Label: MINOCYCLINE tablet
- NDC Code(s): 49884-511-01, 49884-512-01, 49884-513-03
- Packager: Par Pharmaceutical, Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated October 5, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
DESCRIPTION
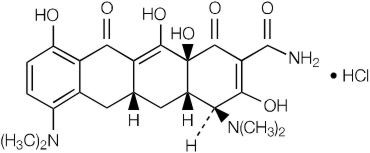
Minocycline hydrochloride, is a semisynthetic derivative of tetracycline, 4,7-Bis (dimethylamino)-1,4,4a,5,5a,6,11,12a-octahydro-3,10,12,12a-tetrahydroxy-1,11-dioxo-2-naphthacenecarboxamide monohydrochloride. Its structural formula is:
Minocycline hydrochloride tablets for oral administration contain minocycline HCl equivalent to 50 mg, 75 mg or 100 mg of minocycline. In addition, 50 mg, 75 mg and 100 mg tablets contain the following inactive ingredients: Microcrystalline Cellulose NF, Lactose Anhydrous NF, Povidone USP, Colloidal Silicon Dioxide NF, Magnesium Stearate NF, and Sodium Starch Glycolate NF. The 50 mg, 75 mg and 100 mg tablets also contain Opadry White which contains: Titanium Dioxide USP, Hypromellose Type 2910 USP, Polyethylene Glycol 400 NF, and Polysorbate 80 NF.
-
CLINICAL PHARMACOLOGY
Following a single dose of one 100 mg tablet of minocycline hydrochloride administered to 28 normal fasting adult volunteers, maximum serum concentrations were attained in 1 to 3 hours (average 1.71 hours) and ranged from 491.71 to 1292.70 ng/mL (average 758.29 ng/mL). The serum half-life in the normal volunteers ranged from 11.38 to 24.31 hours (average 17.03 hours).
When minocycline hydrochloride tablets were given concomitantly with a meal, which included dairy products, the extent of absorption of minocycline hydrochloride tablets was slightly decreased (6%). The peak plasma concentrations were slightly decreased (12%) and delayed by 1.09 hours when administered with food, compared to dosing under fasting conditions. Minocycline HCl may be administered with or without food.
In previous studies with other minocycline dosage forms, the minocycline serum half-life ranged from 11 to 16 hours in 7 patients with hepatic dysfunction, and from 18 to 69 hours in 5 patients with renal dysfunction. The urinary and fecal recovery of minocycline when administered to 12 normal volunteers was one-half to one-third that of other tetracyclines.
Microbiology
The tetracyclines are primarily bacteriostatic and are thought to exert their antimicrobial effect by the inhibition of protein synthesis. The tetracyclines, including minocycline, have similar antimicrobial spectra of activity against a wide range of gram-positive and gram-negative organisms. Cross-resistance of these organisms to tetracyclines is common.
Minocycline has been shown to be active against most strains of the following microorganisms, both in vitro and in clinical infections as described in the INDICATIONS AND USAGE section:
AEROBIC GRAM-POSITIVE MICROORGANISMS:
Because many strains of the following gram-positive microorganisms have been shown to be resistant to tetracyclines, culture and susceptibility testing are especially recommended. Tetracycline antibiotics should not be used for streptococcal diseases unless the organism has been demonstrated to be susceptible. Tetracyclines are not the drug of choice in the treatment of any type of staphylococcal infection.
Bacillus anthracisa
Listeria monocytogenesa
Staphylococcus aureus
Streptococcus pneumoniae
AEROBIC GRAM-NEGATIVE MICROORGANISMS
Bartonella bacilliformis
Brucella species
Calymmatobacterium granulomatis
Campylobacter fetus
Francisella tularensis
Haemophilus ducreyi
Vibrio cholerae
Yersinia pestis
Because many strains of the following groups of gram-negative microorganisms have been shown to be resistant to tetracyclines, culture and susceptibility tests are especially recommended.
Acinetobacter species
Enterobacter aerogenes
Escherichia coli
Haemophilus influenzae
Klebsiella species
Neisseria gonorrhoeaea
Neisseria meningitidisa
Shigella species
“OTHER” MICROORGANISMS
Actinomyces speciesa
Borrelia recurrentis
Chlamydia psittaci
Chlamydia trachomatis
Clostridium speciesa
Entamoeba species
Fusobacterium nucleatum subspecies fusiformea
Mycobacterium marinum
Mycoplasma pneumoniae
Propionibacterium acnes
Rickettsiae
Treponema pallidum subspecies palliduma
Treponema pallidum subspecies pertenuea
Ureaplasma urealyticum
aWhen penicillin is contraindicated, tetracyclines are alternative drugs in the treatment of infections caused by the cited microorganisms.
Susceptibility Tests
Susceptibility testing should be performed with tetracycline since it predicts susceptibility to minocycline. However, certain organisms (e.g., some staphylococci, and Acinetobacter species) may be more susceptible to minocycline and doxycycline than tetracycline.
