Label: YETI POT- SCHNOZ WASH- nasal cleaning salt granule, for solution
-
Contains inactivated NDC Code(s)
NDC Code(s): 70708-786-00 - Packager: HEALTHLAND LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated May 7, 2016
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Warnings
- KEEP OUT OF REACH OF CHILDREN
- Other Information
- Uses
- ACTIVE INGREDIENT
- Directions
-
EASY TO USE INSTRUCTIONS
STEP ONE: Mix one packet of Schnoz Wash with distilled water up to the 240 ml mark in the Yeti Pot. Replace the lid and shake well so that the solution mixes witht he water.
STEP TWO : Tilt your head and insert the Yeti Pot spout gently into the raised nostril, creating a seal between the pot and nostril.Raise the pot slowly to develop a steady flow through the upper nostril and out the lower nostril.
STEP THREE: During the process breathe through your mouth. Upon completion , exhale firmly several times to clear the nasal passages.Reverse the tilt of your head and repeat the other process on the other side.
- Inactive Ingredient
- Yeti Pot -Schnoz wash label
-
INGREDIENTS AND APPEARANCE
YETI POT- SCHNOZ WASH
nasal cleaning salt granule, for solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:70708-786 Route of Administration NASAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SODIUM BICARBONATE (UNII: 8MDF5V39QO) (BICARBONATE ION - UNII:HN1ZRA3Q20) SODIUM BICARBONATE 515 mg in 1 mg SODIUM CHLORIDE (UNII: 451W47IQ8X) (SODIUM CATION - UNII:LYR4M0NH37) SODIUM CHLORIDE 1685 mg in 1 mg Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) 240 mg in 1 mg Product Characteristics Color white (white crystalline granule powder) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70708-786-00 2200 mg in 1 PACKET; Type 0: Not a Combination Product 05/01/2016 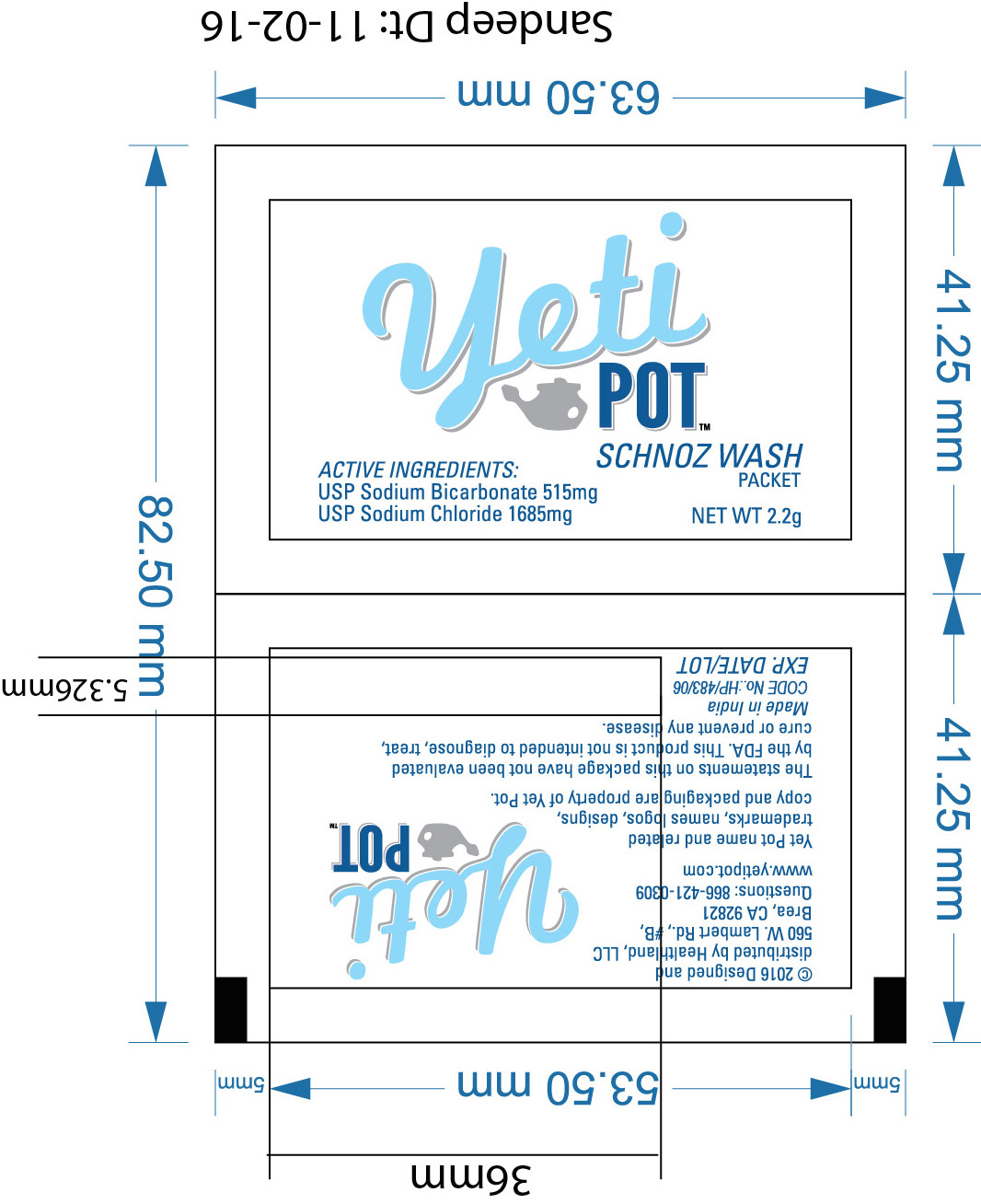
Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 05/01/2016 Labeler - HEALTHLAND LLC (079671169)


