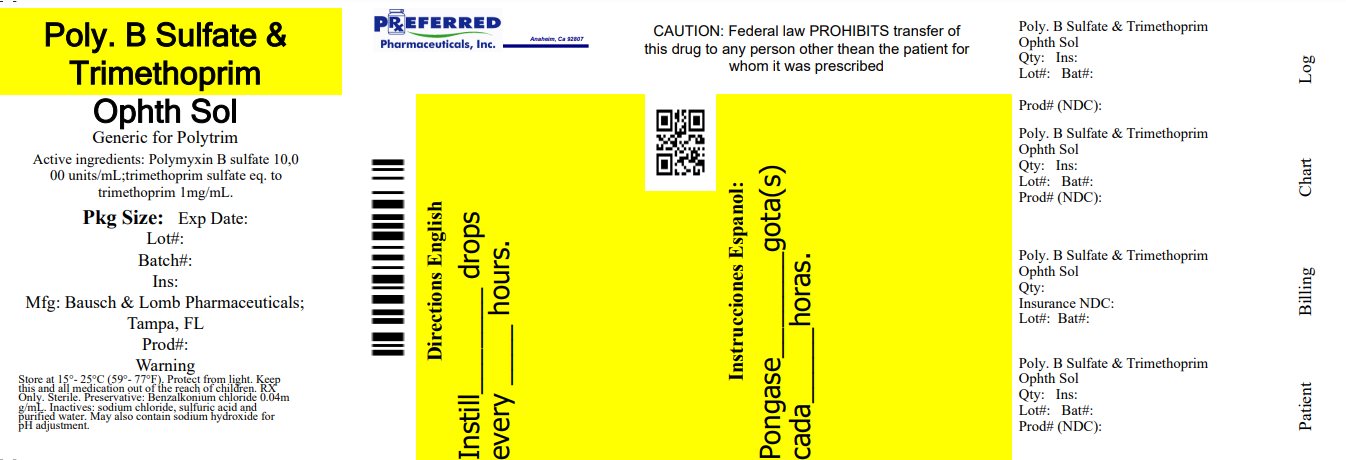
Label: POLYMYXIN B SULFATE AND TRIMETHOPRIM- polymyxin b sulfate and trimethoprim sulfate solution/ drops
- NDC Code(s): 68788-8152-1
- Packager: Preferred Pharmaceuticals Inc.
- This is a repackaged label.
- Source NDC Code(s): 24208-315
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated September 1, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
-
DESCRIPTION
Polymyxin B sulfate and trimethoprim ophthalmic solution, USP is a sterile antimicrobial solution for topical ophthalmic use. It has pH of 4.0 to 5.5 and osmolality of 270 to 310 mOsm/kg.
Chemical Names:
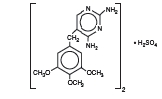
Trimethoprim sulfate, 2,4-diamino-5-(3,4,5-trimethoxybenzyl) pyrimidine sulfate, is a white, odorless, crystalline powder with a molecular weight of 678.72 and the following structural formula:C28H38N8O10S Mol. Wt. 678.72
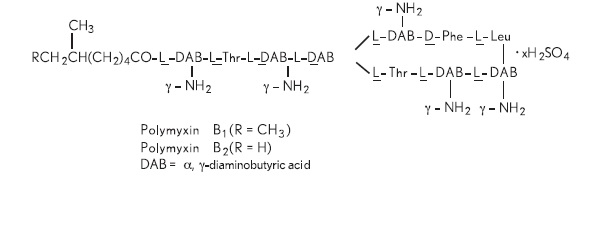
Polymyxin B sulfate is the sulfate salt of polymyxin B1 and B2 which are produced by the growth of Bacillus polymyxa (Prazmowski) Migula (Fam. Bacillaceae). It has a potency of not less than 6,000 polymyxin B units per mg, calculated on an anhydrous basis. The structural formula is:
Each mL contains: Actives: polymyxin B sulfate equal to 10,000 polymyxin B units, trimethoprim sulfate (equivalent to trimethoprim 1 mg); Inactives: purified water, sodium chloride. Sulfuric acid and, if necessary, sodium hydroxide may be added to adjust pH (4.0 – 5.5). Preservative: benzalkonium chloride 0.004%
-
CLINICAL PHARMACOLOGY
Trimethoprim is a synthetic antibacterial drug active against a wide variety of aerobic gram-positive and gram-negative ophthalmic pathogens. Trimethoprim blocks the production of tetrahydrofolic acid from dihydrofolic acid by binding to and reversibly inhibiting the enzyme dihydrofolate reductase. This binding is stronger for the bacterial enzyme than for the corresponding mammalian enzyme and therefore selectively interferes with bacterial biosynthesis of nucleic acids and proteins.
Polymyxin B, a cyclic lipopeptide antibiotic, is bactericidal for a variety of gram-negative organisms, especially Pseudomonas aeruginosa. It increases the permeability of the bacterial cell membrane by interacting with the phospholipid components of the membrane.
Blood samples were obtained from 11 human volunteers at 20 minutes, 1 hour and 3 hours following instillation in the eye of 2 drops of ophthalmic solution containing 1 mg trimethoprim and 10,000 units polymyxin B per mL. Peak serum concentrations were approximately 0.03 mcg/mL trimethoprim and 1 unit/mL polymyxin B.
Microbiology
In vitrostudies have demonstrated that the anti-infective components of trimethoprim sulfate and polymyxin B sulfate ophthalmic solution are active against the following bacterial pathogens that are capable of causing external infections of the eye:Trimethoprim: Staphylococcus aureus and Staphylococcus epidermidis, Streptococcus pyogenes, Streptococcus faecalis, Streptococcus pneumoniae, Haemophilus influenzae, Haemophilus aegyptius, Escherichia coli, Klebsiella pneumoniae, Proteus mirabilis (indole-negative), Proteus vulgaris (indole-positive), Enterobacter aerogenes and Serratia marcescens.
