Label: DONG SUNG MONELYN- zinc pyrithione suspension, panthenol, niacinamide, biotin shampoo
-
Contains inactivated NDC Code(s)
NDC Code(s): 70778-0002-1 - Packager: Daolcosmetic Co.,ltd
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated June 14, 2016
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
-
INACTIVE INGREDIENT
Sodium Polyoxyethylene Laurylether Sulfate, Ammonium Lauryl Sulfate Solution Cocamidopropylbetaine, Cocamide DEA, Dimethicone, Polyquaternium-7, Carbomers, Jojoba Oil, Disodium Edetate Hydrate, Guar Hydroxypropyltrimonium Chloride Sage Extract, Rosemary Extract, Lily Extract, Chamomile Extract, Saffron Extract,Ginseng Extract, Green Tea Extract, Sophora Extract, Paeonia Extract, Loquat Leaf Extract, Water
- PURPOSE
- KEEP OUT OF REACH OF CHILDREN
- INDICATIONS & USAGE
-
WARNINGS
For external use only
Do not use when Your scalp is red, inflamed, irritated or painful
When using this product
Do not apply on other parts of the body
Avoid contact with eyes . If contact occurs, rinse eyes thoroughly with water.
Do not apply directly to wound or open cut.
Stop use and ask doctor if rash or irritation on scalp develops and lasts .
Store at room temperature - DOSAGE & ADMINISTRATION
