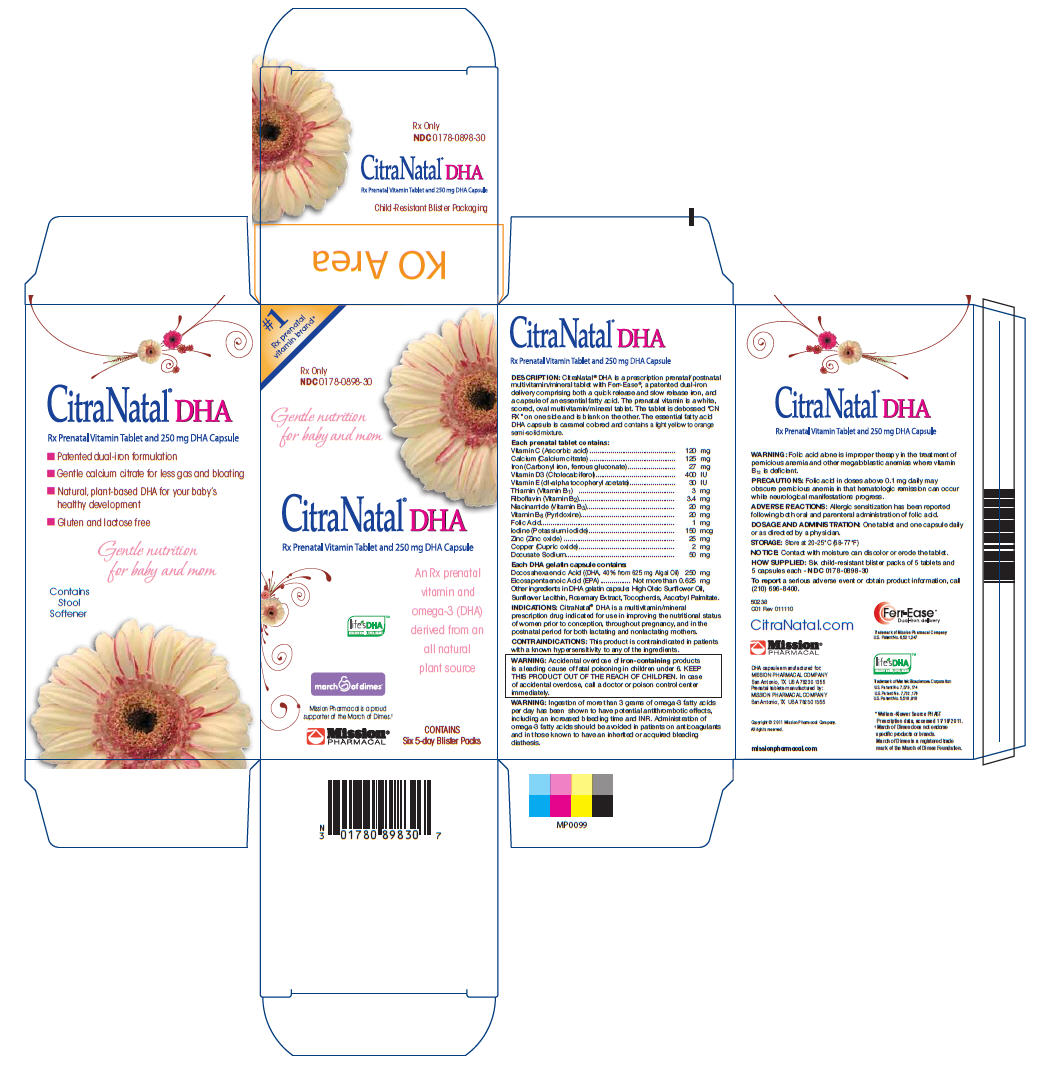
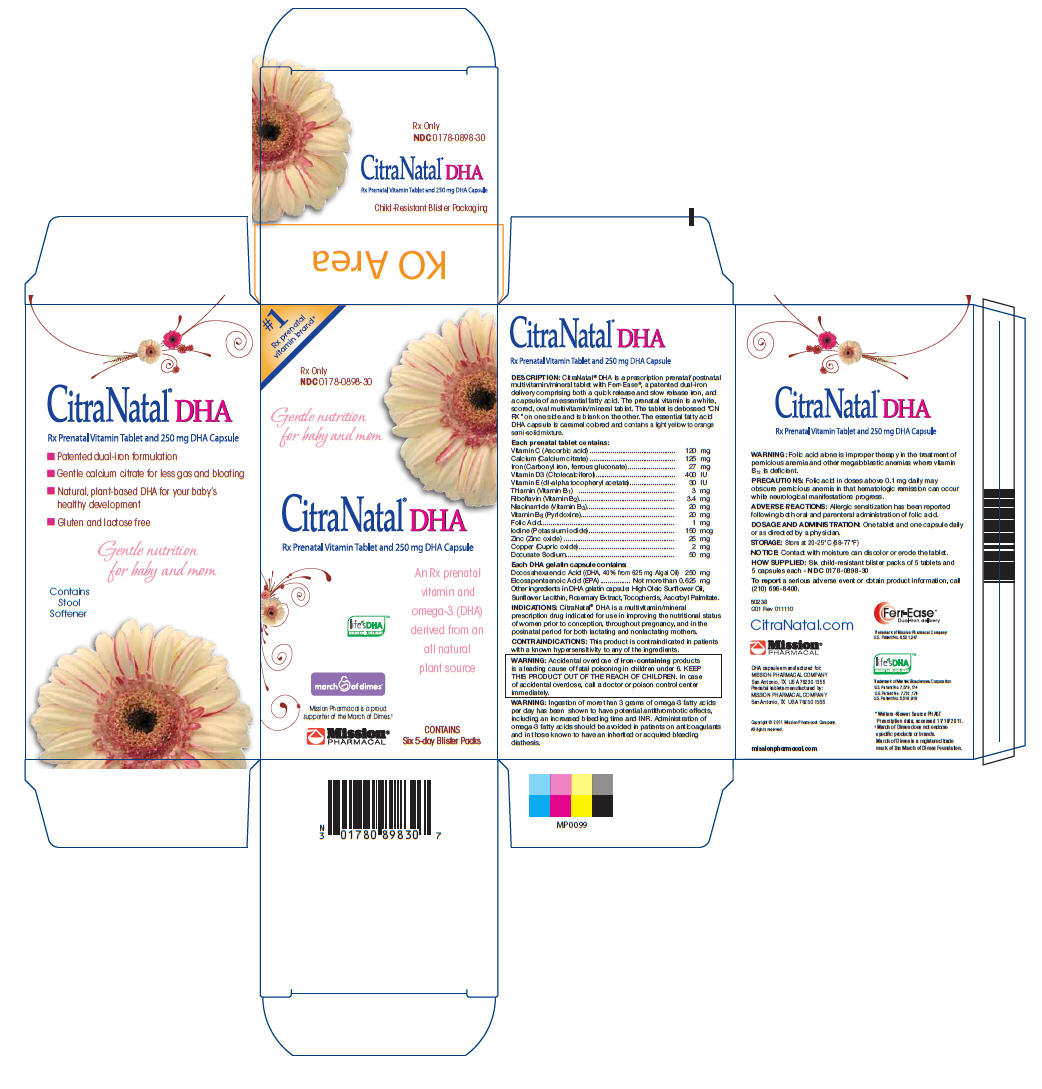
Label: CITRANATAL DHA- vitamin c, calcium, iron, vitamin d3, vitamin e, thiamin, riboflavin, niacinamide, vitamin b6, folic acid, iodine, zinc, copper, docusate sodium tablet
-
Contains inactivated NDC Code(s)
NDC Code(s): 0178-0898-30 - Packager: Mission Pharmacal Company
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated March 25, 2013
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- BOXED WARNING (What is this?)
- KEEP OUT OF REACH OF CHILDREN
-
DESCRIPTION
CitraNatal® DHA is a prescription prenatal/postnatal multivitamin/mineral tablet with Ferr-Ease®, a patented dual-iron delivery comprising both a quick release and slow release iron, and a capsule of an essential fatty acid. The prenatal vitamin is a white, scored, oval multivitamin/mineral tablet. The tablet is debossed “CN RX” on one side and is blank on the other. The essential fatty acid DHA capsule is caramel colored and contains a light yellow to orange semi-solid mixture.
