: XOF597Q3KY) ALUMINUM OXIDE (UNII: LMI26O6933) GLYCERIN (UNII: PDC6A3C0OX) .ALPHA.-TOCOPHEROL (UNII: H4N855PNZ1) ZINC OXIDE (UNII: SOI2LOH54Z) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) MICA (UNII: V8A1AW0880) FERRIC OXIDE RED (UNII: 1K09F3G675) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) FERROSOFERRIC OXIDE (UNII: XM0M87F357) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:58411-458-20 1 in 1 CARTON 01/01/2018 1 10 g in 1 CARTRIDGE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 01/01/2018 SHISEIDO THE MAKEUP FOUNDATION I40
titanium dioxide stickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:58411-459 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 400 mg in 10 g Inactive Ingredients Ingredient Name Strength CYCLOMETHICONE 6 (UNII: XHK3U310BA) TRIETHYLHEXANOIN (UNII: 7K3W1BIU6K) DIMETHICONE (UNII: 92RU3N3Y1O) KAOLIN (UNII: 24H4NWX5CO) CYCLOMETHICONE 5 (UNII: 0THT5PCI0R) PARAFFIN (UNII: I9O0E3H2ZE) SORBITAN SESQUIISOSTEARATE (UNII: VU97D01BF9) CANDELILLA WAX (UNII: WL0328HX19) PHYTOSTERYL/OCTYLDODECYL LAUROYL GLUTAMATE (UNII: 65954KGO9Q) .ALPHA.-TOCOPHEROL ACETATE, DL- (UNII: WR1WPI7EW8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) ALUMINUM HYDROXIDE (UNII: 5QB0T2IUN0) STEARIC ACID (UNII: 4ELV7Z65AP) MICROCRYSTALLINE WAX (UNII: XOF597Q3KY) ALUMINUM OXIDE (UNII: LMI26O6933) GLYCERIN (UNII: PDC6A3C0OX) .ALPHA.-TOCOPHEROL (UNII: H4N855PNZ1) ZINC OXIDE (UNII: SOI2LOH54Z) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) MICA (UNII: V8A1AW0880) FERRIC OXIDE RED (UNII: 1K09F3G675) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) FERROSOFERRIC OXIDE (UNII: XM0M87F357) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:58411-459-20 1 in 1 CARTON 01/01/2018 1 10 g in 1 CARTRIDGE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 01/01/2018 SHISEIDO THE MAKEUP FOUNDATION I60
titanium dioxide stickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:58411-460 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 400 mg in 10 g Inactive Ingredients Ingredient Name Strength CYCLOMETHICONE 6 (UNII: XHK3U310BA) TRIETHYLHEXANOIN (UNII: 7K3W1BIU6K) DIMETHICONE (UNII: 92RU3N3Y1O) KAOLIN (UNII: 24H4NWX5CO) CYCLOMETHICONE 5 (UNII: 0THT5PCI0R) PARAFFIN (UNII: I9O0E3H2ZE) SORBITAN SESQUIISOSTEARATE (UNII: VU97D01BF9) CANDELILLA WAX (UNII: WL0328HX19) PHYTOSTERYL/OCTYLDODECYL LAUROYL GLUTAMATE (UNII: 65954KGO9Q) .ALPHA.-TOCOPHEROL ACETATE, DL- (UNII: WR1WPI7EW8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) ALUMINUM HYDROXIDE (UNII: 5QB0T2IUN0) STEARIC ACID (UNII: 4ELV7Z65AP) MICROCRYSTALLINE WAX (UNII: XOF597Q3KY) ALUMINUM OXIDE (UNII: LMI26O6933) GLYCERIN (UNII: PDC6A3C0OX) .ALPHA.-TOCOPHEROL (UNII: H4N855PNZ1) ZINC OXIDE (UNII: SOI2LOH54Z) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) MICA (UNII: V8A1AW0880) FERRIC OXIDE RED (UNII: 1K09F3G675) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) FERROSOFERRIC OXIDE (UNII: XM0M87F357) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:58411-460-20 1 in 1 CARTON 01/01/2018 1 10 g in 1 CARTRIDGE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 01/01/2018 SHISEIDO THE MAKEUP FOUNDATION B20