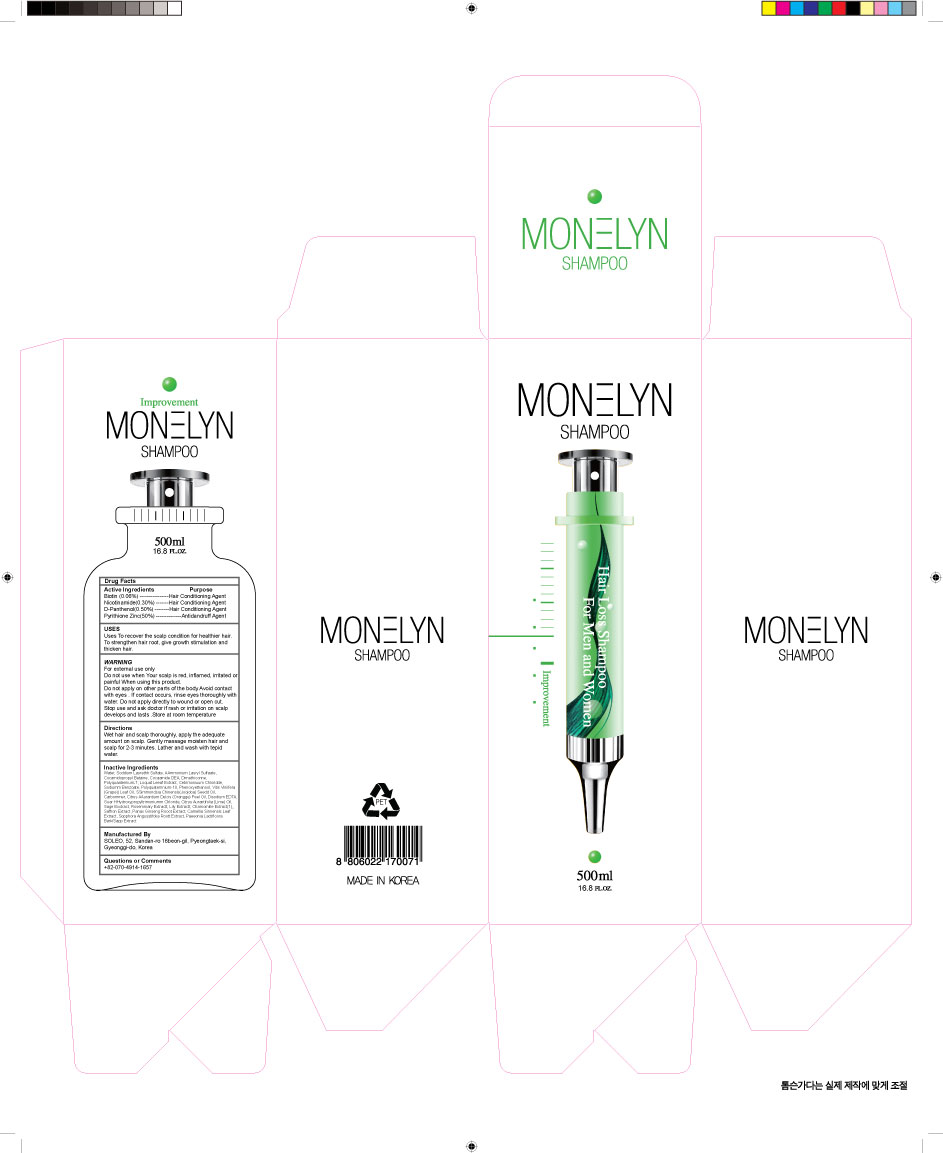
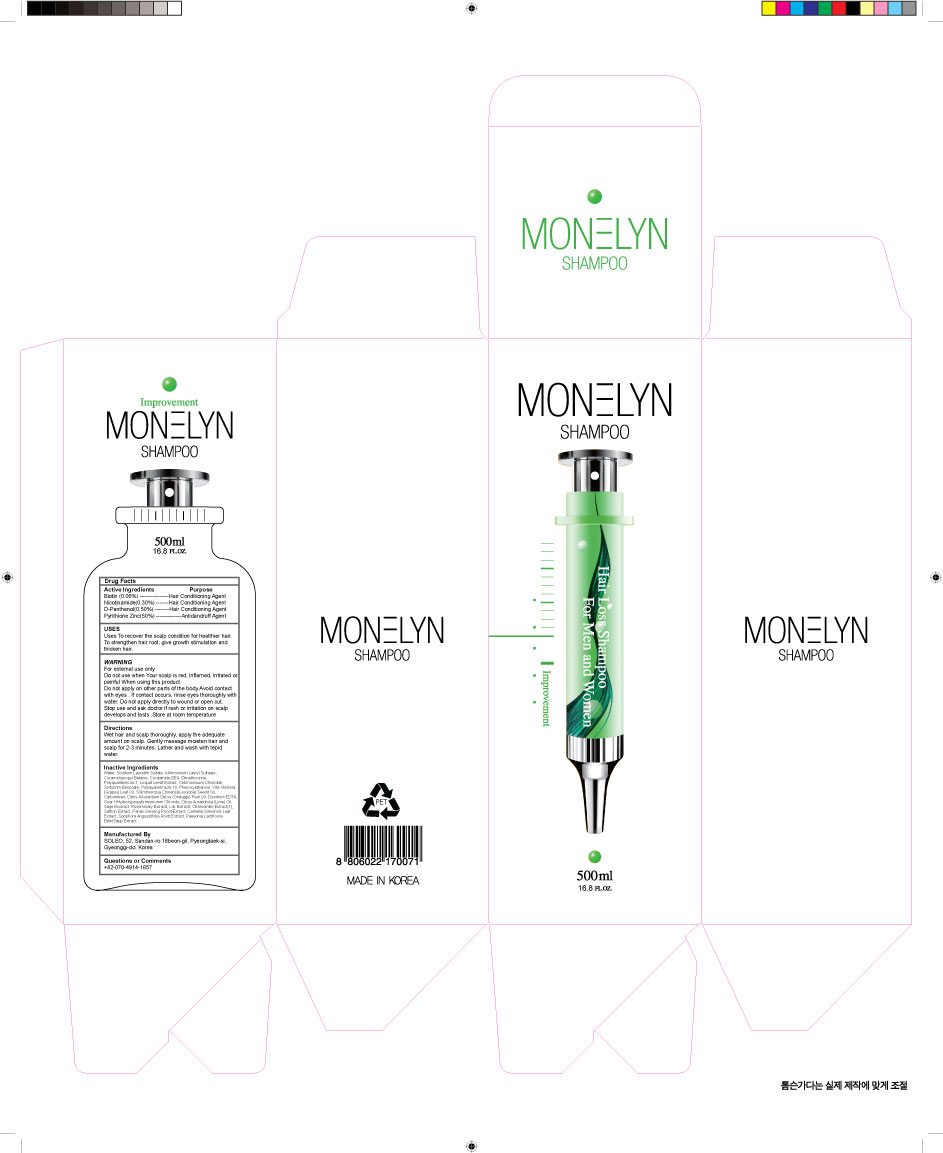
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
DONG SUNG MONELYN
zinc pyrithione suspension, panthenol, niacinamide, biotin shampooProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:70778-0002 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PANTHENOL (UNII: WV9CM0O67Z) (PANTHENOL - UNII:WV9CM0O67Z) PANTHENOL 0.5 g in 100 mL PYRITHIONE ZINC (UNII: R953O2RHZ5) (PYRITHIONE ZINC - UNII:R953O2RHZ5) PYRITHIONE ZINC 2.1 g in 100 mL NIACINAMIDE (UNII: 25X51I8RD4) (NIACINAMIDE - UNII:25X51I8RD4) NIACINAMIDE 0.3 g in 100 mL BIOTIN (UNII: 6SO6U10H04) (BIOTIN - UNII:6SO6U10H04) BIOTIN 0.06 g in 100 mL Inactive Ingredients Ingredient Name Strength SODIUM LAURETH SULFATE (UNII: BPV390UAP0) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70778-0002-1 500 mL in 1 BOTTLE, PUMP; Type 0: Not a Combination Product 03/30/2016 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 03/30/2016 Labeler - Daolcosmetic Co.,ltd (688279560) Registrant - Daolcosmetic Co.,ltd (688279560) Establishment Name Address ID/FEI Business Operations Daolcosmetic Co.,ltd 688279560 manufacture(70778-0002)