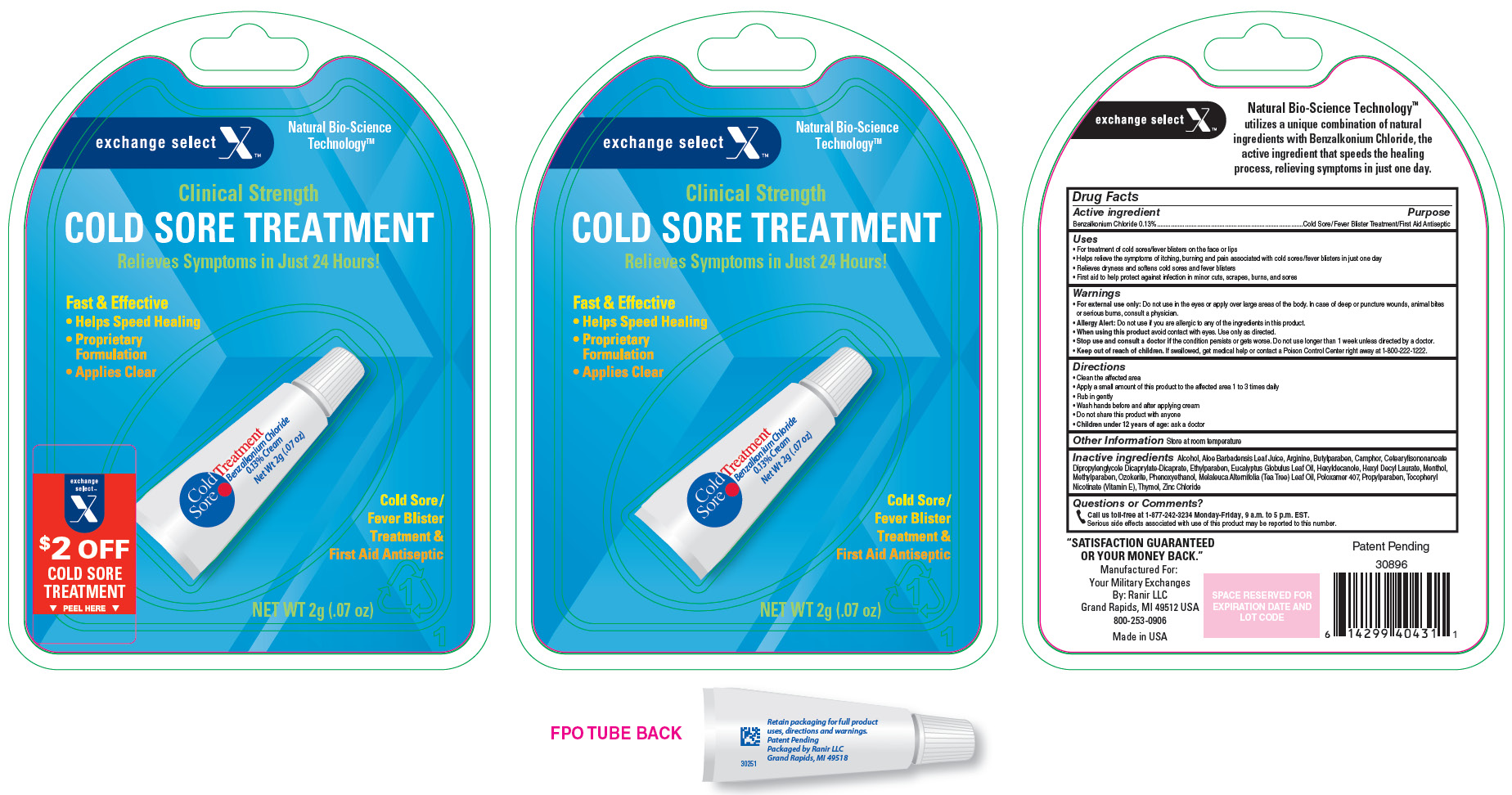
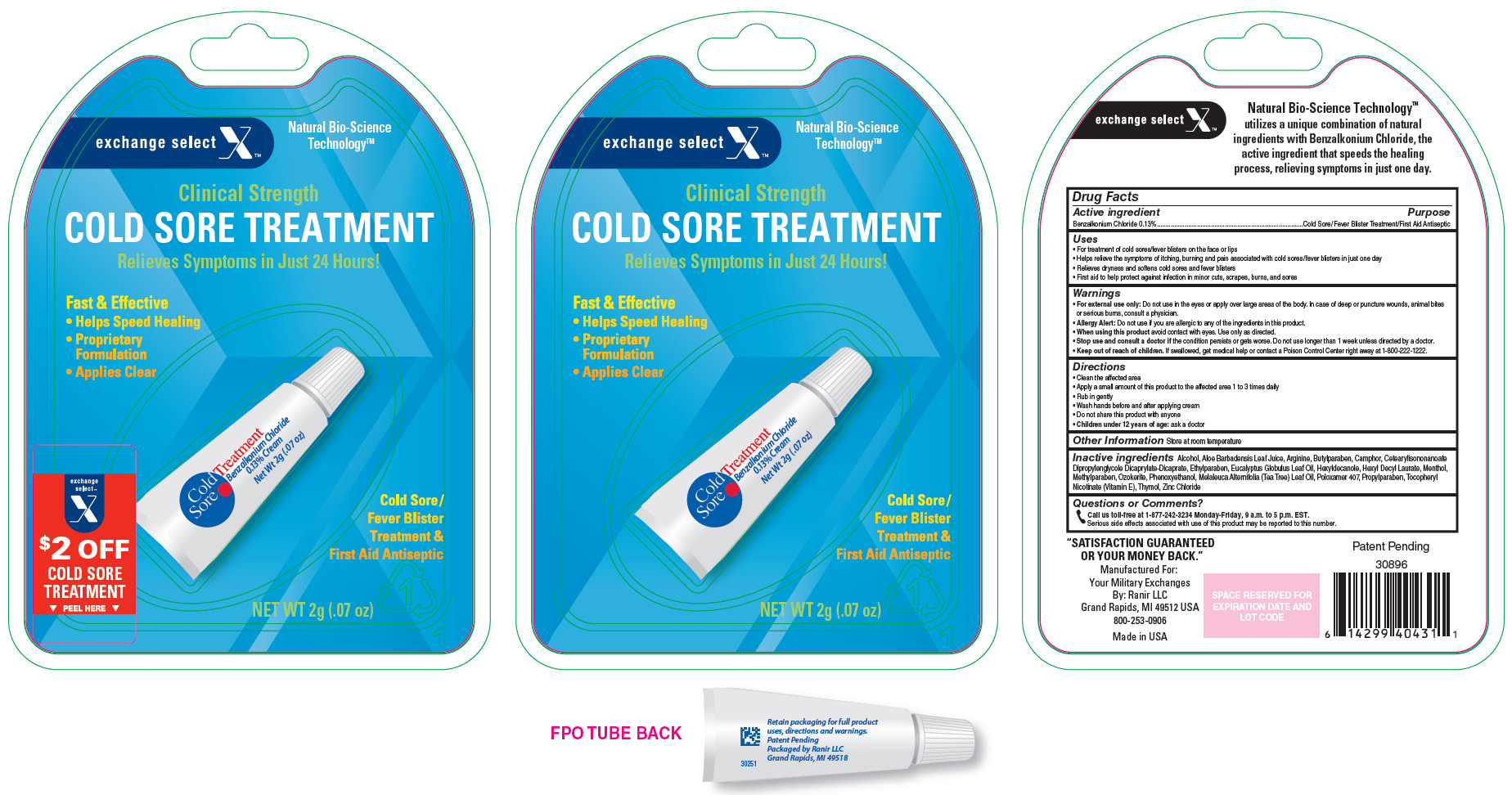
Label: EXCHANGE SELECT COLD SORE TREATMENT- benzalkonium chloride cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 55301-406-01 - Packager: Army + Air Force Exchange Service
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated August 9, 2011
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
- Purpose
-
Uses
- For treatment of cold sores/fever blisters on the face or lips
- Helps relieve the symptoms of itching, burning and pain associated with cold sores/fever blisters in just one day
- Relieves dryness and softens cold sores and fever blisters
- First aid to help protect against infection in minor cuts, scrapes, burns, and sores
-
Warnings
- For external use only: Do not use in the eyes or apply over large areas of the body. In case of deep or puncture wounds, animal bites or serious burns, consult a physician.
- Allergy Alert: Do not use if you are allergic to any of the ingredients in this product.
- When using this product avoid contact with eyes. Use only as directed.
- Stop use and consult a doctor if the condition persists or gets worse. Do not use longer than 1 week unless directed by a doctor.
-
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away. (1-800-222-1222)
- Directions
- Other Information
-
Inactive Ingredients
Alcohol, Aloe Barbadensis Leaf Juice, Arginine, Butylparaben, Camphor, Cetearylisononanoate, Dipropylenglycole Dicaprylate-Dicaprate, Ethylparaben, Eucalyptus Globulus Leaf Oil, Hexyldecanole, Hexyl Decyl Laurate, Menthol,
