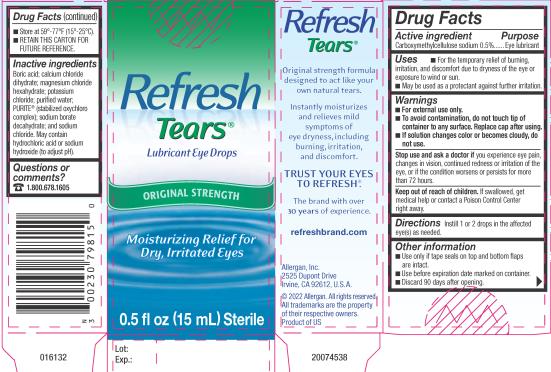
Label: REFRESH TEARS- carboxymethylcellulose sodium solution/ drops
-
NDC Code(s):
0023-0798-01,
0023-0798-02,
0023-0798-03,
0023-0798-04, view more0023-0798-15
- Packager: Allergan, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated June 30, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Uses
-
Warnings
-
For external use only.
-
To avoid contamination, do not touch tip of container to any surface. Replace cap after using.
- If solution changes color or becomes cloudy, do not use.
-
For external use only.
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
REFRESH TEARS
carboxymethylcellulose sodium solution/ dropsProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:0023-0798 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CARBOXYMETHYLCELLULOSE SODIUM (UNII: K679OBS311) (CARBOXYMETHYLCELLULOSE - UNII:05JZI7B19X) CARBOXYMETHYLCELLULOSE SODIUM 5 mg in 1 mL Inactive Ingredients Ingredient Name Strength BORIC ACID (UNII: R57ZHV85D4) CALCIUM CHLORIDE (UNII: M4I0D6VV5M) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) WATER (UNII: 059QF0KO0R) SODIUM CHLORITE (UNII: G538EBV4VF) SODIUM CHLORATE (UNII: T95DR77GMR) CHLORINE DIOXIDE (UNII: 8061YMS4RM) SODIUM BORATE (UNII: 91MBZ8H3QO) SODIUM CHLORIDE (UNII: 451W47IQ8X) HYDROCHLORIC ACID (UNII: QTT17582CB) SODIUM HYDROXIDE (UNII: 55X04QC32I) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0023-0798-04 1 in 1 CARTON 05/16/1997 1 3 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product 2 NDC:0023-0798-03 1 in 1 CARTON 05/16/1997 2 3 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product 3 NDC:0023-0798-15 1 in 1 CARTON 05/16/1997 3 15 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product 4 NDC:0023-0798-01 2 in 1 CARTON 05/16/1997 4 15 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product 5 NDC:0023-0798-02 4 in 1 CARTON 05/16/1997 5 15 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M018 05/16/1997 Labeler - Allergan, Inc. (144796497)