Label: FLUTAMIDE capsule
- NDC Code(s): 69097-915-12, 69097-915-91
- Packager: Cipla USA Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated November 29, 2018
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
BOXED WARNING
(What is this?)
BOXED WARNING
Hepatic Injury
There have been postmarketing reports of hospitalization and rarely death due to liver failure in patients taking flutamide. Evidence of hepatic injury included elevated serum transaminase levels, jaundice, hepatic encephalopathy and death related to acute hepatic failure. The hepatic injury was reversible after discontinuation of therapy in some patients. Approximately half of the reported cases occurred within the initial 3 months of treatment with flutamide.
Serum transaminase levels should be measured prior to starting treatment with flutamide. Flutamide is not recommended in patients whose ALT values exceed twice the upper limit of normal. Serum transaminase levels should then be measured monthly for the first 4 months of therapy, and periodically thereafter. Liver function tests also should be obtained at the first signs and symptoms suggestive of liver dysfunction, e.g., nausea, vomiting, abdominal pain, fatigue, anorexia, "flu-like" symptoms, hyperbilirubinuria, jaundice or right upper quadrant tenderness. If at any time, a patient has jaundice, or their ALT rises above 2 times the upper limit of normal, flutamide should be immediately discontinued with close follow-up of liver function tests until resolution.
-
DESCRIPTION
Flutamide capsules, USP contain flutamide, an acetanilid, nonsteroidal, orally active antiandrogen having the chemical name, α,α,α-Trifluoro-2-methyl-4'-nitro-m-propionotoluidide and has the following structural formula:
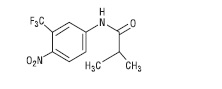
Flutamide is a buff to yellow powder. Each capsule, for oral administration, contains 125 mg flutamide and has the following inactive ingredients: black iron oxide, corn starch, D&C yellow #10 aluminum lake, FD&C blue #1 aluminum lake, FD&C blue #2 aluminum lake, FD&C red #40 aluminum lake, gelatin, lactose monohydrate, magnesium stearate, red iron oxide, sodium lauryl sulfate, titanium dioxide and yellow iron oxide.
-
CLINICAL PHARMACOLOGY
General
In animal studies, flutamide demonstrates potent antiandrogenic effects. It exerts its antiandrogenic action by inhibiting androgen uptake and/or by inhibiting nuclear binding of androgen in target tissues or both. Prostatic carcinoma is known to be androgen-sensitive and responds to treatment that counteracts the effect of androgen and/or removes the source of androgen, e.g., castration. Elevations of plasma testosterone and estradiol levels have been noted following flutamide administration.
Pharmacokinetics
Absorption
Analysis of plasma, urine, and feces following a single oral 200 mg dose of tritium-labeled flutamide to human volunteers showed that the drug is rapidly and completely absorbed. Following a single 250 mg oral dose to normal adult volunteers, the biologically active alpha-hydroxylated metabolite reaches maximum plasma concentrations in about 2 hours, indicating that it is rapidly formed from flutamide. Food has no effect on bioavailability of flutamide.
Distribution
In male rats neither flutamide nor any of its metabolites is preferentially accumulated in any tissue except the prostate after an oral 5 mg/kg dose of 14C-flutamide. Total drug levels were highest 6 hours after drug administration in all tissues. Levels declined at roughly similar rates to low levels at 18 hours. The major metabolite was present at higher concentrations than flutamide in all tissues studied. Following a single 250 mg oral dose to normal adult volunteers, low plasma levels of flutamide were detected. The plasma half-life for the alpha-hydroxylated metabolite of flutamide is about 6 hours. Flutamide, in vivo, at steady-state plasma concentrations of 24 to 78 ng/mL, is 94% to 96% bound to plasma proteins. The active metabolite of flutamide, in vivo, at steady-state plasma concentrations of 1556 to 2284 ng/mL, is 92% to 94% bound to plasma proteins.
Metabolism
The composition of plasma radioactivity, following a single 200 mg oral dose of tritium-labeled flutamide to normal adult volunteers, showed that flutamide is rapidly and extensively metabolized, with flutamide comprising only 2.5% of plasma radioactivity 1 hour after administration. At least 6 metabolites have been identified in plasma. The major plasma metabolite is a biologically active alpha-hydroxylated derivative which accounts for 23% of the plasma tritium 1 hour after drug administration. The major urinary metabolite is 2-amino-5-nitro-4-(trifluoromethyl)phenol.