Methylparaben, Ozokerite, Phenoxyethanol, Melaleuca Alternifolia (Tea Tree) Leaf Oil, Poloxamer 407, Propylparaben, Tocopheryl Nicotinate (Vitamin E), Thymol, Zinc Chloride - Questions or Comments?
- PRINCIPAL DISPLAY PANEL
- KEEP OUT OF REACH OF CHILDREN
-
INGREDIENTS AND APPEARANCE
EXCHANGE SELECT COLD SORE TREATMENT
benzalkonium chloride creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:55301-406 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Benzalkonium Chloride (UNII: F5UM2KM3W7) (Benzalkonium - UNII:7N6JUD5X6Y) Benzalkonium Chloride 1.3 mg in 1 g Inactive Ingredients Ingredient Name Strength Alcohol (UNII: 3K9958V90M) Aloe Vera Leaf (UNII: ZY81Z83H0X) Arginine (UNII: 94ZLA3W45F) Butylparaben (UNII: 3QPI1U3FV8) Camphor (Synthetic) (UNII: 5TJD82A1ET) Cetearyl Isononanoate (UNII: P5O01U99NI) Dipropylene Glycol Caprate/Caprylate Diester (UNII: R6G12EY23X) Ethylparaben (UNII: 14255EXE39) Corymbia Citriodora Leaf Oil (UNII: M63U6N96EB) Hexyldecanol (UNII: 151Z7P1317) Hexyldecyl Laurate (UNII: 0V595C1P6M) Levomenthol (UNII: BZ1R15MTK7) Methylparaben (UNII: A2I8C7HI9T) Ceresin (UNII: Q1LS2UJO3A) Tea Tree Oil (UNII: VIF565UC2G) Poloxamer 407 (UNII: TUF2IVW3M2) Propylparaben (UNII: Z8IX2SC1OH) Alpha-Tocopherol (UNII: H4N855PNZ1) Thymol (UNII: 3J50XA376E) Zinc Chloride (UNII: 86Q357L16B) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:55301-406-01 1 in 1 PACKAGE 1 2 g in 1 TUBE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333A 08/04/2011 Labeler - Army + Air Force Exchange Service (001695568) Registrant - Ranir LLC (364567615) Establishment Name Address ID/FEI Business Operations Ranir LLC 364567615 label