Dilution techniques:
Quantitative methods are used to determine antimicrobial minimal inhibitory concentrations (MICs). These MICs provide estimates of the susceptibility of bacteria to antimicrobial compounds. The MICs should be determined using a standardized procedure. Standardized procedures are based on a dilution method1,3 (broth or agar) or equivalent with standardized inoculum concentrations and standardized concentrations of tetracycline powder. The MIC values should be interpreted according to the following criteria:
For testing aerobic gram-negative microorganisms (Enterobacteriaceae), Acinetobacter species and Staphylococcus aureus:
MIC (mcg/mL) Interpretation ≤ 4 Susceptible (S) 8 Intermediate (I) ≥ 16 Resistant (R) For testing Haemophilus influenzaeb and Streptococcus pneumoniaec:
MIC (mcg/mL) Interpretation ≤ 2 Susceptible (S) 4 Intermediate (I) ≥ 8 Resistant (R) b. These interpretative standards are applicable only to broth microdilution susceptibility testing with Haemophilus influenzae using Haemophilus Test Medium.1
c. These interpretative standards are applicable only to broth microdilution susceptibility testing using cation-adjusted Muller-Hinton broth with 2 to 5% lysed horse blood.1
For testing Neisseria gonorrhoeaed:
MIC (mcg/mL) Interpretation ≤ 0.25 Susceptible (S) 0.5-1 Intermediate (I) ≥ 2 Resistant (R) d. These interpretative standards are applicable only to agar dilution susceptibility testing using GC agar base and 1% defined growth supplements.1
A report of “Susceptible” indicates that the pathogen is likely to be inhibited if the antimicrobial compound in the blood reaches the concentrations usually achievable. A report of “Intermediate” indicates that the result should be considered equivocal, and, if the microorganism is not fully susceptible to alternative, clinically feasible drugs, the test should be repeated. This category implies possible clinical applicability in body sites where the drug is physiologically concentrated or in situations where high dosage of drug can be used. This category also provides a buffer zone which prevents small uncontrolled technical factors from causing major discrepancies in interpretation. A report of “Resistant” indicates that the pathogen is not likely to be inhibited if the antimicrobial compound in the blood reaches the concentrations usually achievable; other therapy should be selected.
Standardized susceptibility test procedures require the use of laboratory control microorganisms to control the technical aspects of the laboratory procedures. Standard tetracycline powder should provide the following MIC values:
Microorganism MIC (mcg/mL) Escherichia coli ATCC 25922 0.5-2 Enterococcus faecalis ATCC 29212 8-32 Staphylococcus aureus ATCC 29213 0.25-1 Haemophilus influenzae ATCC 49247 4-32 Streptococcus pneumoniae ATCC 49619 0.12-0.5 Neisseria gonorrhoeae ATCC 49226 0.25-1 Diffusion techniques:
Quantitative methods that require measurement of zone diameters also provide reproducible estimates of the susceptibility of bacteria to antimicrobial compounds. One such standardized procedure2,3 requires the use of standardized inoculum concentrations. This procedure uses paper disks impregnated with 30 mcg tetracycline (class disk) or 30 mcg minocycline to test the susceptibility of microorganisms to minocycline.
Reports from the laboratory providing results of the standard single-disk susceptibility test with a 30 mcg tetracycline or minocycline disk should be interpreted according to the following criteria:
For testing aerobic gram-negative microorganisms (Enterobacteriaceae), Acinetobacter species and Staphylococcus aureus:
Zone Diameter (mm) Interpretation ≥ 19 Susceptible (S) 15-18 Intermediate (I) ≤ 14 Resistant (R) For testing Haemophilus influenzaee:
Zone Diameter (mm) Interpretation ≥ 29 Susceptible (S) 26-28 Intermediate (I) ≤ 25 Resistant (R) e. These zone diameter standards are applicable only to susceptibility testing with Haemophilus influenzae using Haemophilus Test Medium and a 30 mcg tetracycline disk.2
For testing Neisseria gonorrhoeaef:
Zone Diameter (mm) Interpretation ≥ 38 Susceptible (S) 31-37 Intermediate (I) ≤ 30 Resistant (R) f. These interpretative standards are applicable only to disk diffusion testing using GC agar and 1% growth supplements, and a 30 mcg tetracycline disk.2
For testing Streptococcus pneumoniaeg:
Zone Diameter (mm) Interpretation ≥ 23 Susceptible (S) 19-22 Intermediate (I) ≤ 18 Resistant (R) g. These interpretative standards are applicable only to disk diffusion testing using Muller-Hinton agar adjusted with 5% sheep blood and a 30 mcg tetracycline class disk.2
For testing Vibrio choleraeh:
Zone Diameter (mm) Interpretation ≥ 19 Susceptible (S) 15-18 Intermediate (I) ≤ 14 Resistant (R) h. These interpretative standards are applicable only to disk diffusion testing performed with a 30 mcg tetracycline class disk.2
Interpretation should be as stated above for results using dilution techniques. Interpretation involves correlation of the diameter obtained in the disk test with the MIC for tetracycline.