Excretion
Flutamide and its metabolites are excreted mainly in the urine with only 4.2% of the dose excreted in the feces over 72 hours.
Plasma Pharmacokinetics of Flutamide and Hydroxyflutamide in Geriatric Volunteers (mean ± SD)
Single Dose
Steady State
Flutamide
Hydroxyflutamide
Flutamide
Hydroxyflutamide
Cmax (ng/mL)
25.2 ± 34.2
894 ± 406
113 ± 213
1629 ± 586
Elimination half-life (hr)
-
8.1 ± 1.3
7.8
9.6 ± 2.5
Tmax (hr)
1.9 ± 0.7
2.7 ± 1
1.3 ± 0.7
1.9 ± 0.6
Cmin (ng/mL)
-
-
-
673 ± 316
Special Populations
Geriatric
Following multiple oral dosing of 250 mg t.i.d. in normal geriatric volunteers, flutamide and its active metabolite approached steady-state plasma levels (based on pharmacokinetic simulations) after the fourth flutamide dose. The half-life of the active metabolite in geriatric volunteers after a single flutamide dose is about 8.1 hours and at steady state in 9.6 hours.
Race
There are no known alterations in flutamide absorption, distribution, metabolism, or excretion due to race.
Renal Impairment
Following a single 250 mg dose of flutamide administered to subjects with chronic renal insufficiency, there appeared to be no correlation between creatinine clearance and either Cmax or AUC of flutamide. Renal impairment did not have an effect on the Cmax or AUC of the biologically active alpha-hydroxylated metabolite of flutamide. In subjects with creatinine clearance of < 29 mL/min, the half-life of the active metabolite was slightly prolonged. Flutamide and its active metabolite were not well dialyzed. Dose adjustment in patients with chronic renal insufficiency is not warranted.
Clinical Studies
Flutamide has been demonstrated to interfere with testosterone at the cellular level. This can complement medical castration achieved with LHRH agonists which suppresses testicular androgen production by inhibiting luteinizing hormone secretion.
The effects of combination therapy have been evaluated in two studies. One study evaluated the effects of flutamide and an LHRH agonist as neoadjuvant therapy to radiation in stage B2-C prostatic carcinoma and the other study evaluated flutamide and an LHRH agonist as the sole therapy in stage D2 metastatic carcinoma.
Stage B2-C Prostatic Carcinoma
The effects of hormonal treatment combined with radiation were studied in 466 patients (231 flutamide capsules + goserelin acetate implant + radiation, 235 radiation alone) with bulky primary tumors confined to the prostate (stage B2) or extending beyond the capsule (stage C), with or without pelvic node involvement.
In this multicentered, controlled trial, administration of flutamide capsules (250 mg t.i.d.) and goserelin acetate (3.6 mg depot) prior to and during radiation was associated with a significantly lower rate of local failure compared to radiation alone (16% vs. 33% at 4 years, P < 0.001). The combination therapy also resulted in a trend toward reduction in the incidence of distant metastases (27% vs. 36% at 4 years, P = 0.058). Median disease-free survival was significantly increased in patients who received complete hormonal therapy combined with radiation as compared to those patients who received radiation alone (4.4 vs. 2.6 years, P < 0.001). Inclusion of normal PSA level as a criterion for disease-free survival also resulted in significantly increased median disease-free survival in patients receiving the combination therapy (2.7 vs. 1.5 years, P < 0.001).
Stage D2 Prostatic Carcinoma
To study the effects of combination therapy in metastatic disease, 617 patients (311 leuprolide + flutamide, 306 leuprolide + placebo) with previously untreated advanced prostatic carcinoma were enrolled in a large multicentered, controlled clinical trial.
Three and one-half years after the study was initiated, median survival had been reached. The median actuarial survival time was 34.9 months for patients treated with leuprolide and flutamide versus 27.9 months for patients treated with leuprolide alone. This 7 month increment represents a 25% improvement in overall survival time with the flutamide therapy. Analysis of progression-free survival showed a 2.6 month improvement in patients who received leuprolide plus flutamide, a 19% increment over leuprolide and placebo.
-
INDICATIONS AND USAGE
Flutamide capsules are indicated for use in combination with LHRH agonists for the management of locally confined Stage B2-C and Stage D2 metastatic carcinoma of the prostate.
- CONTRAINDICATIONS
-
WARNINGS
Hepatic Injury
Use in Women
Flutamide capsules are for use only in men. This product has no indication for women and should not be used in this population, particularly for nonserious or non life-threatening conditions.