titanium dioxide stickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:58411-461 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 400 mg in 10 g Inactive Ingredients Ingredient Name Strength CYCLOMETHICONE 6 (UNII: XHK3U310BA) TRIETHYLHEXANOIN (UNII: 7K3W1BIU6K) DIMETHICONE (UNII: 92RU3N3Y1O) KAOLIN (UNII: 24H4NWX5CO) CYCLOMETHICONE 5 (UNII: 0THT5PCI0R) PARAFFIN (UNII: I9O0E3H2ZE) SORBITAN SESQUIISOSTEARATE (UNII: VU97D01BF9) CANDELILLA WAX (UNII: WL0328HX19) PHYTOSTERYL/OCTYLDODECYL LAUROYL GLUTAMATE (UNII: 65954KGO9Q) .ALPHA.-TOCOPHEROL ACETATE, DL- (UNII: WR1WPI7EW8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) ALUMINUM HYDROXIDE (UNII: 5QB0T2IUN0) STEARIC ACID (UNII: 4ELV7Z65AP) MICROCRYSTALLINE WAX (UNII: XOF597Q3KY) ALUMINUM OXIDE (UNII: LMI26O6933) GLYCERIN (UNII: PDC6A3C0OX) .ALPHA.-TOCOPHEROL (UNII: H4N855PNZ1) ZINC OXIDE (UNII: SOI2LOH54Z) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) MICA (UNII: V8A1AW0880) FERRIC OXIDE RED (UNII: 1K09F3G675) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) FERROSOFERRIC OXIDE (UNII: XM0M87F357) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:58411-461-20 1 in 1 CARTON 01/01/2018 1 10 g in 1 CARTRIDGE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 01/01/2018 SHISEIDO THE MAKEUP FOUNDATION B40
titanium dioxide stickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:58411-462 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 400 mg in 10 g Inactive Ingredients Ingredient Name Strength CYCLOMETHICONE 6 (UNII: XHK3U310BA) TRIETHYLHEXANOIN (UNII: 7K3W1BIU6K) DIMETHICONE (UNII: 92RU3N3Y1O) KAOLIN (UNII: 24H4NWX5CO) CYCLOMETHICONE 5 (UNII: 0THT5PCI0R) PARAFFIN (UNII: I9O0E3H2ZE) SORBITAN SESQUIISOSTEARATE (UNII: VU97D01BF9) CANDELILLA WAX (UNII: WL0328HX19) PHYTOSTERYL/OCTYLDODECYL LAUROYL GLUTAMATE (UNII: 65954KGO9Q) .ALPHA.-TOCOPHEROL ACETATE, DL- (UNII: WR1WPI7EW8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) ALUMINUM HYDROXIDE (UNII: 5QB0T2IUN0) STEARIC ACID (UNII: 4ELV7Z65AP) MICROCRYSTALLINE WAX (UNII: XOF597Q3KY) ALUMINUM OXIDE (UNII: LMI26O6933) GLYCERIN (UNII: PDC6A3C0OX) .ALPHA.-TOCOPHEROL (UNII: H4N855PNZ1) ZINC OXIDE (UNII: SOI2LOH54Z) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) MICA (UNII: V8A1AW0880) FERRIC OXIDE RED (UNII: 1K09F3G675) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) FERROSOFERRIC OXIDE (UNII: XM0M87F357) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:58411-462-20 1 in 1 CARTON 01/01/2018 1 10 g in 1 CARTRIDGE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 01/01/2018 SHISEIDO THE MAKEUP FOUNDATION B60
titanium dioxide stickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:58411-534 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 400 mg in 10 g Inactive Ingredients Ingredient Name Strength CYCLOMETHICONE 6 (UNII: XHK3U310BA) TRIETHYLHEXANOIN (UNII: 7K3W1BIU6K) DIMETHICONE (UNII: 92RU3N3Y1O) KAOLIN (UNII: 24H4NWX5CO) CYCLOMETHICONE 5 (UNII: 0THT5PCI0R) PARAFFIN (UNII: I9O0E3H2ZE) SORBITAN SESQUIISOSTEARATE (UNII: VU97D01BF9) CANDELILLA WAX (UNII: WL0328HX19) PHYTOSTERYL/OCTYLDODECYL LAUROYL GLUTAMATE (UNII: 65954KGO9Q) .ALPHA.-TOCOPHEROL ACETATE, DL- (UNII: WR1WPI7EW8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) CARNAUBA WAX (UNII: R12CBM0EIZ) ALUMINUM HYDROXIDE (UNII: 5QB0T2IUN0) STEARIC ACID (UNII: 4ELV7Z65AP) MICROCRYSTALLINE WAX (UNII: XOF597Q3KY) ALUMINUM OXIDE (UNII: LMI26O6933) GLYCERIN (UNII: PDC6A3C0OX) .ALPHA.-TOCOPHEROL (UNII: H4N855PNZ1) ZINC OXIDE (UNII: SOI2LOH54Z) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) MICA (UNII: V8A1AW0880) FERRIC OXIDE RED (UNII: 1K09F3G675) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) FERROSOFERRIC OXIDE (UNII: XM0M87F357) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:58411-534-20 1 in 1 CARTON 01/01/2018 01/31/2022 1 10 g in 1 CARTRIDGE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 01/01/2018 01/31/2022 Labeler - SHISEIDO AMERICAS CORPORATION (193691821) Establishment Name Address ID/FEI Business Operations SHISEIDO AMERICAS INC. 782677132 MANUFACTURE(58411-458, 58411-459, 58411-460, 58411-461, 58411-462, 58411-534) , ANALYSIS(58411-458, 58411-459, 58411-460, 58411-461, 58411-462, 58411-534)