As with standardized dilution techniques, diffusion methods require the use of laboratory control microorganisms that are used to control the technical aspects of the laboratory procedures. For the diffusion technique, the 30 mcg tetracycline or minocycline disk should provide the following zone diameters in these laboratory test quality control strains:
Microorganism Zone Diameter Range (mm) Tetracycline Minocycline Escherichia coli ATCC25922 18-25 19-25 Staphylococcus aureus ATCC 29213 24-30 25-30 Haemophilus influenzae ATCC 49247 14-22 --- Neisseria gonorrhoeae ATCC 49226 30-42 --- Streptococcus pneumoniae ATCC 49619 27-31 --- -
INDICATIONS AND USAGE
Minocycline hydrochloride tablets are indicated in the treatment of the following infections due to susceptible strains of the designated microorganisms:
- Rocky Mountain spotted fever, typhus fever and the typhus group, Q fever, rickettsialpox and tick fevers caused by Rickettsiae.
- Respiratory tract infections caused by Mycoplasma pneumoniae.
- Lymphogranuloma venereum caused by Chlamydia trachomatis.
- Psittacosis (Ornithosis) due to Chlamydia psittaci.
- Trachoma caused by Chlamydia trachomatis, although the infectious agent is not always eliminated, as judged by immunofluorescence.
- Inclusion conjunctivitis caused by Chlamydia trachomatis.
- Nongonococcal urethritis, endocervical, or rectal infections in adults caused by Ureaplasma urealyticum or Chlamydia trachomatis.
- Relapsing fever due to Borrelia recurrentis.
- Chancroid caused by Haemophilus ducreyi
- Plague due to Yersinia pestis.
- Tularemia due to Francisella tularensis.
- Cholera caused by Vibrio cholerae.
- Campylobacter fetus infections caused by Campylobacter fetus.
- Brucellosis due to Brucella species (in conjunction with streptomycin).
- Bartonellosis due to Bartonella bacilliformis.
- Granuloma inguinale caused by Calymmatobacterium granulomatis.
Minocycline is indicated for treatment of infections caused by the following gram-negative microorganisms when bacteriologic testing indicates appropriate susceptibility to the drug:
- Escherichia coli.
- Enterobacter aerogenes.
- Shigella species.
- Acinetobacter species.
- Respiratory tract infections caused by Haemophilus influenzae.
- Respiratory tract and urinary tract infections caused by Klebsiella species.
Minocycline hydrochloride tablets are indicated for the treatment of infections caused by the following gram-positive microorganisms when bacteriologic testing indicates appropriate susceptibility to the drug:
- Upper respiratory tract infections caused by Streptococcus pneumoniae
- Skin and skin structure infections caused by Staphylococcus aureus. (Note: Minocycline is not the drug of choice in the treatment of any type of staphylococcal infection.)
When penicillin is contraindicated, minocycline is an alternative drug in the treatment of the following infections:
- Uncomplicated urethritis in men due to Neisseria gonorrhoeae and for the treatment of other gonococcal infections
- Infections in women caused by Neisseria gonorrhoeae.
- Syphilis caused by Treponema pallidum subspecies pallidum.
- Yaws caused by Treponema pallidum subspecies pertenue.
- Listeriosis due to Listeria monocytogenes.
- Anthrax due to Bacillus anthracis.
- Vincent’s infection caused by Fusobacterium fusiforme.
- Actinomycosis caused by Actinomyces israelii.
- Infections caused by Clostridium species.
In acuteintestinal amebiasis, minocycline may be a useful adjunct to amebicides.
In severe acne, minocycline may be useful adjunctive therapy.
Oral minocycline is indicated in the treatment of asymptomatic carriers of Neisseria meningitidis to eliminate the meningococci from the nasopharynx. In order to preserve the usefulness of minocycline in the treatment of asymptomatic meningococcal carriers, diagnostic laboratory procedures, including serotyping and susceptibility testing, should be performed to establish the carrier state and the correct treatment. It is recommended that the prophylactic use of minocycline be reserved for situations in which the risk of meningococcal meningitis is high.
Oral minocycline is not indicated for the treatment of meningococcal infection.
Although no controlled clinical efficacy studies have been conducted, limited clinical data show that oral minocycline hydrochloride has been used successfully in the treatment of infections caused by Mycobacterium marinum.
To reduce the development of drug-resistant bacteria and maintain the effectiveness of, minocycline hydrochloride tablets and other antibacterial drugs, minocycline hydrochloride tablets should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
- CONTRAINDICATIONS
-
WARNINGS
MINOCYCLINE HYDROCHLORIDE TABLETS, LIKE OTHER TETRACYCLINE-CLASS ANTIBIOTICS, CAN CAUSE FETAL HARM WHEN ADMINISTERED TO A PREGNANT WOMAN. IF ANY TETRACYCLINE IS USED DURING PREGNANCY OR IF THE PATIENT BECOMES PREGNANT WHILE TAKING THESE DRUGS, THE PATIENT SHOULD BE APPRISED OF THE POTENTIAL HAZARD TO THE FETUS. THE USE OF DRUGS OF THE TETRACYCLINE CLASS DURING TOOTH DEVELOPMENT (LAST HALF OF PREGNANCY, INFANCY, AND CHILDHOOD TO THE AGE OF 8 YEARS) MAY CAUSE PERMANENT DISCOLORATION OF THE TEETH (YELLOW-GRAY-BROWN).