Fetal Toxicity
Flutamide may cause fetal harm when administered to a pregnant woman (see Pregnancy).
Aniline Toxicity
One metabolite of flutamide is 4-nitro-3-fluoro-methylaniline. Several toxicities consistent with aniline exposure, including methemoglobinemia, hemolytic anemia and cholestatic jaundice have been observed in both animals and humans after flutamide administration. In patients susceptible to aniline toxicity (e.g., persons with glucose-6-phosphate dehydrogenase deficiency, hemoglobin M disease and smokers), monitoring of methemoglobin levels should be considered.
-
PRECAUTIONS
General
In clinical trials, gynecomastia occurred in 9% of patients receiving flutamide together with medical castration.
Information for Patients
Patients should be informed that flutamide capsules and the drug used for medical castration should be administered concomitantly, and that they should not interrupt their dosing or stop taking these medications without consulting their physician.
Laboratory Tests
Regular assessment of serum Prostate Specific Antigen (PSA) may be helpful in monitoring the patient's response. If PSA levels rise significantly and consistently during flutamide therapy the patient should be evaluated for clinical progression. For patients who have objective progression of disease together with an elevated PSA, a treatment-free period of antiandrogen while continuing the LHRH analogue may be considered.
Drug Interactions
Increases in prothrombin time have been noted in patients receiving long-term warfarin therapy after flutamide was initiated. Therefore close monitoring of prothrombin time is recommended and adjustment of the anticoagulant dose may be necessary when flutamide capsules are administered concomitantly with warfarin.
Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 1 year dietary study in male rats, interstitial cell adenomas of the testes were present in 49% to 75% of all treated rats (daily oral doses of 10, 30, and 50 mg/kg/day were administered). These produce plasma Cmax values that are 1, 2 to 3, and 4 fold, respectively, those associated with therapeutic doses in humans. In male rats similarly dosed for 1 year, tumors were still present after 1 year of a drug-free period, but the incidences were 43% to 47%. In a 2 year carcinogenicity study in male rats, daily administration of flutamide at these same doses produced testicular interstitial cell adenomas in 91% to 95% of all treated rats as opposed to 11% of untreated control rats. Mammary adenomas, adenocarcinomas, and fibroadenomas were increased in treated male rats at exposure levels that were 1 to 4 fold those observed during therapeutic dosing in humans. There are likewise reports of malignant breast neoplasms in men treated with flutamide capsules (see ADVERSE REACTIONS section).
Flutamide did not demonstrate DNA modifying activity in the Ames Salmonella/Microsome Mutagenesis Assay. Dominant lethal tests in rats were negative.
Reduced sperm counts were observed during a 6 week study of flutamide monotherapy in normal human volunteers.
Flutamide did not affect estrous cycles or interfere with the mating behavior of male and female rats when the drug was administered at 25 and 75 mg/kg/day prior to mating. Males treated with 150 mg/kg/day (30 times the minimum effective antiandrogenic dose) failed to mate; mating behavior returned to normal after dosing was stopped. Conception rates were decreased in all dosing groups. Suppression of spermatogenesis was observed in rats dosed for 52 weeks at approximately 3, 8, or 17 times the human dose and in dogs dosed for 78 weeks at 1.4, 2.3, and 3.7 times the human dose.
Animal Toxicology
Serious cardiac lesions were observed in 2/10 beagle dogs receiving 25 mg/kg/day for 78 weeks and 3/16 receiving 40 mg/kg/day for 2 to 4 years. These lesions, indicative of chronic injury and repair processes, included chronic myxomatous degeneration, intra-atrial fibrosis, myocardial acidophilic degeneration, vasculitis and perivasculitis. The doses at which these lesions occurred were associated with 2-hydroxyflutamide levels that were 1 to 12 fold greater than those observed in humans at therapeutic levels.
Pregnancy
Teratogenic Effects
There was decreased 24 hour survival in the offspring of pregnant rats treated with flutamide at doses of 30, 100, or 200 mg/kg/day (approximately 3, 9, and 19 times the human dose). A slight increase in minor variations in the development of the sternebrae and vertebrae was seen in fetuses of cats treated with two higher doses. Feminization of the male cats also occurred at the two higher dose levels. There was a decreased survival rate in the offspring of rabbits receiving the highest dose (15 mg/kg/day, equal to 1.4 times the human dose).
