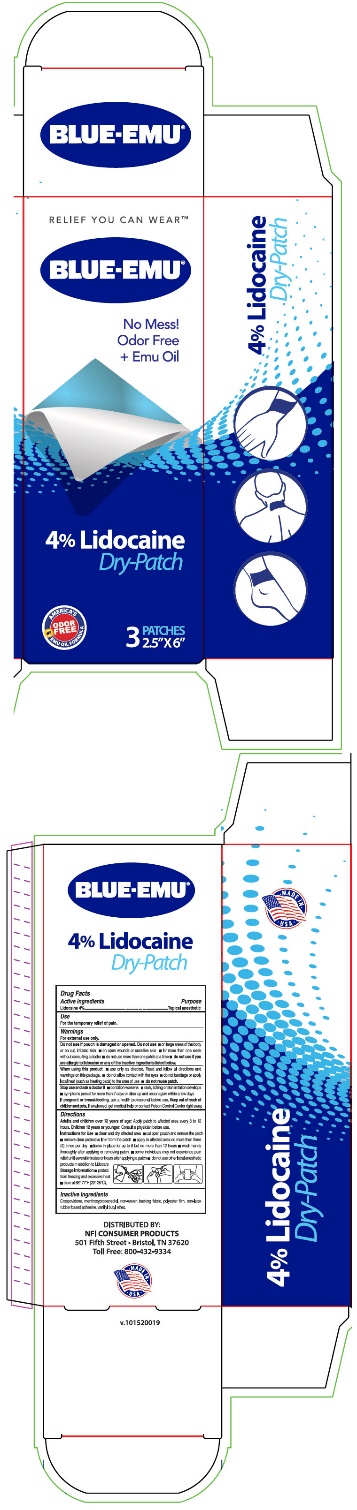
Label: BLUE-EMU LIDOCAINE DRY- lidocaine patch
- NDC Code(s): 69993-425-03
- Packager: NFI, LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated May 1, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredients
- Purpose
- Use
-
Warnings
For external use only.
Do not use if pouch is damaged or opened. Do not use
- •
- on large areas of the body or on cut, irritated skin
- •
- on open wounds or sensitive skin
- •
- for more than one week without consulting a doctor
- •
- do not use more than one patch at a time
- •
- do not use if you are allergic to lidocaine or any of the inactive ingredients listed below.
When using this product
- •
- use only as directed. Read and follow all directions and warnings on this package.
- •
- do not allow contact with the eyes
- •
- do not bandage or apply local heat (such as heating pads) to the area of use
- •
- do not reuse patch.
-
Directions
Instructions for Use
- •
- clean and dry affected area
- •
- cut open pouch and remove the patch
- •
- remove clear protective liner from the patch
- •
- apply to affected area no more than three (3) times per day
- •
- leave in place for up to 8 but no more than 12 hours
- •
- wash hands thoroughly after applying or removing patch
- •
- some individuals may not experience pain relief until several minutes or hours after applying a patch
- •
- do not use other local anesthetic products in addition to Lidocare
- Storage Information
- Inactive Ingredients
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 3 Patch Carton Label
-
INGREDIENTS AND APPEARANCE
BLUE-EMU LIDOCAINE DRY
lidocaine patchProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69993-425 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Lidocaine (UNII: 98PI200987) (Lidocaine - UNII:98PI200987) Lidocaine 22 mg Inactive Ingredients Ingredient Name Strength Isopropyl Alcohol (UNII: ND2M416302) Heptane (UNII: 456148SDMJ) Emu Oil (UNII: 344821WD61) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69993-425-03 3 in 1 CARTON 04/08/2020 1 1 in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 04/08/2020 Labeler - NFI, LLC (121681919) Establishment Name Address ID/FEI Business Operations ProSolus, Inc. 969793079 MANUFACTURE(69993-425)