This adverse reaction is more common during long-term use of the drug but has been observed following repeated short-term courses. Enamel hypoplasia has also been reported. TETRACYCLINE DRUGS, THEREFORE, SHOULD NOT BE USED DURING TOOTH DEVELOPMENT UNLESS OTHER DRUGS ARE NOT LIKELY TO BE EFFECTIVE OR ARE CONTRAINDICATED.
All tetracyclines form a stable calcium complex in any bone-forming tissue. A decrease in the fibula growth rate has been observed in premature human infants given oral tetracycline in doses of 25 mg/kg every six hours. This reaction was shown to be reversible when the drug was discontinued.
Results of animal studies indicate that tetracyclines cross the placenta, are found in fetal tissues, and can have toxic effects on the developing fetus (often related to retardation of skeletal development). Evidence of embryotoxicity has been noted in animals treated early in pregnancy.
Drug Rash with Eosinophilia and Systemic Symptoms (DRESS) including fetal cases have been reported with minocycline use. If this syndrome is recognized, the drug should be discontinued immediately.
The anti-anabolic action of the tetracyclines may cause an increase in BUN. While this is not a problem in those with normal renal function, in patients with significantly impaired function, higher serum levels of tetracycline may lead to azotemia, hyperphosphatemia, and acidosis. Under such conditions, monitoring of creatinine and BUN is recommended, and the total daily dosage should not exceed 200 mg in 24 hours (see DOSAGE AND ADMINISTRATION). If renal impairment exists, even usual oral or parenteral doses may lead to systemic accumulation of the drug and possible liver toxicity.
Photosensitivity manifested by an exaggerated sunburn reaction has been observed in some individuals taking tetracyclines. This has been reported with minocycline.
Central nervous system side effects including light-headedness, dizziness, or vertigo have been reported with minocycline therapy. Patients who experience these symptoms should be cautioned about driving vehicles or using hazardous machinery while on minocycline therapy. These symptoms may disappear during therapy and usually disappear rapidly when the drug is discontinued.
Clostridium difficile associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including minocycline hydrochloride, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibiotic use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibiotic use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibiotic treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
-
PRECAUTIONS
General
As with other antibiotic preparations, use of this drug may result in overgrowth of non-susceptible organisms, including fungi. If superinfection occurs, the antibiotic should be discontinued and appropriate therapy instituted.
Pseudotumor cerebri (benign intracranial hypertension) in adults has been associated with the use of tetracyclines. The usual clinical manifestations are headache and blurred vision. Bulging fontanels have been associated with the use of tetracyclines in infants. While both of these conditions and related symptoms usually resolve after discontinuation of the tetracycline, the possibility for permanent sequelae exists.
Hepatotoxicity has been reported with minocycline; therefore, minocycline should be used with caution in patients with hepatic dysfunction and in conjunction with other hepatotoxic drugs.
Incision and drainage or other surgical procedures should be performed in conjunction
with antibiotic therapy when indicated.
Prescribing minocycline hydrochloride tablets in the absence of a proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug resistant bacteria.
Information For Patients
Diarrhea is a common problem caused by antibiotics which usually ends when the antibiotic is discontinued. Sometimes after starting treatment with antibiotics, patients can develop watery and bloody stools (with or without stomach cramps and fever) even as late as two or more months after having taken the last dose of the antibiotic. If this occurs, patients should contact their physician as soon as possible.
Photosensitivity manifested by an exaggerated sunburn reaction has been observed in some individuals taking tetracyclines. Patients apt to be exposed to direct sunlight or ultraviolet light should be advised that this reaction can occur with tetracycline drugs, and treatment should be discontinued at the first evidence of skin erythema. This reaction has been reported with use of minocycline.
Patients who experience central nervous system symptoms should be cautioned about driving vehicles or using hazardous machinery while on minocycline therapy (see WARNINGS).
Concurrent use of tetracycline with oral contraceptives may render oral contraceptives less effective (see Drug Interactions).
Patients should be counseled that antibacterial drugs including minocycline hydrochloride tablets should only be used to treat bacterial infections. They do not treat viral infections (e.g., the common cold). When minocycline hydrochloride tablets are prescribed to treat a bacterial infection, patients should be told that although it is common to feel better early in the course of therapy, the medication should be taken exactly as directed. Skipping doses or not completing the full course of therapy may: (1) decrease the effectiveness of the immediate treatment and (2) increase the likelihood that bacteria will develop resistance and will not be treatable by minocycline hydrochloride tablets or other antibacterial drugs in the future.
Unused supplies of tetracycline antibiotics should be discarded by the expiration date.
Laboratory Tests
In venereal disease when coexistent syphilis is suspected, a dark-field examination should be done before treatment is started and the blood serology repeated monthly for at least four months.
Periodic laboratory evaluations of organ systems, including hematopoietic, renal, and hepatic, should be performed.
Drug Interactions
Because tetracyclines have been shown to depress plasma prothrombin activity, patients who are on anticoagulant therapy may require downward adjustment of their anticoagulant dosage.
Since bacteriostatic drugs may interfere with the bactericidal action of penicillin, it is advisable to avoid giving tetracycline-class drugs in conjunction with penicillin.