-
ADVERSE REACTIONS
Stage B2-C Prostatic Carcinoma
Treatment with flutamide capsules and the goserelin acetate implant did not add substantially to the toxicity of radiation treatment alone. The following adverse experiences were reported during a multicenter clinical trial comparing flutamide capsules + goserelin acetate implant + radiation versus radiation alone. The most frequently reported (greater than 5%) adverse experiences are listed below.
Adverse Events During Acute Radiation Therapy
(within first 90 days of radiation therapy)
Adverse Events During Late Radiation Phase
(after 90 days of radiation therapy)
(n = 231)
Goserelin Acetate Implant + Flutamide Capsules + Radiation
% All
(n = 235)
Radiation Only
% All
(n = 231)
Goserelin Acetate Implant + Flutamide Capsules + Radiation
% All
(n = 235)
Radiation Only
% All
Rectum/large bowel
80
76
Diarrhea
36
40
Bladder
58
60
Cystitis
16
16
Skin
37
37
Rectal bleeding
14
20
Proctitis
8
8
Hematuria
7
12
Stage D2 Metastatic Carcinoma
The following adverse experiences were reported during a multicenter clinical trial comparing flutamide capsules + LHRH agonist versus placebo + LHRH agonist.
The most frequently reported (greater than 5%) adverse experiences during treatment with flutamide capsules in combination with an LHRH agonist are listed in the table below. For comparison, adverse experiences seen with an LHRH agonist and placebo are also listed in the following table.
(n = 294)
Flutamide + LHRH agonist
% All
(n = 285)
Placebo + LHRH agonist
% All
Hot flashes
61
57
Loss of libido
36
31
Impotence
33
29
Diarrhea
12
4
Nausea/vomiting
11
10
Gynecomastia
9
11
Other
7
9
Other GI
6
4
Central Nervous System
CNS (drowsiness/confusion/depression/anxiety/nervousness) reactions occurred in 1% of patients
Hematopoietic System
Anemia occurred in 6%, leukopenia in 3%, and thrombocytopenia in 1% of patients
Other
Edema occurred in 4%, genitourinary and neuromuscular symptoms in 2%, and pulmonary symptoms in less than 1% of patients.
In addition, the following spontaneous adverse experiences have been reported during the marketing of flutamide: hemolytic anemia, macrocytic anemia, methemoglobinemia, sulfhemoglobinemia, photosensitivity reactions (including erythema, ulceration, bullous eruptions, and epidermal necrolysis), and urine discoloration. The urine was noted to change to an amber or yellow-green appearance which can be attributed to the flutamide and/or its metabolites. Also reported were cholestatic jaundice, hepatic encephalopathy, and hepatic necrosis. The hepatic conditions were often reversible after discontinuing therapy; however, there have been reports of death following severe hepatic injury associated with use of flutamide.
Malignant breast neoplasms have occurred rarely in male patients being treated with flutamide.
-
OVERDOSAGE
In animal studies with flutamide alone, signs of overdose included hypoactivity, piloerection, slow respiration, ataxia, and/or lacrimation, anorexia, tranquilization, emesis, and methemoglobinemia.
Clinical trials have been conducted with flutamide in doses up to 1500 mg per day for periods up to 36 weeks with no serious adverse effects reported. Those adverse reactions reported included gynecomastia, breast tenderness, and some increases in SGOT. The single dose of flutamide ordinarily associated with symptoms of overdose or considered to be life-threatening has not been established.
Flutamide is highly protein bound and is not cleared by hemodialysis. As in the management of overdosage with any drug, it should be borne in mind that multiple agents may have been taken. If vomiting does not occur spontaneously, it should be induced if the patient is alert. General supportive care, including frequent monitoring of the vital signs and close observation of the patient, is indicated.
- DOSAGE AND ADMINISTRATION
-
HOW SUPPLIED
Flutamide capsules, USP, 125 mg are available as light brown opaque body and light brown opaque cap linear printed with 'Cipla' logo in black ink on cap and '915' along with '125 mg' in black ink on body. They are supplied as follows:
Bottle of 180 NDC 69097-915-91
Bottle of 500 NDC 69097-915-12
Dispense in a tight, light-resistant container as defined in the USP, with a child-resistant closure (as required).
KEEP THIS AND ALL MEDICATIONS OUT OF THE REACH OF CHILDREN.
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature].
Manufactured by:
CIPLA Verna.
Goa, India
Manufactured for:
Cipla USA, Inc.