Each prenatal tablet contains: Vitamin C (Ascorbic acid) 120 mg Calcium (Calcium citrate) 125 mg Iron (Carbonyl iron, ferrous gluconate) 27 mg Vitamin D3 (Cholecalciferol) 400 IU Vitamin E (dl-alpha tocopheryl acetate) 30 IU Thiamin (Vitamin B1) 3 mg Riboflavin (Vitamin B2) 3.4 mg Niacinamide (Vitamin B3) 20 mg Vitamin B6 (Pyridoxine) 20 mg Folic Acid 1 mg Iodine (Potassium iodide) 150 mcg Zinc (Zinc oxide) 25 mg Copper (Cupric oxide) 2 mg Docusate Sodium 50 mg Each DHA gelatin capsule contains:
Docosahexaenoic Acid ((DHA, 40% from 625 mg Algal Oil) 250 mg Eicosapentaenoic Acid (EPA)................. Not more than 0.625 mg Other ingredients in DHA gelatin capsule: High Oleic Sunflower Oil, Sunflower Lecithin, Rosemary Extract, Tocopherols, Ascorbyl Palmitate. - ACTIVE INGREDIENT
- PURPOSE
- INACTIVE INGREDIENT
- INDICATIONS
-
WARNING
Ingestion of more than 3 grams of omega-3 fatty acids per day has been shown to have potential antithrombotic effects, including an increased bleeding time and INR. Administration of omega-3 fatty acids should be avoided in patients on anticoagulants and in those known to have an inherited or acquired bleeding diathesis.