Absorption of tetracyclines is impaired by antacids containing aluminum, calcium, or magnesium, and iron-containing preparations.
The concurrent use of tetracycline and methoxyflurane has been reported to result in fatal renal toxicity.
Concurrent use of tetracyclines with oral contraceptives may render oral contraceptives less effective.
Administration of isotretinoin should be avoided shortly before, during, and shortly after minocycline therapy. Each drug alone has been associated with pseudotumor cerebri (see PRECAUTIONS).
Increased risk of ergotism when ergot alkaloids or their derivatives are given with tetracyclines.
Drug/Laboratory Test Interactions
False elevations of urinary catecholamine levels may occur due to interference with the fluorescence test.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Dietary administration of minocycline in long term tumorigenicity studies in rats resulted in evidence of thyroid tumor production. Minocycline has also been found to produce thyroid hyperplasia in rats and dogs. In addition, there has been evidence of oncogenic activity in rats in studies with a related antibiotic, oxytetracycline (i.e., adrenal and pituitary tumors). Likewise, although mutagenicity studies of minocycline have not been conducted, positive results in in vitro mammalian cell assays (i.e., mouse lymphoma and Chinese hamster lung cells) have been reported for related antibiotics (tetracycline hydrochloride and oxytetracycline). Segment I (fertility and general reproduction) studies have provided evidence that minocycline impairs fertility in male rats.
Pregnancy
Teratogenic Effects:Pregnancy Category D (see WARNINGS).
All pregnancies have a background risk of birth defects, loss, or other adverse outcome regardless of drug exposure. There are no adequate and well-controlled studies on the use of minocycline in pregnant women. Minocycline, like other tetracycline-class antibiotics, crosses the placenta and may cause fetal harm when administered to a pregnant woman. Rare spontaneous reports of congenital anomalies including limb reduction have been reported in post-marketing experience. Only limited information is available regarding these reports; therefore, no conclusion on causal association can be established. If minocycline is used during pregnancy or if the patient becomes pregnant while taking this drug, the patient should be apprised of the potential hazard to the fetus.
Nonteratogenic Effects: (see WARNINGS).
Nursing Mothers
Tetracyclines are excreted in human milk. Because of the potential for serious adverse reactions in nursing infants from the tetracyclines, a decision should be made whether to discontinue nursing or discontinue the drug, taking into account the importance of the drug to the mother (see WARNINGS).
Pediatric Use
Minocycline is not recommended for use in children below 8 years of age unless the expected benefits of therapy outweigh the risks (see WARNINGS).
Geriatric Use
Clinical studies of oral minocycline did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy (see WARNINGS, DOSAGE AND ADMINISTRATION).
-
ADVERSE REACTIONS
Due to oral minocycline’s virtually complete absorption, side effects to the lower bowel, particularly diarrhea, have been infrequent. The following adverse reactions have been observed in patients receiving tetracyclines.
Body as a whole: Fever, and discoloration of secretions.
Gastrointestinal: Anorexia, nausea, vomiting, diarrhea, dyspepsia, stomatitis, glossitis, dysphagia, enamel hypoplasia, enterocolitis, pseudomembranous colitis, pancreatitis, inflammatory lesions (with monilial overgrowth) in the oral and anogenital regions. Instances of esophagitis and esophageal ulcerations have been reported in patients taking the tetracycline-class antibiotics in capsule and tablet form. Most of these patients took the medication immediately before going to bed (see DOSAGE AND ADMINISTRATION).
Genitourinary: Vulvovaginitis.
Hepatic toxicity: Hyperbilirubinemia, hepatic cholestasis, increases in liver enzymes, fatal hepatic failure, and jaundice. Hepatitis, including autoimmune hepatitis, and liver failure have been reported (see PRECAUTIONS).
Skin: Alopecia, erythema nodosum, hyperpigmentation of nails, pruritus, toxic epidermal necrolysis, and vasculitis. Maculopapular and erythematous rashes. Exfoliative dermatitis has been reported. Fixed drug eruptions have been reported. Lesions occurring on the glans penis have caused balanitis. Erythema multiforme and Stevens-Johnson syndrome have been reported. Photosensitivity is discussed above (see WARNINGS). Pigmentation of the skin and mucous membranes has been reported.
Respiratory: Cough, dyspnea, bronchospasm, exacerbation of asthma, and pneumonitis.
Renal toxicity: Interstitial nephritis. Elevations in BUN have been reported and are apparently dose related (see WARNINGS). Reversible acute renal failure has been reported.
Musculoskeletal: Arthralgia, arthritis, bone discoloration, myalgia, joint stiffness, and joint swelling.
Hypersensitivity reactions: Urticaria, angioneurotic edema, polyarthralgia, anaphylaxis/ anaphylactoid reaction (including shock and fatalities) anaphylactoid purpura, myocarditis, pericarditis, exacerbation of systemic lupus erythematosus and pulmonary infiltrates with eosinophilia have been reported. A transient lupus-like syndrome and serum sickness-like reactions have also been reported.
Blood: Agranulocytosis, hemolytic anemia, thrombocytopenia, leukopenia, neutropenia, pancytopenia, and eosinophilia have been reported.
