Label: SINUS CONGESTION PE- guaifenesin and phenylephrine hcl tablet, film coated
-
Contains inactivated NDC Code(s)
NDC Code(s): 68788-0845-2, 68788-0845-3, 68788-0845-6 - Packager: Preferred Pharmaceuticals, Inc.
- This is a repackaged label.
- Source NDC Code(s): 0904-5792
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated November 1, 2019
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredients (in each immediate-release tablet)
- Purpose
-
Uses
- •
- helps loosen phlegm (mucus) and thin bronchial secretions to rid the bronchial passageways of bothersome mucus and make coughs more productive
- •
- temporarily relieves nasal congestion due to the common cold, hay fever or other upper respiratory allergies
- •
- promotes nasal and/or sinus drainage
- •
- temporarily relieves sinus congestion and pressure
-
Warnings
Do not use
if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have
- •
- heart disease
- •
- high blood pressure
- •
- thyroid disease
- •
- diabetes
- •
- difficulty in urination due to enlargement of the prostate gland
- •
- persistent or chronic cough such as occurs with smoking, asthma, chronic bronchitis, or emphysema
- •
- cough accompanied by too much phlegm (mucus)
Stop use and ask a doctor if
- •
- nervousness, dizziness, or sleeplessness occur
- •
- symptoms do not improve within 7 days or are accompanied by fever
- •
- cough persists more than 7 days, tends to recur, or is accompanied by a fever, rash, or persistent headache. These could be signs of a serious condition.
- Directions
- Other information
- Inactive ingredients
-
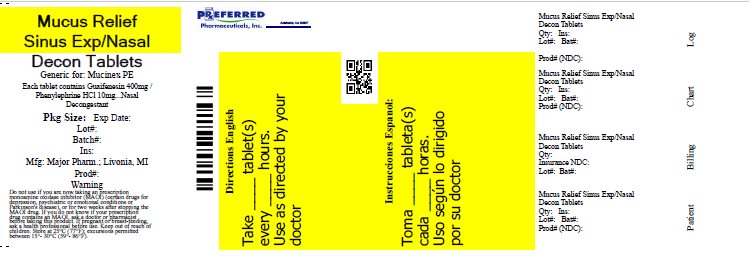
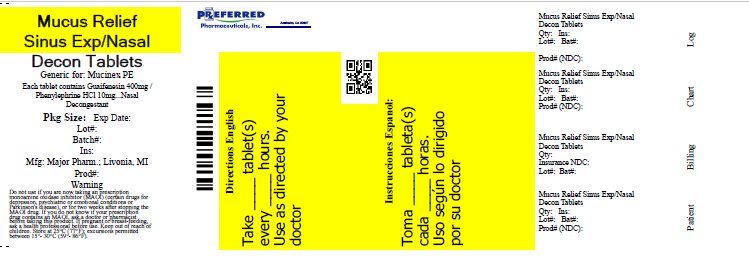
Principal Display Panel
MAJOR®
NDC 0904-5792-46
Mucus Relief
SinusGuaifenesin 400 mg /
Phenylephrine HCl 10 mgExpectorant / Nasal Decongestant
- •
- Alleviates Chest Congestion
- •
- Relieves Nasal/Sinus Congestion
- •
- Does Not Contain Pseudoephedrine
30 Tablets
Immediate
ReleaseTAMPER EVIDENT: DO NOT USE IF IMPRINTED SAFETY SEAL UNDER CAP IS BROKEN OR MISSING
50844 REV0118E54201
Distributed by MAJOR PHARMACEUTICALS
17177 N Laurel Park Drive, Suite 233
Livonia, MI 48152 USA
REV. 02/18 M-17 Re-order No. 006474
-
INGREDIENTS AND APPEARANCE
SINUS CONGESTION PE
guaifenesin and phenylephrine hcl tablet, film coatedProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:68788-0845(NDC:0904-5792) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength GUAIFENESIN (UNII: 495W7451VQ) (GUAIFENESIN - UNII:495W7451VQ) GUAIFENESIN 400 mg PHENYLEPHRINE HYDROCHLORIDE (UNII: 04JA59TNSJ) (PHENYLEPHRINE - UNII:1WS297W6MV) PHENYLEPHRINE HYDROCHLORIDE 10 mg Inactive Ingredients Ingredient Name Strength HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) MAGNESIUM STEARATE (UNII: 70097M6I30) MALTODEXTRIN (UNII: 7CVR7L4A2D) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) STEARIC ACID (UNII: 4ELV7Z65AP) Product Characteristics Color WHITE Score no score Shape OVAL Size 16mm Flavor Imprint Code 44;542 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68788-0845-2 20 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 05/14/2014 2 NDC:68788-0845-3 30 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 05/14/2014 3 NDC:68788-0845-6 60 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 05/14/2014 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part341 05/14/2014 Labeler - Preferred Pharmaceuticals, Inc. (791119022) Registrant - Preferred Pharmaceuticals, Inc. (791119022) Establishment Name Address ID/FEI Business Operations Preferred Pharmaceuticals, Inc. 791119022 REPACK(68788-0845)