1560 Sawgrass Corporate Parkway.
Suite 130 Miami, FL 33323
Issued: 05/2017
-
PATIENT MEDICATION INFORMATION SECTION
Important information for patients taking flutamide capsules
Read this information carefully each time your prescription is refilled because there may be new information available. This summary does not tell you everything you need to know about flutamide therapy. Your doctor is the best source of information about your treatment. Ask your doctor about questions you have.
What is flutamide therapy?
Flutamide, in combination with other therapies, is a treatment option for men with some types of prostate cancer.
Prostate cancer results from the abnormal growth of prostate cells. Medical scientists do not know exactly what causes the abnormal cells, but age, environment and genetics are important factors. Male hormones ("androgens") cause the cancer to grow. The cancer growth can be slowed down by blocking the effect of androgens.
The flutamide product is used together with an injection called "LHRH agonist," as a combined treatment called "total androgen blockade." The goal of this treatment is to reduce androgen levels and to block the effect of androgen on the tumor. The LHRH agonist reduces androgen levels. Flutamide therapy blocks the effect of androgen on the tumor.
Who should not take the flutamide product?
You should not take flutamide if you have liver problems or if you are allergic to it. Flutamide is for use only in men; therefore women should not take flutamide.
Are there important risks I should know about flutamide therapy?
Some men taking the flutamide product had liver injury and needed to be hospitalized. In rare cases, men died because of liver failure while they were taking flutamide. In about half of these cases, the liver failure occurred in the first 3 months that they were taking flutamide.
Because the flutamide product may cause liver failure, it is very important that you have all blood tests recommended by your doctor. These tests help identify whether you are having liver problems. A recommended schedule for these blood tests is:
- before starting flutamide treatment
- every month for the first 4 months of therapy
- periodically after the first 4 months
In addition, you should call your doctor right away if you have any of the following signs or symptoms:
- loss of appetite
- nausea and vomiting
- stomach or abdominal pain
- fatigue (feeling extremely tired)
- flu-like symptoms (muscle aches, soreness)
- brown urine
- jaundice (yellowing of the skin or whites of the eyes)
- drink plenty of fluids
- reduce your intake of dairy products (for example, milk, cheese, yogurt)
- increase your intake of whole grains, fruits, and vegetables
- stop laxative use
- take non-prescription anti-diarrheal medicines
If your diarrhea continues or it becomes severe, contact your doctor right away.
Are there any other lab tests my doctor will be performing?
Your doctor may perform other regular tests (such as the PSA blood test) to ensure that your body is responding to treatment. Ask your doctor if you have any questions about how your flutamide therapy is being monitored.
Where can I get further support?
Please ask your doctor about any questions concerning prostate cancer or flutamide therapy, or you can also ask for a more detailed leaflet that is written for healthcare professionals.
Manufactured by:
CIPLA Verna.
Goa, India
Manufactured for:
Cipla USA, Inc.
1560 Sawgrass Corporate Parkway.
Suite 130 Miami, FL 33323
Issued: 05/2017
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
FLUTAMIDE
flutamide capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:69097-915 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength FLUTAMIDE (UNII: 76W6J0943E) (FLUTAMIDE - UNII:76W6J0943E) FLUTAMIDE 125 mg Inactive Ingredients Ingredient Name Strength FERROSOFERRIC OXIDE (UNII: XM0M87F357) STARCH, CORN (UNII: O8232NY3SJ) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FD&C BLUE NO. 2 (UNII: L06K8R7DQK) FD&C RED NO. 40 (UNII: WZB9127XOA) ALUMINUM OXIDE (UNII: LMI26O6933) GELATIN (UNII: 2G86QN327L) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) FERRIC OXIDE RED (UNII: 1K09F3G675) SODIUM LAURYL SULFATE (UNII: 368GB5141J) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) Product Characteristics Color BROWN (LIGHT) Score no score Shape CAPSULE Size 22mm Flavor Imprint Code CIPLA;915;125 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69097-915-91 180 in 1 BOTTLE; Type 0: Not a Combination Product 09/21/2016 2 NDC:69097-915-12 500 in 1 BOTTLE; Type 0: Not a Combination Product 09/21/2016 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA075780 09/21/2016 Labeler - Cipla USA Inc. (078719707) Registrant - Cipla USA Inc. (078719707) Establishment Name Address ID/FEI Business Operations Cipla Ltd.- Goa 650072015 MANUFACTURE(69097-915) Establishment Name Address ID/FEI Business Operations Cipla Limited, Bangalore 915154892 ANALYSIS(69097-915) , API MANUFACTURE(69097-915)