Central Nervous System: Convulsions, dizziness, hypesthesia, paresthesia, sedation, and vertigo. Bulging fontanels in infants and benign intracranial hypertension (pseudotumor cerebri) in adults have been reported (see PRECAUTIONS-General). Headache has also been reported.
Other: Thyroid cancer has been reported in the post-marketing setting in association with minocycline products. When minocycline therapy is given over prolonged periods, monitoring for signs of thyroid cancer should be considered. When given over prolonged periods, tetracyclines have been reported to produce brown-black microscopic discoloration of the thyroid gland. Cases of abnormal thyroid function have been reported.
Tooth discoloration in children less than 8 years of age, and also in adults has been reported (see WARNINGS).
Oral cavity discoloration (including tongue, lip, and gum) have been reported.
Tinnitus and decreased hearing have been reported in patients on minocycline hydrochloride.
The following syndromes have been reported. In some cases involving these syndromes, death has been reported. As with other serious adverse reactions, if any of these syndromes are recognized, the drug should be discontinued immediately:
Hypersensitivity syndrome consisting of cutaneous reaction (such as rash or exfoliative dermatitis), eosinophilia, and one or more of the following: hepatitis, pneumonitis, nephritis, myocarditis, and pericarditis. Fever and lymphadenopathy may be present.
Lupus-like syndrome consisting of positive antinuclear antibody; arthralgia, arthritis, joint stiffness, or joint swelling; and one or more of the following: fever, myalgia, hepatitis, rash, and vasculitis.
Serum sickness-like syndrome consisting of fever; urticaria or rash; and arthralgia, arthritis, joint stiffness, or joint swelling. Eosinophilia may be present.
-
OVERDOSAGE
The adverse events more commonly seen in overdose are dizziness, nausea, and vomiting.
No specific antidote for minocycline is known.
In case of overdosage, discontinue medication, treat symptomatically and institute supportive measures. Minocycline is not removed in significant quantities by hemodialysis or peritoneal dialysis.
-
DOSAGE AND ADMINISTRATION
THE USUAL DOSAGE AND FREQUENCY OF ADMINISTRATION OF MINOCYCLINE DIFFERS FROM THAT OF THE OTHER TETRACYCLINES. EXCEEDING THE RECOMMENDED DOSAGE MAY RESULT IN AN INCREASED INCIDENCE OF SIDE EFFECTS.
Minocycline hydrochloride tablets may be taken with or without food (see CLINICAL PHARMACOLOGY).
Ingestion of adequate amounts of fluids along with capsule and tablet forms of drugs in the tetracycline-class is recommended to reduce the risk of esophageal irritation and ulceration. The tablets should be swallowed whole.
For Pediatric Patients Above 8 Years of Age
Usual pediatric dose: 4 mg/kg initially followed by 2 mg/kg every 12 hours, not to exceed the usual adult dose.
Adults
The usual dosage of minocycline hydrochloride tablets is 200 mg initially followed by 100 mg every 12 hours. Alternatively, if more frequent doses are preferred, two or four 50 mg tablets may be given initially followed by one 50 mg tablet four times daily.
Uncomplicated gonococcal infections other than urethritis and anorectal infections in men: 200 mg initially, followed by 100 mg every 12 hours for a minimum of four days, with post-therapy cultures within 2 to 3 days.
In the treatment of uncomplicated gonococcal urethritis in men, 100 mg every 12 hours for 5 days is recommended.
For the treatment of syphilis, the usual dosage of minocycline hydrochloride should be administered over a period of 10 to 15 days. Close follow-up, including laboratory tests, is recommended.
In the treatment of meningococcal carrier state, the recommended dosage is 100 mg every 12 hours for five days.
Mycobacterium marinum infections: Although optimal doses have not been established, 100 mg every 12 hours for 6 to 8 weeks have been used successfully in a limited number of cases.
Uncomplicated urethral, endocervical, or rectal infection in adults caused by Chlamydia trachomatis or Ureaplasma urealyticum: 100 mg orally, every 12 hours for at least seven days.
Ingestion of adequate amounts of fluids along with capsule and tablet forms of drugs in the tetracycline-class is recommended to reduce the risk of esophageal irritation and ulceration.
The pharmacokinetics of minocycline in patients with renal impairment (CLCR <80mL/min) have not been fully characterized. Current data are insufficient to determine if a dosage adjustment is warranted. The total daily dosage should not exceed 200 mg in 24 hours. However, due to the anti-anabolic effect of tetracyclines, BUN and creatinine should be monitored (see WARNINGS).
-
HOW SUPPLIED
Minocycline hydrochloride tablets are supplied as aqueous film coated tablets containing minocycline hydrochloride equivalent to 50 mg, 75 mg and 100 mg minocycline.
The 50 mg tablets are white, unscored, modified capsule shaped, coated tablet debossed “Par” on one side and “511" on the other. Each tablet contains minocycline hydrochloride equivalent to 50 mg minocycline, supplied as follows:
NDC 49884-511-01 Bottle of 100
The 75 mg tablets are white, unscored, modified capsule shaped, coated tablet debossed “Par" on one side and “512" on the other. Each tablet contains minocycline hydrochloride equivalent to 75 mg minocycline, supplied as follows:
NDC 49884-512-01 Bottle of 100
The 100 mg tablets are white, unscored, modified capsule shaped, coated tablet debossed “Par" on one side and “513" on the other. Each tablet contains minocycline hydrochloride equivalent to 100 mg minocycline, supplied as follows:
NDC 49884-513-03 Bottle of 50
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature].