WARNING: Folic acid alone is improper therapy in the treatment of pernicious anemia and other megaloblastic anemias where vitamin B12 is deficient.
- PRECAUTIONS
- ADVERSE REACTIONS
- DOSAGE AND ADMINISTRATION
- STORAGE
-
HOW SUPPLIED
Six child-resistant blister packs of 5 tablets and 5 capsules each - NDC 0178-0898-30
To report a serious adverse event or obtain product information, call (210) 696-8400.
DHA capsules manufactured for:
MISSION PHARMACAL COMPANY
San Antonio, TX USA 78230 1355
Prenatal tablets manufactured by:
MISSION PHARMACAL COMPANY
San Antonio, TX USA 78230 1355 - PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
CITRANATAL DHA
vitamin c, calcium, iron, vitamin d3, vitamin e, thiamin, riboflavin, niacinamide, vitamin b6, folic acid, iodine, zinc, copper, docusate sodium tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:0178-0898 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ASCORBIC ACID (UNII: PQ6CK8PD0R) (ASCORBIC ACID - UNII:PQ6CK8PD0R) ASCORBIC ACID 120 mg CALCIUM CITRATE (UNII: MLM29U2X85) (CALCIUM CATION - UNII:2M83C4R6ZB) CALCIUM CITRATE 125 mg IRON (UNII: E1UOL152H7) (IRON - UNII:E1UOL152H7) IRON 27 mg VITAMIN D (UNII: 9VU1KI44GP) (CHOLECALCIFEROL - UNII:1C6V77QF41) VITAMIN D 400 [iU] .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) (.ALPHA.-TOCOPHEROL - UNII:H4N855PNZ1) .ALPHA.-TOCOPHEROL 30 [iU] THIAMINE (UNII: X66NSO3N35) (THIAMINE ION - UNII:4ABT0J945J) THIAMINE 3 mg RIBOFLAVIN (UNII: TLM2976OFR) (RIBOFLAVIN - UNII:TLM2976OFR) RIBOFLAVIN 3.4 mg NIACINAMIDE (UNII: 25X51I8RD4) (NIACINAMIDE - UNII:25X51I8RD4) NIACINAMIDE 20 mg PYRIDOXINE HYDROCHLORIDE (UNII: 68Y4CF58BV) (PYRIDOXINE - UNII:KV2JZ1BI6Z) PYRIDOXINE HYDROCHLORIDE 20 mg FOLIC ACID (UNII: 935E97BOY8) (FOLIC ACID - UNII:935E97BOY8) FOLIC ACID 1 mg POTASSIUM IODIDE (UNII: 1C4QK22F9J) (IODIDE ION - UNII:09G4I6V86Q) POTASSIUM IODIDE 150 mg ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 25 mg CUPRIC OXIDE (UNII: V1XJQ704R4) (CUPRIC CATION - UNII:8CBV67279L) CUPRIC CATION 2 mg DOCUSATE SODIUM (UNII: F05Q2T2JA0) (DOCUSATE - UNII:M7P27195AG) DOCUSATE SODIUM 50 mg Inactive Ingredients Ingredient Name Strength POVIDONES (UNII: FZ989GH94E) 132.2 mg CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) 30.6 mg TITANIUM DIOXIDE (UNII: 15FIX9V2JP) 30.5 mg NIACINAMIDE (UNII: 25X51I8RD4) 25 mg MAGNESIUM SILICATE (UNII: 9B9691B2N9) 5.1 mg THIAMINE MONONITRATE (UNII: 8K0I04919X) 3.9 mg SHELLAC (UNII: 46N107B71O) 2.6 mg POLYETHYLENE GLYCOLS (UNII: 3WJQ0SDW1A) 2 mg ETHYL VANILLIN (UNII: YC9ST449YJ) .7 mg VITAMIN A PALMITATE (UNII: 1D1K0N0VVC) .15 mg VITAMIN D (UNII: 9VU1KI44GP) .02 mg Product Characteristics Color BROWN (caramel) Score 2 pieces Shape CAPSULE Size 10mm Flavor Imprint Code CN;RX Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0178-0898-30 6 in 1 CARTON 1 5 in 1 BLISTER PACK 1 5 in 1 BLISTER PACK Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date Unapproved drug other 11/20/2007 Labeler - Mission Pharmacal Company (008117095) Registrant - Mission Pharmacal Company (927726893) Establishment Name Address ID/FEI Business Operations Mission Pharmacal Company 927726893 MANUFACTURE(0178-0898) Establishment Name Address ID/FEI Business Operations Robinson Pharma, Inc. 831560578 MANUFACTURE(0178-0898) Establishment Name Address ID/FEI Business Operations Accucaps Industries Limited 248441727 MANUFACTURE(0178-0898)