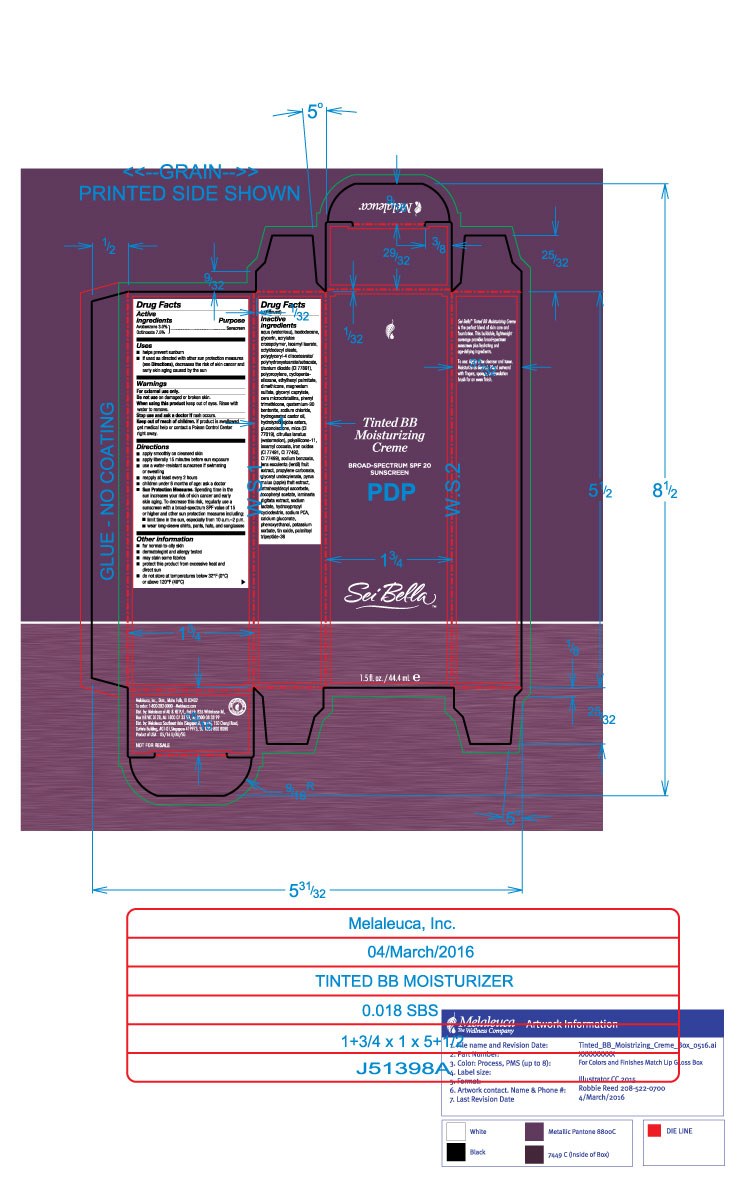
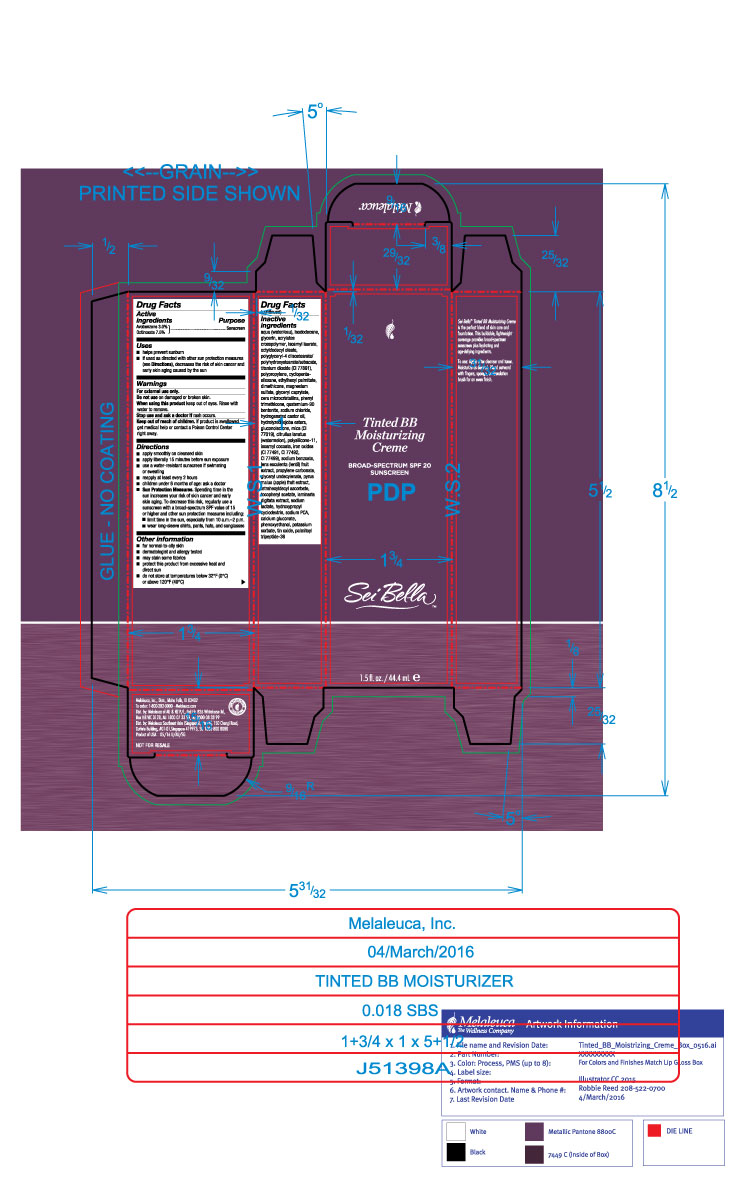
Label: SEI BELLA TINTED BB MOISTURIZING CREME BROADSPECTRUM SPF 20 BLUSH 140 C- avobenzone and octinoxate lotion
-
Contains inactivated NDC Code(s)
NDC Code(s): 54473-261-01 - Packager: Melaleuca Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated May 6, 2016
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredients
- Purpose
- Uses
- Warnings
-
Directions
Directions
- apply smoothly on cleased skin
- apply liberally 15 minutes before sun exposure
- use a water-resistant sunscreen if swimming or sweating
- reapply at least every 2 hours
- children under 6 months of age: ask a doctor
- Sun Protection Measures: Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a broad-spectrum SPF value of 15 or higher and other sun protection measures including:
- limit time in the sun, especially from 10a.m.-2p.m
- wear long-sleeve shirts, pants, hats, and sunglasses
- Other Information
-
Inactive ingredients
aqua (water/eau), isododecane, glycerin, acrylates crosspolymer, isoamyl laurate, octyldodecyl oleate, polyglyceryl-4 diisostearate/polyhydroxustearate/sebacate, titanium dioxide (CI 77891), polypropylene, cyclopentasiloxane, ethylhexyl palmitate, dimethicone, magnesium sulfate, glyceryl caprylate, microcrystalline wax, phenyl trimethicone, quaternium-90 