Polymyxin B: Pseudomonas aeruginosa, Escherichia coli, Klebsiella pneumoniae, Enterobacter aerogenes and Haemophilus influenzae.
- INDICATIONS AND USAGE
- CONTRAINDICATIONS
-
PRECAUTIONS
General
As with other antimicrobial preparations, prolonged use may result in overgrowth of nonsusceptible organisms, including fungi. If superinfection occurs, appropriate therapy should be initiated.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis: Long-term studies in animals to evaluate carcinogenic potential have not been conducted with polymyxin B sulfate or trimethoprim.
Mutagenesis: Trimethoprim was demonstrated to be non-mutagenic in the Ames assay. In studies at two laboratories no chromosomal damage was detected in cultured Chinese hamster ovary cells at concentrations approximately 500 times human plasma levels after oral administration; at concentrations approximately 1,000 times human plasma levels after oral administration in these same cells, a low level of chromosomal damage was induced at one of the laboratories. Studies to evaluate mutagenic potential have not been conducted with polymyxin B sulfate.
Impairment of Fertility: Polymyxin B sulfate has been reported to impair the motility of equine sperm, but its effects on male or female fertility are unknown.
No adverse effects on fertility or general reproductive performance were observed in rats given trimethoprim in oral dosages as high as 70 mg/kg/day for males and 14 mg/kg/day for females.
Pregnancy
Teratogenic Effects
Animal reproduction studies have not been conducted with polymyxin B sulfate. It is not known whether polymyxin B sulfate can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity.
Trimethoprim has been shown to be teratogenic in the rat when given in oral doses 40 times the human dose. In some rabbit studies, the overall increase in fetal loss (dead and resorbed and malformed conceptuses) was associated with oral doses 6 times the human therapeutic dose.
While there are no large well-controlled studies on the use of trimethoprim in pregnant women, Brumfitt and Pursell, in a retrospective study, reported the outcome of 186 pregnancies during which the mother received either placebo or oral trimethoprim in combination with sulfamethoxazole. The incidence of congenital abnormalities was 4.5% (3 of 66) in those who received placebo and 3.3% (4 of 120) in those receiving trimethoprim and sulfamethoxazole. There were no abnormalities in the 10 children whose mothers received the drug during the first trimester. In a separate survey, Brumfitt and Pursell also found no congenital abnormalities in 35 children whose mothers had received oral trimethoprim and sulfamethoxazole at the time of conception or shortly thereafter.
Because trimethoprim may interfere with folic acid metabolism, trimethoprim should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when polymyxin B sulfate and trimethoprim ophthalmic solution is administered to a nursing woman.
-
ADVERSE REACTIONS
The most frequent adverse reaction to polymyxin B sulfate and trimethoprim ophthalmic solution is local irritation consisting of increased redness, burning, stinging, and/or itching. This may occur on instillation, within 48 hours, or at any time with extended use. There are also multiple reports of hypersensitivity reactions consisting of lid edema, itching, increased redness, tearing, and/or circumocular rash. Photosensitivity has been reported in patients taking oral trimethoprim.
To report SUSPECTED ADVERSE REACTIONS, contact Bausch & Lomb Incorporated at 1-800-553-5340 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
- DOSAGE AND ADMINISTRATION
-
HOW SUPPLIED
Polymyxin B sulfate and trimethoprim ophthalmic solution, USP* containing 10,000 polymyxin B units and 1 mg trimethoprim per mL, is supplied in a plastic bottle with a controlled drop tip and a natural cap in the following size:
NDC 68788-8152-1 10 mL
DO NOT USE IF IMPRINTED NECKBAND IS NOT INTACT.
Storage:
Store at 15°C to 25°C (59°F to 77°F). PROTECT FROM LIGHT.
*Does not meet USP packaging specification for light resistance.
RETAIN IN CARTON UNTIL TIME OF USE.
Distributed by:
Bausch & Lomb Americas Inc.
Bridgewater, NJ 08807 USAManufactured by:
Bausch & Lomb Incorporated
Tampa, FL 33637 USA
© 2023 Bausch & Lomb Incorporated or its affiliatesRelabeled By: Preferred Pharmaceuticals Inc.
Revised: June 2023
9117804 (Folded)
9117904 (Flat) - Principal Display Panel
-
INGREDIENTS AND APPEARANCE
POLYMYXIN B SULFATE AND TRIMETHOPRIM
polymyxin b sulfate and trimethoprim sulfate solution/ dropsProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:68788-8152(NDC:24208-315) Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength POLYMYXIN B SULFATE (UNII: 19371312D4) (POLYMYXIN B - UNII:J2VZ07J96K) POLYMYXIN B 10000 [USP'U] in 1 mL TRIMETHOPRIM SULFATE (UNII: E377MF8EQ8) (TRIMETHOPRIM - UNII:AN164J8Y0X) TRIMETHOPRIM 1 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) SODIUM CHLORIDE (UNII: 451W47IQ8X) SULFURIC ACID (UNII: O40UQP6WCF) SODIUM HYDROXIDE (UNII: 55X04QC32I) BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68788-8152-1 1 in 1 CARTON 03/14/2022 1 10 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA064120 03/14/2022 Labeler - Preferred Pharmaceuticals Inc. (791119022) Registrant - Preferred Pharmaceuticals Inc. (791119022) Establishment Name Address ID/FEI Business Operations Preferred Pharmaceuticals Inc. 791119022 RELABEL(68788-8152)