Label: OMEPRAZOLE tablet, delayed release
-
NDC Code(s):
0363-0915-01,
0363-0915-03,
0363-0915-30,
0363-0915-55, view more0363-0915-74
- Packager: Walgreen Company
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated January 13, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
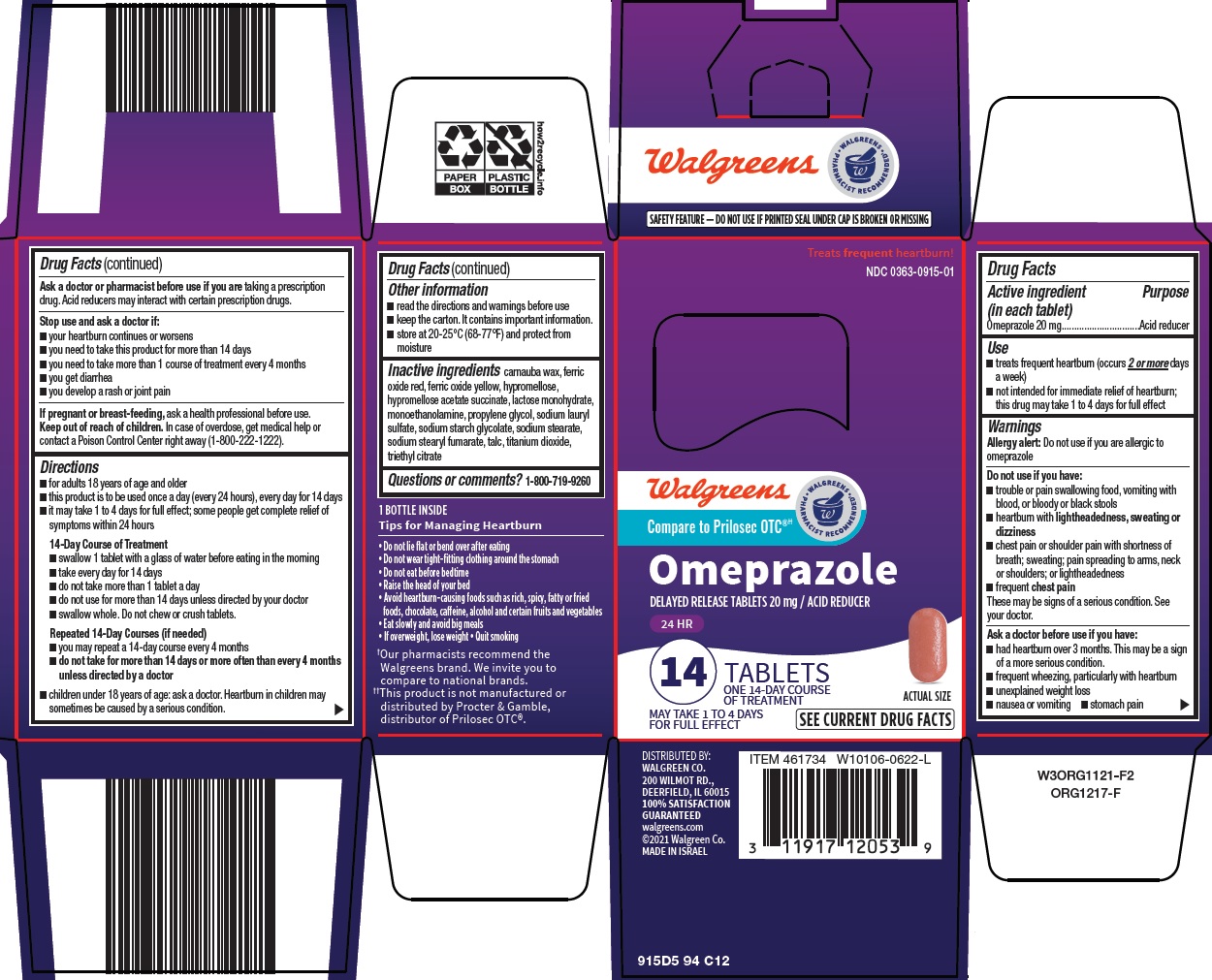
- Active ingredient (in each tablet)
- Purpose
- Use
-
Warnings
Allergy alert: Do not use if you are allergic to omeprazole
Do not use if you have:
- •
- trouble or pain swallowing food, vomiting with blood, or bloody or black stools
- •
- heartburn with lightheadedness, sweating or dizziness
- •
- chest pain or shoulder pain with shortness of breath; sweating; pain spreading to arms, neck or shoulders; or lightheadedness
- •
- frequent chest pain
These may be signs of a serious condition. See your doctor.
Ask a doctor before use if you have:
- •
- had heartburn over 3 months. This may be a sign of a more serious condition.
- •
- frequent wheezing, particularly with heartburn
- •
- unexplained weight loss
- •
- nausea or vomiting
- •
- stomach pain
Ask a doctor or pharmacist before use if you are
taking a prescription drug. Acid reducers may interact with certain prescription drugs.
-
Directions
- •
- for adults 18 years of age and older
- •
- this product is to be used once a day (every 24 hours), every day for 14 days
- •
- it may take 1 to 4 days for full effect; some people get complete relief of symptoms within 24 hours
14-Day Course of Treatment
- •
- swallow 1 tablet with a glass of water before eating in the morning
- •
- take every day for 14 days
- •
- do not take more than 1 tablet a day
- •
- do not use for more than 14 days unless directed by your doctor
- •
- swallow whole. Do not chew or crush tablets
Repeated 14-Day Courses (if needed)
- •
- you may repeat a 14-day course every 4 months
- •
- do not take for more than 14 days or more often than every 4 months unless directed by a doctor
- •
- children under 18 years of age: ask a doctor. Heartburn in children may sometimes be caused by a serious condition.
- Other information
- Inactive ingredients
- Questions or comments?
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
OMEPRAZOLE
omeprazole tablet, delayed releaseProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:0363-0915 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OMEPRAZOLE (UNII: KG60484QX9) (OMEPRAZOLE - UNII:KG60484QX9) OMEPRAZOLE 20 mg Inactive Ingredients Ingredient Name Strength CARNAUBA WAX (UNII: R12CBM0EIZ) FERRIC OXIDE RED (UNII: 1K09F3G675) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MONOETHANOLAMINE (UNII: 5KV86114PT) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SODIUM LAURYL SULFATE (UNII: 368GB5141J) SODIUM STEARATE (UNII: QU7E2XA9TG) SODIUM STEARYL FUMARATE (UNII: 7CV7WJK4UI) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) TRIETHYL CITRATE (UNII: 8Z96QXD6UM) Product Characteristics Color BROWN Score no score Shape OVAL Size 12mm Flavor Imprint Code 20 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0363-0915-30 2 in 1 CARTON 02/27/2008 1 14 in 1 CARTON 1 1 in 1 BLISTER PACK; Type 0: Not a Combination Product 2 NDC:0363-0915-55 3 in 1 CARTON 02/27/2008 2 14 in 1 CARTON 2 1 in 1 BLISTER PACK; Type 0: Not a Combination Product 3 NDC:0363-0915-74 14 in 1 CARTON 02/27/2008 04/01/2013 3 1 in 1 BLISTER PACK; Type 0: Not a Combination Product 4 NDC:0363-0915-01 1 in 1 CARTON 10/06/2010 4 14 in 1 BOTTLE; Type 0: Not a Combination Product 5 NDC:0363-0915-03 3 in 1 CARTON 03/25/2011 5 14 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA022032 02/27/2008 Labeler - Walgreen Company (008965063)