Protect from light, moisture and excessive heat.
Dispense in a tight, light-resistant container as defined in the USP.
-
ANIMAL PHARMACOLOGY AND/OR TOXICOLOGY
Minocycline hydrochloride has been observed to cause a dark discoloration of the thyroid in experimental animals (rats, minipigs, dogs and monkeys). In the rat, chronic treatment with minocycline hydrochloride has resulted in goiter accompanied by elevated radioactive iodine uptake and evidence of thyroid tumor production. Minocycline hydrochloride has also been found to produce thyroid hyperplasia in rats and dogs.
-
REFERENCES
- National Committee for Clinical Laboratory Standards, Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria that Grow Aerobically - Fourth Edition; Approved Standard. NCCLS Document M7-A4, Vol . 17, No. 2, NCCLS, 940 West Valley Road, Suite 1400, Wayne, PA. January 1997.
- National Committee for Clinical Laboratory Standards, Performance Standards for Antimicrobial Disk Susceptibility Tests - Sixth Edition; Approved Standard. NCCLS Document M2-A6, Vol. 17, No. 1, NCCLS, 940 West Valley Road, Suite 1400, Wayne, PA. January 1997.
- National Committee for Clinical Laboratory Standards, Performance Standards for Antimicrobial Susceptibility Tests - Eighth Edition; Approved Standard. NCCLS Document M100-S8, Vol. 18, No. 1, NCCLS, 940 West Valley Road, Suite 1400, Wayne, PA. January 1998.
-
INFORMATION FOR PATIENTS
Minocycline Hydrochloride Tablets, USP
50, 75, and 100 mg
Read the Patient Information that comes with Minocyline HydrochlorideTablets before you or a family member starts taking it and each time you get a refill. There may be new information. This leaflet does not take the place of talking to your doctor about your medical condition or treatment.
What is Minocycline Hydrochloride?
Minocycline Hydrochloride is a tetracycline-class antibiotic medicine. Minocycline Hydrochloride is used to treat certain infections caused by bacteria. These include infections of the skin, respiratory tract, urinary tract, some sexually transmitted diseases, and others. Minocycline Hydrochloride may be used along with other treatments for severe acne.
Sometimes, other germs, called viruses cause infections. The common cold is a virus. Minocycline Hydrochloride, like other antibiotics, does not treat viruses.
Who should not use Minocycline Hydrochloride?
Do not take Minocycline Hydrochloride if you are allergic to minocycline or other tetracycline antibiotics.
Ask your doctor or pharmacist for a list of these medications if you are not sure. See the end of this leaflet for a complete list of ingredients in Minocycline Hydrochloride.
Minocycline Hydrochloride is not recommended for pregnant women or children up to 8 years old because:
- Minocycline Hydrochloride may harm an unborn baby.
- Minocycline Hydrochloride may permanently turn a baby’s or child’s teeth yellow-gray-brown during tooth development. Tooth development happens in the last half of pregnancy and birth to age 8 years.
What should I tell my doctor before starting Minocycline HydrochlorideTablets?
Tell your doctor about all of your medical conditions, including if you:
- have liver or kidney problems.
- are pregnant or planning to become pregnant. Minocycline Hydrochloride may harm your unborn baby. Stop taking Minocycline Hydrochloride and call your doctor if you become pregnant while taking it.
- are breast feeding. Minocycline Hydrochloride passes into your milk and may harm your baby. You should decide whether to use Minocycline Hydrochloride or breastfeed, but not both.
Tell your doctor about all the medicines you are taking including prescription and non- prescription medications, vitamins, and herbal supplements. Minocycline Hydrochloride and other medicines may interact. Especially tell your doctor if you take:
- birth control pills. Minocycline Hydrochloride may make your birth control pills less effective.
- a blood thinner medicine. The dose of your blood thinner may have to be lowered.
- a penicillin antibiotic medicine. Minocycline Hydrochloride and penicillins should not be used together.
- Migraine medicines called ergot alkaloids.
- An acne medicine called isotretinoin (Accutane®, Amnesteem®, Claravis®, Sotret®).
- Antacids that contain aluminum, calcium, or magnesium, or iron-containing products.
Know the medicines you take, keep a list of them to show your doctor and pharmacist each time you get a new medicine.
How should I take Minocycline HydrochlorideTablets?
-
Take Minocycline Hydrochloride Tablets exactly as your doctor tells you to take them. Skipping doses or not taking all your Minocycline Hydrochloride may:
- Decrease the effectiveness of the treatment.
- Increase the chance that bacteria will develop resistance to Minocycline Hydrochloride.
- Take Minocycline Hydrochloride with a full glass of liquid. Taking Minocycline Hydrochloride with enough liquid may lower your chance of getting irritation or ulcers in your esophagus. Your esophagus is the tube that connects your mouth to your stomach.