bentonite, sodium chloride, hydrogenated castor oil, hydrolyzed jojoba esters, gluconolactone, mica (CI 77019), citrullur lanatus (watermelon), polysilicone-11, isoamyl cocoate, iron oxides (CI 77491, CI 77492, CI 77499), sodium benzoate, lens esculenta (lentil) fruit extract, propylene carbonate, glyceryl undecylenate, pyrus malus (apple) fruit extract, tetrahexyldecyl ascorbate, tocopheryl acetate, laminaria digitata extract, sodium lactate, hydroxypropyl cyclodextrin, sodium PCA, calcium gluconate, phenoxyethanol, potassium sorbate, tin oxide, palmitoyl tripeptide-38
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
SEI BELLA TINTED BB MOISTURIZING CREME BROADSPECTRUM SPF 20 BLUSH 140 C
avobenzone and octinoxate lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:54473-261 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 1.389 g in 44.4 mL OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 3.473 g in 44.4 mL Inactive Ingredients Ingredient Name Strength LENTIL (UNII: 6O38V6B52O) PROPYLENE CARBONATE (UNII: 8D08K3S51E) TETRAHEXYLDECYL ASCORBATE (UNII: 9LBV3F07AZ) ALPHA-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) SODIUM LACTATE (UNII: TU7HW0W0QT) WATER (UNII: 059QF0KO0R) ISODODECANE (UNII: A8289P68Y2) 1,1'-((1-METHYL-1,2-ETHANEDIYL)2-PROPENOIC ACID, BIS(OXY(1-METHYL-2,1-ETHANEDIYL))) ESTER (UNII: 2O70KL79K2) ISOAMYL