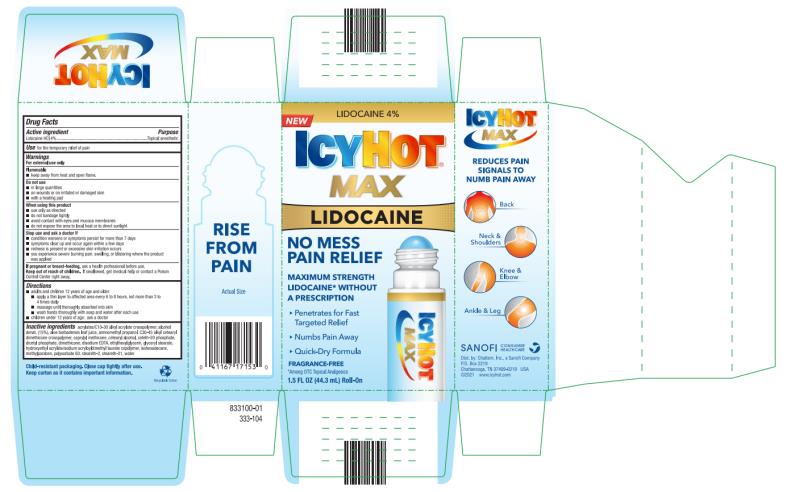
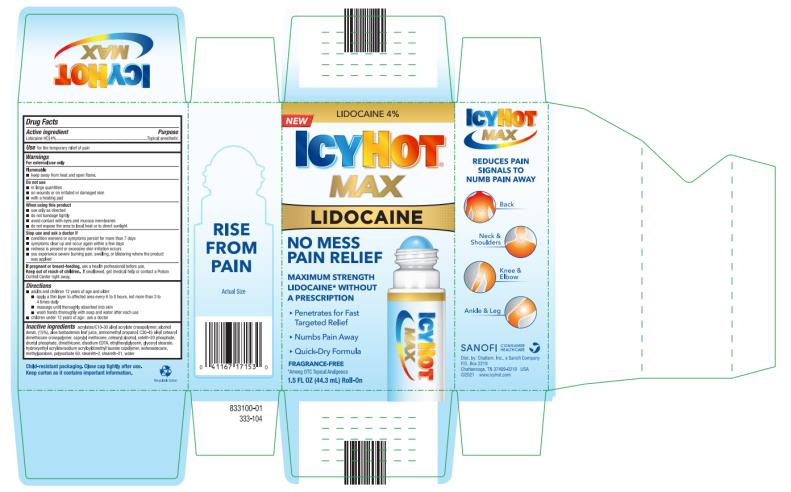
Label: ICY HOT LIDOCAINE NO-MESS- lidocaine hydrochloride liquid
- NDC Code(s): 41167-1711-0
- Packager: Chattem, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated November 1, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredient
- Purpose
- Use
-
Warnings
For external use only
Flammable
■ keep away from heat and open flame.
When using this product
■ use only as directed
■ do not bandage tightly
■ avoid contact with eyes and mucous membranes
■ do not expose the area to local heat or to direct sunlight
- Directions
-
Inactive ingredients
acrylates/C10-30 alkyl acrylate crosspolymer, alcohol denat. (15%), aloe barbadensis leaf juice, aminomethyl propanol, C30-45 alkyl cetearyl dimethicone crosspolymer, caprylyl methicone, cetearyl alcohol, ceteth-20 phosphate, dicetyl phosphate, dimethicone, disodium EDTA, ethylhexylglycerin, glyceryl stearate, hydroxyethyl acrylate/sodium acryloyldimethyl taurate copolymer, isohexadecane, methylparaben, polysorbate 60, steareth-2, steareth-21, water
333-104
Child-resistant packaging. Close cap tightly after use.
Keep carton as it contains important information.
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ICY HOT LIDOCAINE NO-MESS
lidocaine hydrochloride liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:41167-1711 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LIDOCAINE HYDROCHLORIDE (UNII: V13007Z41A) (LIDOCAINE - UNII:98PI200987) LIDOCAINE HYDROCHLORIDE ANHYDROUS 4 g in 100 g Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) ALOE VERA LEAF (UNII: ZY81Z83H0X) AMINOMETHYLPROPANOL (UNII: LU49E6626Q) C30-45 ALKYL CETEARYL DIMETHICONE CROSSPOLYMER (UNII: 4ZK9VP326R) CETETH-20 PHOSPHATE (UNII: 921FTA1500) DIMETHICONE (UNII: 92RU3N3Y1O) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) HYDROXYETHYL ACRYLATE/SODIUM ACRYLOYLDIMETHYL TAURATE COPOLYMER (100000 MPA.S AT 1.5%) (UNII: 86FQE96TZ4) ISOHEXADECANE (UNII: 918X1OUF1E) METHYLPARABEN (UNII: A2I8C7HI9T) POLYSORBATE 60 (UNII: CAL22UVI4M) STEARETH-2 (UNII: V56DFE46J5) STEARETH-21 (UNII: 53J3F32P58) WATER (UNII: 059QF0KO0R) CARBOMER INTERPOLYMER TYPE A (55000 CPS) (UNII: 59TL3WG5CO) EDETATE DISODIUM ANHYDROUS (UNII: 8NLQ36F6MM) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) DIHEXADECYL PHOSPHATE (UNII: 2V6E5WN99N) CAPRYLYL TRISILOXANE (UNII: Q95M2P1KJL) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:41167-1711-0 42.5 g in 1 BOTTLE; Type 0: Not a Combination Product 02/10/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M017 02/10/2022 Labeler - Chattem, Inc. (003336013)