- Minocycline Hydrochloride Tablets may be taken with or without food. If you forget to take Minocycline Hydrochloride, take it as soon as you remember.
- If you take too much Minocycline Hydrochloride, call you doctor or poison control center right away.
What are the possible side effects of Minocycline Hydrochloride?
Minocycline Hydrochloridemay cause serious side effects. Stop Minocycline Hydrochlorideand call your doctor if you have:
- watery diarrhea
- bloody stools
- stomach cramps
- unusual headaches
- blurred vision
- fever
- rash
- joint pain
- feeling very tired
Minocycline Hydrochloridemay also cause:
- central nervous system effects. Symptoms include light-headedness, dizziness, and a spinning feeling (vertigo). You should not drive or operate machines if you have these symptoms.
- sun sensitivity (photosensitivity). You may get a worse sunburn with Minocycline Hydrochloride. Avoid sun exposure and the use of sunlamps or tanning beds. Protect your skin while out in the sunlight. Stop Minocycline Hydrochloride and call your doctor if your skin turns red.
These are not all the side effects with Minocycline Hydrochloride. Ask your doctor or pharmacist for more information.
How should I store Minocycline HydrochlorideTablets?
- Store Minocycline Hydrochloride Tablets at room temperature and away from excess heat and moisture.
- Throw away any Minocycline Hydrochloride that is outdated or no longer needed.
- Keep Minocycline Hydrochloride Tablets and all medicines out of the reach of children.
General advice about Minocycline HydrochlorideTablets
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use Minocycline HydrochlorideTablets for a condition for which it was not prescribed. Do not give Minocycline HydrochlorideTablets to other people, even if they have the same symptoms you have. It may harm them.
This Patient Information leaflet summarizes the most important information about Minocycline Hydrochloride. If you would like more information, talk with your doctor.
Your doctor or pharmacist can give you information about Minocycline Hydrochloridethat is written for health care professionals. For more information, you can also call Par Pharmaceutical at 1-800-828-9393.
What are the ingredients in Minocycline HydrochlorideTablets?
Active ingredient: minocycline hydrochloride, 50 mg, 75 mg, and 100 mg
Inactive ingredients: Microcrystalline Cellulose NF, Lactose Anhydrous NF, Povidone USP, Colloidal Silicon Dioxide NF, Magnesium Stearate NF, and Sodium Starch Glycolate NF. The 50 mg, 75 mg and 100 mg tablets also contain Opadry White which contains: Titanium Dioxide USP, Hypromellose Type 2910 USP, Polyethylene Glycol 400 NF, and Polysorbate 80 NF.
All other trademarks are the property of their respective owners.
Manufactured and Distributed by:
PAR PHARMACEUTICAL
Chestnut Ridge, NY 10977
Revised: 12/16 OS511-01-1-08
- PRINCIPAL DISPLAY PANEL 50 MG TABLETS
- PRINCIPAL DISPLAY PANEL 75 MG TABLETS
- PRINCIPAL DISPLAY PANEL 100 MG TABLETS
-
INGREDIENTS AND APPEARANCE
MINOCYCLINE
minocycline tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:49884-511 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MINOCYCLINE HYDROCHLORIDE (UNII: 0020414E5U) (MINOCYCLINE - UNII:FYY3R43WGO) MINOCYCLINE 50 mg Inactive Ingredients Ingredient Name Strength CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) MAGNESIUM STEARATE (UNII: 70097M6I30) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) POLYSORBATE 80 (UNII: 6OZP39ZG8H) POVIDONE K30 (UNII: U725QWY32X) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) Product Characteristics Color white Score no score Shape CAPSULE Size 10mm Flavor Imprint Code Par;511 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:49884-511-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 04/16/2003 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA065131 04/16/2003 MINOCYCLINE
minocycline tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:49884-512 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MINOCYCLINE HYDROCHLORIDE (UNII: 0020414E5U) (MINOCYCLINE - UNII:FYY3R43WGO) MINOCYCLINE 75 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) MAGNESIUM STEARATE (UNII: 70097M6I30) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) POLYSORBATE 80 (UNII: 6OZP39ZG8H) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) POVIDONE K30 (UNII: U725QWY32X) Product Characteristics Color white Score no score Shape CAPSULE Size 11mm Flavor Imprint Code Par;512 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:49884-512-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 04/16/2003 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA065131 04/16/2003 MINOCYCLINE
minocycline tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:49884-513 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MINOCYCLINE HYDROCHLORIDE (UNII: 0020414E5U) (MINOCYCLINE - UNII:FYY3R43WGO) MINOCYCLINE 100 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) MAGNESIUM STEARATE (UNII: 70097M6I30) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) POLYSORBATE 80 (UNII: 6OZP39ZG8H) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) POVIDONE K30 (UNII: U725QWY32X) Product Characteristics Color white Score no score Shape CAPSULE Size 13mm Flavor Imprint Code Par;513 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:49884-513-03 50 in 1 BOTTLE; Type 0: Not a Combination Product 04/16/2003 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA065131 04/16/2003 Labeler - Par Pharmaceutical, Inc. (092733690) Registrant - Par Pharmaceutical Inc. (092733690)