LAURATE (UNII: M1SLX00M3M) ETHYLHEXYL PALMITATE (UNII: 2865993309) MAGNESIUM SULFATE (UNII: DE08037SAB) MICROCRYSTALLINE WAX (UNII: XOF597Q3KY) PHENYL TRIMETHICONE (UNII: DR0K5NOJ4R) HYDROGENATED CASTOR OIL (UNII: ZF94AP8MEY) STANNIC OXIDE (UNII: KM7N50LOS6) CITRULLUS LANATUS VAR. LANATUS WHOLE (UNII: 3J5I6254YO) GLYCERYL MONOUNDECYLENATE (UNII: IK8A76Q5G8) GLYCERIN (UNII: PDC6A3C0OX) OCTYLDODECYL OLEATE (UNII: MCA43PK7MH) ISOLAN (UNII: K7716TIH63) CYCLOMETHICONE (UNII: NMQ347994Z) ISOAMYL COCOATE (UNII: 14OG46E98E) SODIUM BENZOATE (UNII: OJ245FE5EU) GLUCONOLACTONE (UNII: WQ29KQ9POT) PHENOXYETHANOL (UNII: HIE492ZZ3T) MICA (UNII: V8A1AW0880) APPLE FRUIT OIL (UNII: 9NT987I3A8) LAMINARIA DIGITATA (UNII: 15E7C67EE8) DIMETHICONE (UNII: 92RU3N3Y1O) GLYCERYL CAPRYLATE (UNII: TM2TZD4G4A) SODIUM CHLORIDE (UNII: 451W47IQ8X) HYDROLYZED JOJOBA ESTERS (ACID FORM) (UNII: UDR641JW8W) CALCIUM GLUCONATE (UNII: SQE6VB453K) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:54473-261-01 1 in 1 BOX 09/01/2016 1 44.4 mL in 1 TUBE; Type 0: Not a Combination Product 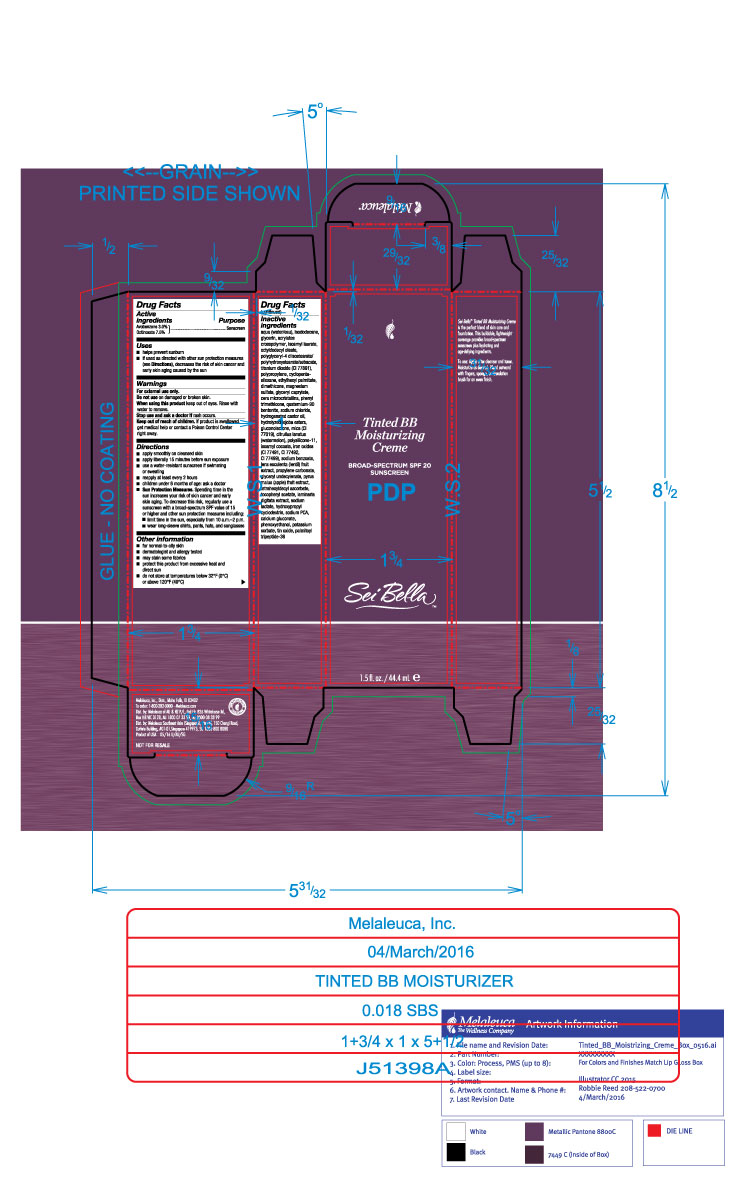
Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 09/01/2016 Labeler - Melaleuca Inc (139760102) Registrant - Melaleuca Inc (079287544) Establishment Name Address ID/FEI Business Operations Northwest Cosmetic Labs 929572014 manufacture(54473-261)